PHYTOCHEMICAL, IN VITRO ANTIOXIDANT AND ANTIBACTERIAL ACTIVITIES OF ARGYREIA PILOSA WIGHT & ARN. (WHOLE PLANT)
HTML Full TextPHYTOCHEMICAL, IN-VITRO ANTIOXIDANT AND ANTIBACTERIAL ACTIVITIES OF ARGYREIA PILOSA WIGHT & ARN. (WHOLE PLANT)
D. S. N. B. K. Prasanth * 1, Atla Srinivasa Rao 2 and Rajendra Prasad Yejella 3
Department of Pharmacy 1, JNTUK, Kakinada - 533003, Andhra Pradesh, India.
Shri Vishnu College of Pharmacy 2, Bhimavaram - 534202, Andhra Pradesh, India.
University College of Pharmaceutical Sciences 3, Visakhapatnam - 530003, Andhra Pradesh, India.
ABSTRACT: Background: The whole plant of Argyreia pilosa Wight & Arn. (Convolvulaceae) continuously to be utilized in indigenous Indian systems as a remedy for cough, quinsy and applied externally in case of itch, eczema and other skin troubles, antidiabetic, antiphlogistic, rheumatism and reduce burning sensation. Still, there were constrained phytochemical or biological studies on the whole plant of A. pilosa, as well as no studies which align using its conventional medicinal uses. Aim: This study aimed to determine the total tannin and flavonoids contents, antimicrobial and antioxidant activity of two extracts of the whole plant of A. pilosa and compare them to find out the better extract for upcoming studies. Materials and Methods: The whole plant material was subjected to cold maceration with ethyl acetate and methanol to obtain ethyl acetate extract (APEE) and methanolic extract (APME). Phytochemical constituents of these extracts were determined as per standard procedure. Total phenolics had been estimated by the Folin–Ciocalteu colorimetric method using tannic acid as standard. Total flavonoids had been estimated by aluminium chloride assay using quercetin as standard, and the antioxidant capacity was determined by the 2,2-diphenyl-1-picrylhydrazyl radical scavenging assay, hydrogen peroxide assay and reducing capacity. The antimicrobial activity was studied with the agar diffusion method, and minimal inhibitory concentrations (MIC) of these extracts were determined. Results: APEE has a better yield than APME. The phytochemical constituents present in both APEE and APME have revealed the presence of flavonoids, alkaloids, phenols, tannins, glycosides, amino acids and proteins except for steroids and acid compounds which are only present in APME. Total tannin content and total flavonoids content of APEE is more than in APME. Both REE and RME have antioxidant and antibacterial potency. Statistically, there is no significant difference between the antioxidant potency of APEE and APME. But APEE and APME are statistically different from each other in terms of their antibacterial strength, APEE being better than APME in this case.
| Keywords: |
Argyreia pilosa, Antioxidant, Tannins, Flavonoids, Antibacterial and Phytochemical analysis
INTRODUCTION: Phytochemicals tend to be widely available at diverse ranges in several medicinal plants. Most of the plants utilized in conventional drugs are efficient for different disorders triggered by oxidative stress and microbial infections.
Studies have revealed that medicinal plants show antioxidant along with antimicrobial activity 1. Oxygen, the most crucial element for the endurance of living organisms is exceptionally reactive and may do severe harm to healthy cells of the body as free radical. Oxidation generates free radicals that begin sequence reaction within the cell that triggers harm to and also fatality of cell 2.
An antioxidant ends these type of radical responses by eliminating free radical intermediates, that can causes aging and disorders such as atherosclerosis, cancer, cardiovascular diseases and CNS disorders 3. Therefore, play an important role in prohibiting or lagging the oxidation at the cellular level 4. Man-made antioxidants, i.e., butylated hydroxytoluene, butylated hydroxyanisole, and tert-butyl hydro-quinone being examined all over the world for their possible health risks and have been found to promote hepatic damage and leading to cancer to laboratory animals 5. In contrast, the herbs prove to have less or no side effects, 3 thus channelizing the interest towards the nature for antioxidant potency.
