PHYSICO-CHEMICAL CHANGES THAT OCCURS DURING FERMENTATION OF ARISHTAM (A HYDRO ALCOHOLIC CLASS OF DRUG OF AYURVEDA)
HTML Full TextPHYSICO-CHEMICAL CHANGES THAT OCCURS DURING FERMENTATION OF ARISHTAM (A HYDRO ALCOHOLIC CLASS OF DRUG OF AYURVEDA)
S. Thirukannan, M. Kishore Krithivel, P. Ilansuriyan and M. Marimuthu *
Nutraceutical Chemistry Lab, Department of Food Process Engineering, School of Bioengineering, SRM University, Kattankulathur - 603203, Tamil Nadu, India.
ABSTRACT: The present study was to evaluate the physicochemical changes that take place in vasarishta during the manufacturing of the arishtams (hydro-alcohol class of drug of Ayurveda). The manufacturing process of the Arishtams usually takes 21 -30 days. The time period of the entire study was 21 days. Batch fermentation was done for a period of 14 days after which the fermentation process was inhibited using methods recommended in Ayurvedic literature and as per the company’s standard operating procedure. The arishtam was analysed for its physicochemical parameters periodically at an interval of 24 h. The parameters chosen for the physicochemical characterisation of the fermentation process were pH, Temperature, Specific gravity, Brix value, Sugar content, Alcohol content, Free amino acid content and Tannin content. The pH was found to vary between 4.30 and 5.57. The temperature was found to vary randomly between 25 ºC and 29 ºC throughout the period. The specific gravity was found to decrease drastically up to the 14th day followed by a gradual increase up to the 21st day. The brix value of the arishtam, on the other hand was found to decrease gradually up to the 14th day and then it maintained a constant value. The sugar content was found to gradually decrease over the period of fermentation while the alcohol content was found to increase up to day 16 followed by a drastic downfall. The free amino acid and tannin content were constantly fluctuating throughout the entire period. Using the values obtained from the experiments, an attempt has been made to understand a few physicochemical changes that take place in the vasarishta during fermentation.
| Keywords: |
Arishtam, total free amino acids, Tannin, Alcohol content, Brix, Sugar
INTRODUCTION: Ayurveda is an important, prominent natural system of health care all over the world 1. It is a traditional system of Indian medicinal field which being practiced for thousands of years.
More than 1,200 species of plants, nearly 100 minerals, and over 100 animal products include in the Ayurvedic Pharmacopoeia.
Considerable research on chemistry, pharmacognosy, pharmacology and clinical therapeutics of Ayurveda has been carried out and tends to numerous drugs have entered into the international pharmacopeia 2. Ayurvedic system of treatment has been estimated to meet 70-80% of the healthcare needs of India 3. WHO notes that of 119 plant-derived pharmaceutical medicines, about 74% are used in modern medicine. Normally Herbal formulation must be standardized as per WHO guidelines 4.
The aim of WHO guidelines are to define basic criteria for the evaluation of safety, quality, and efficacy of herbal drugs 5. The herbal drugs singularly or in combinations contain compounds in complex matrices in which no single active constituent is responsible for overall efficacy New England Journal of Medicine had given a report as 70% of the well-educated and higher than average income people opt for herbal medicines. It starts to create a challenge in establishing quality standards for raw materials and standardization of the finished herbal drugs.
The importance of conducting clinical research in herbal drugs, simple bioassays for biological standardization, pharmacological and toxicological evaluation, toxic herbal drugs in use, various animal models for toxicity and safety evaluation, were the major research areas in this field 6. The most commonly used five basic formulations has some drawback like higher chances of microbial growth, low level of shelf life and its require high dose and are not palatable. Therefore, they developed secondary preparations like fermented preparation, confectionaries, medicated oil, pills, etc. The short shelf life is the major drawback of the decoctions for the large-scale production 7.