As a result of developing resistance to antibiotics of several microbes, plant extracts and plant-derived compounds are of current interest as antiseptics as well as antimicrobial agents in medication. The effectiveness of the plants in treating various disorders is certainly well established along with a significant level of study has been carried out within this area by investigators in India as well as abroad. The worldwide breakthrough of multi-drug resistant bacterial strains is progressively restricting the potency of existing medicines and considerably leads to remedy failing of infections. For example include methicillin - resistant Staphylococci, Pneumococci resistant to penicillin and macrolides, vancomycin-resistant Enterococci and multi-drug resistant gram-negative organisms 1.
A. pilosa is an ornamental, in addition to a medicinal plant. All parts of this plant are widely used as a folklore medicine for the treatment of various ailments by the traditional Indian healer. Its root is utilized to cure various illness like sexually transmitted diseases viz., gonorrhoea and syphilis, blood diseases. Traditionally, the paste of the leaves is applied to the neck region for cough, quinsy and applied externally in case of itch, eczema and other skin troubles, anti-diabetic, anti-phlogistic, rheumatism, reduce burning sensation and anti-diabetic 6. Young wines are mixed with rhizome of ginger are spread all around the body to relieve from fever 7. The decoction of its root used to treat diarrhoea and cathartic 8, 9.
A vast range of phytochemical constituents has been separated from the genus Argyreia, i.e., glycosides, alkaloids, amino acids, proteins, flavonoids, triterpene, and steroids 10. The genus Argyreia has been reported various biological activities including nootropic, aphrodisiac, anti-oxidant, antiulcer, immune-modulatory, hepato-protective, anti-inflammatory, anti-hyperglycemic, anti-diarrheal, antimicrobial, antiviral, nematicidal, anti-convulsant, analgesic, anti-inflammatory, wound healing and central nervous depressant activities 10-14. Even though the drug has many uses, it’s pharmacological and phytochemistry is very poorly explored 15.
Although, the plant has been extensively used for its traditional value, antibacterial and antioxidant activities remain unexplored. Therefore, the current investigation had been carried out to study the antimicrobial and antioxidant potency of this medicinal plant.
MATERIALS AND METHODS:
Chemicals: All analytical grade chemicals were utilized in this study were procured from E. Merck, Germany. 2, 2-diphenyl-1picrylhydrazyl (DPPH), tannic acid, ascorbic acid, quercetin, Folin-Ciocalteu’s reagent, aluminium chloride, hydrogen peroxide, ferric chloride, potassium ferricyanide, Dimethyl sulfoxide, agar powder, beef extract, and peptone were obtained from Hi-Media, Mumbai; petroleum ether, toluene, acetone, ethyl acetate and methanol.
Plant Material: The plant material was obtained from Tirupati, Chittoor district of Andhra Pradesh, India during March 2016 and authenticated by Dr. K. Madhava Chetty, Taxonomist, Sri Venkateswara University Tirupati, India. Voucher specimen No. 1922 was deposited at the herbarium for future reference. Then it was shade dried, powdered and sieved through 20 mesh and kept in an airtight container for future use 16, 17.
Extraction Method: The whole plant of A. pilosa were collected, washed in tap water to get rid of any contaminants and then dried to remove water from the surface of the plant. The plant was cut into small pieces and dried in the shade in room temperature for 7 days. The dried plant material was ground to coarse powder and defatted using petroleum ether. An equal quantity of defatted dried material of Argyreia pilosa was subjected to maceration with ethyl acetate and methanol for 72 h each. It was then filtered and concentrated (rotary-evaporator) to obtain dried ethyl acetate extract (APEE) and methanolic extract (APME). Percentage yield was calculated using the following Eqn. 1.
Percentage yield (%) = Weight of Extract (g) / Weight of defatted dried plant material (g) × 100
Qualitative Phytochemical Analysis: The extracts had been analyzed for the presence of alkaloid, glycoside, steroids, triterpenoids, flavonoids, proteins, amino acids, carbohydrates, reducing sugar, tannin, and saponin using standard methods 15, 16, 17.
Quantitative Phytochemical Analysis:
Determination of Total Flavonoids Content: Total flavonoids contents of two extracts had been estimated by aluminium chloride colorimetric assay depending on the development of a complex flavonoid-aluminum, having a maximum absorbance at 510 nm 18. Quercetin utilized to make the calibration curve. One milliliter of sample or quercetin standard solution was added into a 10 ml volumetric flask, mixed with 4 ml of 60% ethanol and 0.3 ml of 5% NaNO2 for 6 min, after that, 0.3 ml of 10% AlCl3 was added to react for another 6 min. The reaction was stopped by 4 ml of 4% NaOH, and the total volume was topped up to 10 ml with 60% ethanol. The absorbance was measured after 15 min at 510 nm. Total flavonoids contents were expressed as mg Quercetin equivalents per gram dry weight of each extract 19. All samples were analyzed in triplicate.
Determination of Total Tannin Content: Total tannin content (TTC) was estimated using Folin-Ciocalteu reagent (FCR) taking tannic acid as standard 20. FCR being sensitive to reducing compounds including polyphenols produce blue color upon reaction. The extracts utilized in this analysis should be in the concentration of 1 mg/ml. The reaction mixture was made by combining 0.5 ml of extract, 2.5 ml of 10% Folin-Ciocalteu’s reagent mixed with water, in addition, to add 2.5 ml of 7.5% Na2CO3. Blank was prepared by the same procedure without a sample. The samples had been incubated in a thermostat at 45 °C for 45 min. The absorbance was determined by making use of spectrophotometer at λmax = 765 nm.
The samples have been prepared in triplicate for each analysis, and the mean value of absorbance had been acquired. The same method was repeated for the standard solution of tannic acid, and the calibration line was construed. Based on the measured absorbance, the concentration of phenolics was calculated (mg/ml) from the calibration line. All the determinations were done in triplicate. The total phenolic content is expressed in mg tannic acid equivalent (TAE) per gram.
Antioxidant Assays:
DPPH Free Radical Scavenging Assay: DPPH scavenging capacity of each extract was determined according to the method of Md. Nur Alam et al. A stock solution made up of 40 µg/mL of DPPH (in anhydrous ethanol, w/v) had been prepared. 1 ml of sample was mixed with 4 ml of ethanol DPPH solution. The mixture had been shaken vigorously as well as incubated at room temperature for 30 min in the dark. The absorbance was determined at 517 nm. A control was prepared without a sample. Ascorbic acid was used as positive controls. The DPPH radical scavenging ability was calculated according to the following equation:
DPPH scavenging rate (%) = [(A0-A1)/A0] × 100
Where, A0 is the absorbance of control, and A1 is the absorbance of the sample. Each sample was analyzed in triplicate 21.
Hydrogen Peroxide Scavenging Activity Assay: A solution of hydrogen peroxide (40 mM) is prepared in phosphate buffer (50 mM pH 7.4). The concentration of hydrogen peroxide is determined by absorption at 230 nm using a spectrophotometer. Extract (20–60 lg/mL) in distilled water is added to hydrogen peroxide, and the absorbance at 230 nm is determined after 10 min against a blank solution containing phosphate buffer without hydrogen peroxide. The percentage of hydrogen peroxide scavenging is calculated as follows:
% scavenged (H2O2) = [(Ai – At) / Ai] × 100
Where, Ai is the absorbance of control and At is the absorbance of test 21, 22.
Reducing Power Method: In the method, 2.5 ml of 0.2 M phosphate buffer with pH 6.6 and 2.5 ml of K3Fe (CN)6 (1% w/v) are combined to 1.0 ml of sample mixed in distilled water. The resulting mixture is incubated at 50 °C for 20 min, followed by the addition of 2.5 ml of trichloroacetic acid (10% w/v).