Nowadays people find very hard and tedious to prepare and administer decoctions. To overcome these problems, concentrated and fermented decoctions are prepared nowadays. The self-generated herbal fermentations of traditional Ayurvedic system are Arishtas and asavas. It has considered a unique and valuable therapeutics in Ayurveda 8. Though traditional knowledge in literature as well as in practice exists about arishtas and asavas, there was little effort to document, preserve and improve this knowledge for the betterment of mankind. They are alcoholic medicaments prepared by allowing the herbal juices or their decoctions to undergo fermentation with the addition of sugars. Arishtas are made with decoctions of herbs in boiling water while asavas are prepared by directly using fresh herbal juices. Fermentation of both preparations is brought about by the addition of dhataki flowers which acts as a source of carbohydrates and additionally spices for improving their assimilation. They are moderately alcoholic (upto 12%) and mostly sweetish with slight acidity and acceptable aroma. Medicinal wines have enhanced therapeutic properties, better keeping quality, and improvement in the efficiency of extraction of drug molecules from the herbs and improve in drug delivery into the human body sites 9-13.
The conventional method followed in the production of arishtams is highly time-consuming. Manufacturers are unable to introduce new changes in the production strategy as knowledge regarding the physiochemical changes that take place during fermentation is unknown or understood. Extensive literature search and interaction with Ayurvedic physicians suggested that no or little information is available on the physicochemical and biochemical changes that occur during the fermentation of arishtas and Asavas 14-16. The changes that are likely to happen to the herbal decoctions under anaerobic conditions have not been monitored on a day to day basis, and it becomes difficult to enumerate the microorganisms that bring about the changes during the production of Asavas and Arishtas.
Studies in this direction would help us to understand the biochemical changes that influence the rate of fermentation. Results of such studies would enable us to suggest modifications to be undertaken in the fermentation process based on modern scientific concepts to reduce the time frame required for the manufacture of Asavas and Arishtas 17-20. Hence in the present study, an attempt has been made to follow up the physicochemical changes that are likely to happen during the fermentation of vasarishta- an arishtam of commercial importance for the cure of bronchitis and asthmam 20-24.
MATERIALS AND METHODS:
Collection of Arishtam Sample: The arishtam (vasarishta) sample was obtained from AVN Arogya Formulations Pvt. Ltd., Madurai. They were authenticated by Dr.Baburaj Production Manager, AVN Arogya Formulations Pvt. Ltd., Madurai.
Processing of Arishtam Sample: The arishtam (vasarishta) sample got from AVN Arogya Formulations was made into 21 aliquots of equal volume and maintained in 21 airtight containers. The containers were stored in a cold place to maintain 25 ºC until the completion of fermentation. On daily basis the containers were opened, and the physicochemical characteristics were evaluated every day. Once the alcohol was produced, the fermentation was stopped by adding sodium benzoate, and the medicament powder and camphor were added. This was done as per the methodology adopted by the company. The physicochemical characterization was carried out until the date, decided to be the end stage of the production process by the company. The end stage is usually decided once the desired organoleptic characteristics have arrived.
Acidity Estimation: pH is the negative logarithm of hydrogen ion concentration. The pH probe was used to determine the acidity of an aqueous solution of arishtam sample.
Temperature Measurement: Chemical reactions within yeast are facilitated by enzymes, which are large organic catalysts. Each enzyme has an "optimal temperature range" a temperature range at which it performs best. As soon as the airtight container is opened, the temperature of the arishtam sample was checked using a thermometer.
Total Sugar Content (Fehling’s Test): The quantitative estimation of total reducing sugars was performed by Fehling’s test 25. 1 g of the sample was made up to 250 ml with distilled water. It was heated to 70 ºC for 5 min with the addition of 5 ml Hydrochloric acid. It was cooled immediately and neutralized with sodium carbonate. The weight of the sample in the solution was recorded as W. 5 ml of Fehling’s solution A and 5 ml of Fehling’s solution B was added, and 10 ml of sample solution was added in the conical flask which was heated to boiling over asbestos gauze. 1 ml of methylene blue was added as an indicator, and the titration was stopped when the solution becomes reddish brown.
Calculation:
Percentage of total reducing sugar = (250 × 100 × N) / (V × W)
N-Normality of Fehling’s solution
Determination of Brix Value: The index of dissolved solids present in the arishtam sample gives the Brix value. The upper lid of the Refractometer was cleaned with distilled water and was adjusted to zero. The 4 to 5 drops of the sample (28 ± 2 °C) was placed on the flat prism of the hand Refractometer and was observed through the eyepiece. A sharp separation will be seen between the white and blue background. The line of separation is the brix value.