The mixture is centrifuged at 3000 rpm for 10 min to collect the upper layer of the solution (2.5 ml), mixed with distilled water (2.5 ml) and 0.5 ml of FeCl3 (0.1%, w/v). The absorbance is then measured at 700 nm against blank sample 21, 23.
Antibacterial Activity:
Test Microorganisms: The antibacterial activity of the plant extracts had been examined against some gram-positive and gram-negative bacteria, i.e., Pseudomonas aeruginosa, Staphylococcus aureus, streptococcus epidermidis, and Escherichia coli, which are obtained from Laboratory of Micro-biology, NICU, SUM Hospital, Bhubaneswar.
Well Diffusion Method for Determination of Zone of Inhibition: The antibacterial study of APCE, APEE, and APME had been carried out through well diffusion method 24, 25. The inoculation of the microorganism had been prepared from bacterial culture. 15 ml of nutrient agar medium was put in clean sterilized Petri-plates and allowed to cool and solidify. 100 μl of broth of bacterial stain was pipetted out and spread over the medium evenly by a spreading rod till it is dried properly. Wells of 6 mm in diameter are bored using sterile cork borers. Solutions of both the extracts (10 mg/ml) in DMSO (dimethyl sulphoxide) were prepared. 100 μl of plant extract solutions were added to the wells. The Petri-plates were incubated at 37 °C for 24 h. Ciprofloxacin (1 mg/ml) was used as positive control, and DMSO was taken as negative control. Antibacterial activity was evaluated by measuring the zone of inhibition diameters. All the determinations were performed in triplicate.
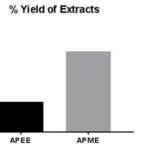
RESULTS AND DISCUSSION: Extraction of defatted plant material of A. pilosa had been performed utilizing chloroform, ethyl acetate and methanol as a liquid extractor to yield dried APCE (Buff powder), APEE (Blackish green powder) and APME (Dark Green Powder). Defatting of plant material is a pre-extraction process that eliminates unwanted fatty and waxy substances from the plant material. On determining percentage yield (%) using Eqn.1, % yield of APME (5.38%) and APEE (2.25%) are shown in Fig. 1.
Percentage (%) yield= Weight of extract (g) / Weight of plant material (g) × 100
FIG. 1: PERCENTAGE (%) YIELD OF EXTRACTS
The results of qualitative phytochemical analysis of crude powder of A. pilosa are tabulated in Table 1.
TABLE 1: PHYTOCHEMICAL ANALYSIS OF VARIOUS EXTRACTS OF THE WHOLE PLANT OF ARGYREIA PILOSA WIGHT & ARN.
| Phytoconstituents | Method | Ethylacetate Extract | Methanolic Extract |
| Flavonoids | Shinoda Test | + | + |
| Zn. Hydrochloride test | + | + | |
| Lead acetate Test | + | + | |
| Volatile oil | Stain test | - | - |
| Alkaloids | Wagner Test | + | + |
| Hager’s Test | + | + | |
| Tannins & Phenols | Fecl3 Test | + | + |
| Potassium dichromate test | + | + | |
| Saponins | Foaming Test | - | - |
| Steroids | Salkowski test | - | + |
| Carbohydrates | Molish test | - | - |
| Acid compounds | Litmus test | - | + |
| Glycoside | Keller-Killani Test | + | + |
| Amino acids | Ninhydrin test | + | + |
| Proteins | Biuret | + | + |
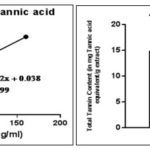
FIG. 2: STANDARD CURVE OF TANNIC ACID AND COMPARISON BY GRAPHICAL REPRESENTATION OF TOTAL TANNIN CONTENT
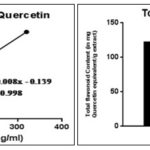
FIG. 3: STANDARD CURVE OF QUERCETIN AND COMPARISON BY GRAPHICAL REPRESENTATION OF TOTAL FLAVONOIDS CONTENT
The outcomes of quantitative estimation of total tannin content (TTC) and total flavonoid content (TFC) are being represented in Fig. 2 and 3 respectively. It is observable through the results that TTC and TFC in REE> RME. Antioxidant activity of any crude extract might be because of several mechanisms.
Binding of transition metal ion, radical scavenging, decomposition of peroxide, reducing capacity, prevention of continued hydrogen abstraction and prevention of chain initiation are some of these mechanisms. DPPH is a stable free radical having absorption at 515 nm. When reduced by an antioxidant to form DPPH, the natural deep violet color of DPPH changes to pale yellow 26.
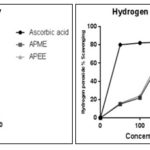
The change in the colour is proportional to the strength of the antioxidants and a significant decrease in the absorbance of the reaction mixture indicates significant free radical activity of the test material. Hydrogen peroxide (H2O2) scavenging activity of the plant extracts is assessed spectrophotometrically with the disappearance of H2O2 at 230 nm 27. H2O2 generates hydroxyl radical within the tissues and is harmful to them. Therefore, removal of H2O2 is extremely important 28. Results of hydrogen peroxide (H2O2) scavenging activity is presented in Fig. 4.
Based on the reducing power of the screening substance, the yellow color of the Fe(III) becomes to Fe(II), Perl’s Prussian blue which may be measured spectrophotometrically at 700 nm 21. The reductive capacity of APEE and APME is compared to that of the Ascorbic acid in Fig. 5.
It is noticed that there exists positive correlation between the antioxidant activity and reducing the power of APEE and APME. Like the antioxidant activity study, reducing the power of REE and RME also increases with the increase in concentration. Reductones, which exhibit the antioxidant activity by breaking the free radical chain and donating a hydrogen atom, is also responsible for the reducing properties 29.
FIG. 4: ANTIOXIDANT ACTIVITY STUDY OF ASCORBIC ACID, APME, AND APEE
FIG. 5: REDUCING POWER ASSAY OF ASCORBIC ACID, APEE, AND APME
The IC50 values of APEE and APME are portrayed in Table 2. The IC50 value of APEE is less than APME, which states the fact that APEE shows better antioxidant property than APME. The antioxidant activity has a positive correlation with total phenolic and flavonoids content 1, which act as scavengers to various oxidizing species 30. For that reason, it was quite sensible to determine the TPC and TFC in the plant extract. TPC and TFC of REE are more than that of RME as evident from Fig. 3B and Fig. 3F, respectively reasoning the result obtained that IC50 of REE is less than that of RME.
TABLE 2: IC50 VALUES OF APEE AND APME
| Testing sample | IC50 (µg/ml) | |
| DPPH radical scavenging activity | Hydrogen peroxide assay | |
| Ascorbic acid | 34.14 | 32.67 |
| APEE | 140.23 | 132.32 |
| APME | 147.34 | 137.53 |
For the current study, the potency of APEE and APME is screened against gram-positive and gram-negative bacteria. The well diffusion method of screening antibacterial activity is considered to provide better results and is associated with the carrier solvent, DMSO which is likely to diffuse highly across the medium 31.
In this study, DMSO is employed as a solvent for solubilizing APEE and APME for antibacterial study. DMSO is a polar aprotic solvent which can dissolve both polar and non-polar compounds. DMSO can be used as a cryoprotectant for long term low temperature conservation of various biomaterials 32 and thus considered to have no impact on the growth or inhibition of growth on microorganisms. Although, the literature facilitates the use of DMSO as a solvent about antibacterial studies, a negative control (i.e., only DMSO) is also studied in each plate to be extra sure.
In the present circumstances of increasing resistance of microbes towards the worldwide used antibacterial 33, it is crucial that the actual ingredients are having potential antibacterial need to be extracted. The current study is useful to find the extract that exhibits better antibacterial activity. In this study, the plant material was extracted with ethyl acetate and methanol by maceration.