Estimation of Total Free Amino Acids: Ninhydrin is a powerful oxidizing agent which oxidizes alpha amino acids and yields an intensely colored bluish purple product 26. The ninhydrin solution, 0.2 M Citrate buffer pH 5.0, the diluent solution was prepared. To 0.1 ml of the extract, 1 ml of ninhydrin solution was added, and the volume was made up to 2 ml with distilled water. It was heated in a boiling water bath for 20 minutes. 5ml of the diluent solution was added and mixed. After 15 min the intensity of the purple color against the reagent blank (reagent blank was prepared as above by taking 0.1 ml of 80% ethanol instead of the extract) in a calorimeter was read at 570 nm.
Determination of the Percentage of Alcohol Content: 25 ml of the arishtam sample was measured and was transferred to a distillation flask and was diluted to 150 ml with water. This was distilled at 70 ºC and the first 90 ml of the distillate was collected in a standard measuring flask. The volume was made up to 100 ml and was cooled to 25 ºC. The specific gravity of the liquid was determined, and corresponding alcoholic content was noted from the table given below.
Determination of Tannin Content: The vanillin reagent reacts with any phenol that has an unsubstituted resorcinol or phloroglucinol nucleus and forms a colored substituted product which was measured at 500 nm 27. The vanillin hydrochloride reagents and the working standard solution were prepared.
1 ml of the working standard solution was taken, and 5 ml of vanillin hydrochloride reagent was added. It was read in a spectrophotometer at 500 nm after 20 min. The blank was prepared using vanillin hydrochloride alone.
HPTLC Analysis: The plain arishtam (without the addition of yeast) sample, intermediate arishtam (day 9) sample, final arishtam (day 21) were taken to compare various chemical constituents present in them. These 3 samples were subjected to chromatographic profiling.
HPTLC Condition: Chromatography was performed on 20 × 10 cm Aluminium backed HPTLC silica gel 60 F254 plates (MERCK). Before use, the plates were dried in an oven at 50 ºC for 5min. Sample was applied as 8 mm bands by spraying by means of Camag Linomat 5 sample applicator equipped with a 100 µl syringe. The distance between bands was 18 mm. The developing solvent was allowed to ascend to 80mm, with Toluene: Ethyl acetate: Methanol (7:2:1) as a mobile phase in a Twin Though Chamber Was saturated for 20 min lined with Whatman filter paper. The room temperature was 25 ± 2 ºC. The average development time was 15minutes. After development, the plates were dried at room temperature. Before densitometric scanning, plates were heated for 5 min in the oven at 105 °C. Densitometric evaluation of the plates was performed at 254 nm, 366 nm using a Camag TLC scanner-3 equipped with win CATS software, using a Deuterium light source, the slit dimensions were 6.00 × 0.45 mm.
RESULT AND DISCUSSION: The herbal industry uses organoleptic characteristic evaluation as one of the quality control standards for the finished products. In the present study too, this was used as an endpoint indicator s shown in Table 1. The different stages of fermentation were chosen as representatives of each important modification that are perceived to happen during the production process in Table 2. The brix value, total free amino acid content and sugar content were by the 4 phases of microbial growth.
A slow decrease in the value was observed during the lag and early log phase (day 1-4) and there was a drastic fall during the log (exponential) phase lasting from day 5- 9. The values fluctuated over a small range during the stationary phase (day 10 -14) followed by a prolonged decrease in the final stationary phase (day 15-21), as in the case of sugar and a constant value as in the case of brix and total free amino acid content. The tannin content and temperature values fluctuated randomly throughout the 4 phases, but tannin maintained an almost constant value after day 18. The specific gravity and pH were decreasing and increasing alternatively. Specific gravity reached a constant value in the final stationary phase, but pH kept increasing till day 21 indicating a shift towards alkaline conditions. Alcohol was found to produce only in the late log phase; it was found to increase up to day 17 followed by a slow decrease indicating the reach of stationary phase among the working microbes. Thus the parameters were found to vary with the four phases of microbial growth. However, characterization and enumeration of the microbial population during the stages will help us understand the biochemical changes during the production of arishtam.
TABLE 1: ORGANOLEPTIC PROPERTIES OF FINAL ARISHTAM
| Colour | Reddish brown |
| Odor | Characteristic |
| Nature | Liquid |
| Taste | Astringent |
TABLE 2: PHYSICOCHEMICAL CHANGES DURING FOUR IMPORTANT PHASE OF THE FERMENTATION OF ARISHTAM
|
Parameters |
Initial
Sample (Day 1) |
Intermediate sample
(Day 9) |
Stop Fermentation
(Day 14) |
Final sample (Day 21) |
| pH | 5.57 | 4.48 | 4.40 | 4.94 |
| Temperature (ºC) | 25 | 30.5 | 27 | 27 |
| specific gravity | 1.215 | 1.1629 | 1.1540 | 1.1624 |
| Sugar content (%) | 101.5 | 58.30 | 50.115 | 41.7 |
| Brix | 47.4 | 42.6 | 42 | 41.8 |
| Total Amino Acids content (µg/ml) | - | 0.1171 | 0.1161 | 0.0048 |
| Tannin (µg/ml) | 0.0360 | 0.1171 | 0.0391 | 0.0388 |
FIG. 1: STORAGE OF ARISHTAM SAMPLE
Table 3 shows that there is a drastic decrease in the sugar percentage from day 1-9 indicating the sugar intake for cell growth and metabolism during the lag and log phase. From day 10 -21 the sugar intake was relatively less showing the microbes had entered into the late log and stationary phase of their growth.
TABLE 3: PERCENTAGE OF SUGAR CONSUMPTION
| Days | Sugar content (%) | Percentage of Sugar Decreased |
| Day 1 | 101.5 | - |
| Day 9 | 58.30 | 43.20 % |
| Day 14 | 50.11 | 51.38 % |
| Day 21 | 41.70 | 59.80 % |
TABLE 4: PHYSICOCHEMICAL CHANGES DURING THE FERMENTATION OF ARISHTAM
| Days | pH | Temperature
(˚C) |
Specific gravity | Sugar
(%) |
Brix | Total Free amino acids (µg/mL) | Alcohol
(%) |
Tannin
(µg/mL) |
| 1 | 5.57 | 25 | 1.215 | 101.5 | 48.4 | - | - | 0.0360 |
| 2 | 5.53 | 27 | 1.220 | 95.19 | 48.2 | 0.0760 | - | 0.0443 |
| 3 | 5.27 | 28 | 1.214 | 80.20 | 48.0 | 0.1597 | - | 0.0347 |
| 4 | 5.04 | 26 | 1.179 | 77.67 | 48.0 | 0.7050 | - | 0.0328 |
| 5 | 4.89 | 27 | 1.195 | 70.61 | 46.0 | 0.2054 | - | 0.0349 |
| 6 | 4.61 | 29 | 1.181 | 69.71 | 44.8 | 0.5687 | - | 0.0355 |
| 7 | 4.58 | 29 | 1.171 | 62.86 | 43.0 | 0.1624 | - | 0.0366 |
| 8 | 4.76 | 28 | 1.174 | 60.75 | 43.6 | 0.2522 | - | 0.0404 |
| 9 | 4.58 | 27 | 1.162 | 58.30 | 42.6 | 0.1171 | - | 0.0382 |
| 10 | 4.50 | 28 | 1.162 | 56.04 | 43.0 | 0.2934 | 7.28 | 0.0446 |
| 11 | 4.46 | 27 | 1.161 | 55.41 | 43.0 | 0.2676 | 7.8 | 0.0459 |
| 12 | 4.45 | 27 | 1.160 | 54.92 | 43.0 | 0.1540 | 8.32 | 0.0386 |
| 13 | 4.43 | 27 | 1.157 | 51.29 | 41.6 | 0.0236 | 9.44 | 0.0418 |
| 14 | 4.40 | 28 | 1.154 | 50.11 | 42.0 | 0.1161 | 11.56 | 0.0391 |
| 15 | 4.30 | 29 | 1.152 | 48.32 | 42.1 | 0.1133 | 12.60 | 0.0394 |
| 16 | 4.42 | 28 | 1.156 | 46.51 | 41.8 | 0.0873 | 13.72 | 0.0387 |
| 17 | 4.48 | 29 | 1.158 | 45.01 | 41.8 | 0.0732 | 13.72 | 0.0388 |
| 18 | 4.53 | 28 | 1.161 | 44.49 | 41.8 | 0.0472 | 14.8 | 0.0411 |
| 19 | 4.89 | 27 | 1.162 | 43.70 | 41.8 | 0.0317 | 13.64 | 0.0393 |
| 20 | 4.94 | 27 | 1.162 | 42.00 | 41.8 | 0.0233 | 12.60 | 0.0388 |
| 21 | 4.96 | 27 | 1.163 | 41.70 | 41.8 | 0.0048 | 11.56 | 0.0388 |
The pH range decreases during the period of fermentation from Day 1 to day 14 and the pH gradually increases later. Fermentation of glucose results in the abundant production of acidic end products, the presence of which can be detected by the pH indicator in the medium. The pH values recorded in the present study reflect the presence of weak acids in the fermenting medium and the pH range of 4-5.5 observed in the present study is indicative of the existence of carbonic acid. Although the presence of carbonic acid depends on the partial pressure of the gaseous carbon dioxide, its pH value is usually 5.7. Hence, it would be appropriate to estimate the levels of CO2 generated and the levels of carbonic anhydrase in the medium to ascertain the fact that carbonic acid is present Table 4 and Fig. 2.