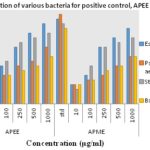
This study also compares the antibacterial activity of ethyl acetate and methanolic extracts of A. pilosa, APEE and APME, respectively. Results depicted in Fig. 6 and 7 show that both REE and RME at different concentrations 1, 10, 100, 250, 500 and 1000 µg/ml (100 μl incorporated in each well) possess antibacterial activity towards both gram-positive and gram-negative bacteria. DMSO shows no zone of inhibition (ZI).
TABLE 3: ZONE OF INHIBITION (ZI) OF THE EXTRACTS (APEE AND APME) AGAINST DIFFERENT GRAM NEGATIVE AND GRAM-POSITIVE BACTERIA
| Test Organisms | APEE(µg/ml) ZI±S.D (mm) | APME(µg/ml) ZI±S.D (mm) | |||||||||||
| Concentration (µg/ml | |||||||||||||
| Std. (5µg/ml) | 10 | 100 | 250 | 500 | 1000 | Std. (5µg/ml) | 10 | 100 | 250 | 500 | 1000 | ||
| E. coli | 18±
0.577 |
5±
0.232 |
8±
0.179 |
12±
0.545 |
15±
0.318 |
17±
0.253 |
18±
0.225 |
4±
0.179 |
6±
0.325 |
12±
0.291 |
14±
0.381 |
16±
0.485 |
|
| P. aeruginosa | 19±
0.623 |
4±
1.332 |
6±
0.265 |
8±
0.21 |
9±
0.229 |
12±
0.789 |
19±
0.623 |
4±
0.21 |
5±
0.212 |
6±
0.296 |
8±
0.288 |
10±
0.568 |
|
| S. aureus | 17±
0.232 |
5±
0.323 |
9±
0.364 |
13±
0.598 |
14±
0.558 |
15±
0.553 |
17±
0.364 |
3±
0.359 |
8±
0.206 |
12±
0.402 |
13±
0.577 |
14±
1.015 |
|
| B. subtilis | 16±
1.23 |
4±
0.623 |
6±
0.485 |
7±
0.307 |
8±
0.615 |
10±
0.458 |
16±
0.485 |
4±
0.489 |
5±
0.223 |
7±
0.525 |
8±
0.750 |
10±
0.5 |
|
FIG. 6: ANTIBACTERIAL ACTIVITY OF APEE AND APME AGAINST ESCHERICHIA COLI, PSEUDOMONAS AERUGINOSA, STREPTOCOCCUS AUREUS, AND STAPHYLOCOCCUS EPIDERMIDIS
FIG. 7: ZONE OF INHIBITION OF VARIOUS BACTERIA FOR STANDARD, APEE, AND APME
All the results are mean ± SD (n=3). The antibacterial activity of APEE and APME may be attributed to TTC and TFC in APEE and RME. Flavonoids and phenolic compounds are recognized for their role in protecting plants against microbial attack 30. Tannins also act as astringents and have the ability to bind and precipitate and shrink proteins 34. This ability of tannins directly damages the bacterial cell wall and precipitate proteins killing the bacteria 35, 36. From the results of the ZI depicted in Table 3 and from Fig. 5 which compares the ZI formed by REE and RME against different bacteria, it is deemed fit to state that REE has better antibacterial activity than RME.
CONCLUSION: The whole plant of A. pilosa was extracted with ethyl acetate and methanol to obtain APEE and APME. Both APEE and APME was subjected to phytochemical screening, and it was found that both contain flavonoids, alkaloids, tannins, glycosides, proteins, and amino acids except for steroids and acid compounds which is only present in APME. TTC and TFC are more in APEE than APME. Both APEE and APME show antioxidant and antibacterial activity, APEE is claimed to have more antibacterial and antioxidant potency than APME.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Kaneria M and Chanda S: Evaluation of antioxidant and antimicrobial properties of Manilkara zapota (chiku) leaves by sequential soxhlet extraction method. Asian Pac J Trop Biomed 2012; 2: S1526-33.