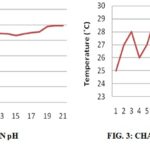
The Temperature increases and decreases randomly within the range of 25 ºC to 29 ºC indicating that the biochemical changes and the microbial flora are highly dynamic and heat is generated during the process of fermentation as shown in Fig. 3.
The specific gravity decreases during the period of fermentation from Day 1 to Day 14, and the specific gravity gradually increases after the addition of sodium benzoate. Specific gravity decrease is suggestive of the reduction in density of the fermenting medium Fig. 4.
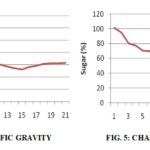
Total sugar content gradually decreases throughout fermentation. Fermentation decreased some of the nitrogenous constituents of the product and much more drastically the sugar content Fig. 5. This is evident from the decrease in sugar content and brix content throughout the study period. It could be inferred that hydrolysis and other metabolic changes freed the nutrients from their bound forms while decreasing the quantity, but increasing the quality and availability of the nutrients.
The total dissolved solids content decreases till Day 14 after which it was maintained at a constant rate till the end of the experiment. The results suggest that during the fermentation process there is the conversion of complex solid entities into liquid and gaseous forms which is also evident from the changes in pH of the medium indicating generation of weaker acids and evolution of alcohol as shown in Fig. 6.
Although there was a drastic reduction in the sugar content throughout the study period more importantly 60% reduction by the end, there was not a drastic decrease in the total dissolved solids content, indicating that the larger biomolecules are broken down into smaller entities and hence the dissolved solid contents levels are maintained as shown in Fig. 7.
The free amino acid randomly increases and decreases during the period of fermentation from day 1 to day 14, and there is a gradual decrease in the free amino acid after the addition of sodium benzoate. During fermentation, yeasts assimilate between 1 and 2 g/L of amino acids Fig. 8. Towards the end of fermentation, yeasts excrete significant but variable amounts of different amino acids. Finally, at the end of alcoholic fermentation, a few hundred mg/L of amino acids remain; proline generally represents 28. In the present study, there is an increase in free amino acid content during the middle of the fermentation process (day1- day14). Individual amino acid profiles have to be established to understand the nature of free amino acids generated and their influence in affecting the texture and flavor of the end product.
The alcohol content increases from day 9 to day 15, and after the addition of sodium benzoate, the alcohol content decreases gradually Fig. 9. The relationship between fermentation conditions and ethanol production rate has been established in this study. From the results of the study, it is evident that alcohol generation seems to start from Day 7 and the alcohol content reaches a 12% level by the 21st day. This is by the Indian pharmacopeia standards established and suggested for the percentage of alcohol present in Arishtams 29.
The tannin content randomly increases and decreases during the period of fermentation. From the results of the present study, it seems that free amino acid content and tannin content can be used effectively as potential indicators of fermentation rate Fig. 10.