- Aruoma OI: Free radicals, oxidative stress, and anti-oxidants in human health and disease. J Amer Oil Chem Soc 1998; 75: 199-212.
- Jayaprakasha GK, Rao LJ and Sakariah KK: Antioxidant activities of flavidin in different in vitro model systems. Bioorg Med Chem 2004; 12: 5141-6.
- Muniandy P, Shori AB and Baba AS: Influence of green, white and black tea addition on the probiotic yogurt during refrigerated storage. Food Packaging and Shelf Life 2016; 8: 1-8.
- Vhangani LN and Wyk JV: Antioxidant activity of Maillard reaction products (MRPs) in a lipid-rich model system. Food Chem 2016; 208: 301-308.
- Marles R. J. and Farnsworth N. R. Antidiabetic plants and their active constituents. Phytomedicine 1995; 2: 137-89.
- Bhandary MJ, Chandrashekar KR and Kaveriappa KM: Medical ethnobotany of the Siddis of Uttara Kannada district, India. J Ethnopharmacol 1995; 47: 149-58.
- Ghorbani A, Langenberger G, Feng L and Sauerborn J: Ethnobotanical study of medicinal plants utilized by Hani ethnicity in Naban River Watershed National Nature Reserve, Yunnan, China. J Ethnopharmacol 2011; 134: 651-67.
- Gurih-Fakim A, Sewraj M, Gueho J and Dulloo E: Medical ethnobotany of some weeds of Mauritius and Rodrigues, J Ethnopharmacol 1993; 39: 175- 85.
- Galani VJ, Patel BG, and Patel NB: Argyreia speciosa (Linn. f.) sweet: A comprehensive review. Pharmacogn Rev 2010; 4: 172-8.
- Lalan BK, Hiray RS and Ghongane BB: Evaluation of analgesic and anti-inflammatory activity of extract of Holoptelea integrifolia and Argyreia speciosa in Animal Models. J Clin Diagn Res 2015; 9: FF01-4.
- Yadav KS, Yadav NP, Rawat B, Rai VK, Shanker K and Venkateswara Rao C: An assessment of wound healing potential of Argyreia speciosa Scientific World J 2014; 1-6.
- Galani VJ and Patel BG: The psychotropic activity of Argyreia speciosa roots in experimental animals. Ayu 2011; 32: 380-4.
- Motawi TK, Hamed MA, Hashem RM, Shabana MH and Ahmed YR: Protective and therapeutic effects of Argyreia speciosa against ethanol-induced gastric ulcer in rats. Z Naturforsch C 2012; 67: 47-57.
- Kataria S, Rao SK, Bhandari A and Kaur D: Pharma-cognostical standardization of Corchorus depressus (L.) Stocks (Tiliaceae)- A promising ethnomedicinal plant. Ind J Tradit Knowl 2013; 12: 489-97.
- Choudhary N, Siddiqui MB and Khatoon S: Pharma-cognostic evaluation of Tinospora cardifolia (Willd.) Miers and identification Markers. Ind J Tradit Knowl 2014; 13: 543-50.
- Stanković MS: Total phenolic content, flavonoid concentration and antioxidant activity of Marrubium peregrinum extracts. Kragujevac J Sci 2011; 33: 63-72.
- Akrout A, Gonzalez LA, El-Jani H and Madrid PC: Antioxidant and antitumor activities of Artemisia campestris and Thymelaea hirsuta from southern Tunisia. Food Chem Toxicol 2011; 49: 342-7.
- D, Ribarova. F and Atanassova M: Total phenolics and total flavonoids in bulgarian fruits and vegetables. J Chem Technol Metall 2005; 40: 255-60.
- Rahate KP, Padma R, Parvathy NG and Renjith V: Quantitative estimation of tannins, phenols and antioxidant activity of methanolic extract of Imperata cylindrical. Ind J Pharm Edu Res 2013; 4: 73-7.
- Alam MN, Bristi NJ and Rafiquzzaman M: Review on in-vivo and in-vitro methods evaluation of antioxidant activity. Saudi Pharm J 2013; 21: 143-52.
- Sur TK, Hazra A, Hazra AK and Bhattacharyya D: Antioxidant and hepatoprotective properties of Indian Sunderban mangrove Bruguiera gymnorrhiza leave. J Basic Clin Pharma 2016; 7: 75-9.
- Walia H, Kumar S and Arora S: Comparative analysis of antioxidant and phenolic content of chloroform extract/ fraction of Terminalia chebula. J Basic Clin Pharma 2011; 2: 115-24.
- Chitravadivu C, Manian S and Kalaichelvi K. Anti-microbial studies on selected medicinal plants, Erode region, Tamil Nadu, India. Middle East J Sci Res 2009; 4: 147-52.
- Nashwa T, Hassan HM, AbdelGhani SMM, Radwan IA and Hammouda O: Comparative chemical and anti-microbial study of nine essential oils obtained from medicinal plants growing in Egypt. Beni-Seuf Univ J Appl Sci 2014; 3: 149-56.
- Shekhar TC and Anju G: Antioxidant activity by DPPH radical scavenging method of Ageratum conyzoides Leaves. Am J Ethnomed 2014; 1: 244-9.
- Fernando CD and Soysa P: Optimized enzymatic colorimetric assay for determination of Hydrogen peroxide (H2O2) scavenging activity of plant extracts. Methods X 2015; 2: 283-91.
- Keser S, Celik S, Turkoglu S, Yilmaz O and Turkoglu I: Hydrogen peroxide radical scavenging and total anti-oxidant activity of Hawthorn. Chem Journal 2012; 2: 9-12.
- Kumaran A and Karunakaran RJ: In-vitro antioxidant activities of methanol extracts of five Phyllanthus species from India; LWT- Food, Science and Technology 2007; 40: 344-52.
- Harborne JB and Williams CA: Advances in flavonoid research since 1992. Phytochemistry 2000; 55: 481-504.
- Valgas C, Machado de Souza S, Smania EFA and Smania A: Screening methods to determine the antibacterial activity of natural products. Braz J Microbiol 2007; 38: 369-80.
- Huang P, Dong A and Caughey WS: Effects of dimethyl sulfoxide, glycerol and ethylene glycol on secondary structures of cytochrome C and lysozyme as observed by Infrared spectroscopy. J Pharm Sci 1995; 84(4): 387-92.
- Zheng B, Li A, Jiang X, Hu X, Yao J and Zhao L: Genome sequencing and genomic characterization of a tigecycline-resistant Klebsiella pneumoniae strain isolated from the bile samples of a cholangiocarcinoma patient. Gut Pathogens 2014; 6: 1-7.
- Ashok PK and Upadhaya K: Tannins are astringent. J Pharmacog Phytochem 2012; 1: 45-50.
- Baskaran C, Rathabai V and Kanimozhi D: Screening of antimicrobial activity and phytochemical analysis of various leaf extract of Murraya koenigii. Int J Res Ayurveda Pharm 2011; 2: 1807-10.
- Taveria de Jesus NZ, Falcao HDS, Gomes IF, Leite TJDA, Lima GRDM and Babosa-Filho JM: Tannins, peptic ulcer and related mechanisms. Int J Mol Sci 2012; 13: 3203-28.
How to cite this article:
Prasanth DSNBK, Rao AS and Yejella RP: Phytochemical, in-vitro antioxidant and antibacterial activities of Argyreia pilosa Wight & Arn. (Whole plant). Int J Pharmacognosy 2017; 4(4): 109-17. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.4(4).109-17.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
2
109-117
741
744
English
IJP
D. S. N. B. K. Prasanth *, A. S. Rao and R. P. Yejella
Department of Pharmacy, JNTUK, Kakinada, Andhra Pradesh, India.
dsnbkprasanth@gmail.com
15 January 2017
22 March 2017
25 March 2017
10.13040/IJPSR.0975-8232.IJP.4(4).109-17
01 April 2017