FIG. 11: VISUALIZATION OF BANDS AT (A) 254 nm AND (B) 366 nm FOR ARISHTM SAMPLES
PA-Plain Arishtam (Initial stage); IA-Intermediate Arishtam; FA-Final Arishtam
TABLE 5: Rf VALUE AND AREA UNDER THE CURVE FOR ARISHTAM SAMPLES AT 254 nm
| Rf Value | area (AU) | ||||
| PA (Day 1) | IA (Day 9) | FA(Day21) | PA (Day 1) | IA (Day 9) | FA(Day21) |
| 0.09 | 0.13 | 0.13 | 168.1 | 410.5 | 419.3 |
| 0.14 | 0.20 | 0.21 | 412.9 | 716.7 | 658.5 |
| 0.21 | 0.23 | 0.24 | 713.4 | 519.0 | 633.4 |
| 0.24 | 0.32 | 0.37 | 597.6 | 2435.6 | 9155.1 |
TABLE 6: Rf VALUE AND AREA UNDER THE CURVE FOR ARISHTAM SAMPLES AT 366 nm
| Rf Value | area (AU) | ||||
| PA (Day 1) | IA (Day 9) | FA(Day21) | PA (Day 1) | IA (Day 9) | FA(Day21) |
| 0.10 | 0.10 | 0.10 | 389.0 | 222.6 | 108.2 |
| 0.30 | 0.33 | 0.34 | 209.4 | 477.3 | 593.5 |
The chromatogram developed at 254 nm and 366 nm indicate the presence of similar phytoconstituents at the different stages of the fermentation process. Identification of the chemical nature of these bands or compounds will help us to use these phytoconstituents as markers to follow up the production process and to maintain batch to batch consistency as shown in Fig. 11 and 12. It is interesting to note here that there are no additional phytoconstituents present in the stages where the generation of alcohol had happened. The change in the hydroalcoholic environment of the medium does not seem to be effective in extracting the phytoconstituents. However, to confirm this, the chromatogram should be developed with few other solvent systems too.
CONCLUSION: There is a decrease in sugar content; however the dissolved solid content remains more or less the same. Fermentation of sugar and conversion into organic acids is evident from the production of alcohol and changes in pH. The HPTLC profile of the different stages of the fermentation reveals the presence of identical compounds, which could be used to develop quality indicators for following up the production process and to maintain batch to batch consistency during the production of arishtams. Results of the present study indicate that amino acid content and tannin content could be used as quality indicators or endpoint indicators for developing standard manufacturing protocols for the production of arishtams.
ACKNOWLEDGEMENT: We express our thankfulness to Professor & Head, Department of Food Process Engineering, SRM University for his support. We also express our gratitude to Dr. C. Muthamizhchelvan, Director (Engineering & Technology) of SRM University for his continued encouragement towards the project.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Joy, Thomas, Mathew and Skaria: Medicinal plants, Edition 2, 2001: 449-632.
- Patwardhan B, Vaidya ADB and Chorghade M: Ayurveda and natural products drug discovery. Curr Sci 2004; 86: 789-799.
- Viswanathan MV, Unnikrishnan PM, Komatsu K, Fushimi H and Basnet P: A brief introduction to Ayurvedic system of Medicine and some of its problems. Indian J Traditional Knowledge 2003; 2: 159-169.
- Traditional Medicines and Modern Health Care. Progress report by the Director-General. Geneva, World Health Organization 1990.
- Guidelines for the Assessment of Herbal Medicines, Document No. WHO/TRM/91: World Health Organization, Geneva 1991.
- Adesara: Herbal drugs: present status and future prospects, Current science 2001; 81: 1.
- Manish Vyas, Shukla VJ, Patgiri BJ and Prajapati PK: An Unique concentrated & Fermented Dosage Form Pravahikwatha. International journal of Pharmaceutical & Biological Archives 2010; 1: 287-290.
- Kumar KA: The need for developing new dosage presentation forms for traditional medicine, Indian Healthcare Tradition-A Contemporary view, edited by Paulose KG, Murali TS and Kumar NM, Arya Vaidya Sala, Kottakkal 2002; 120-128.
- Murthy KRS: Astanga Hrdayam, Krishnadas Academy, Varanasi 1994: 68-73.
- Shastri MV and Ratnavali VY: IMPCOPS, Madras 1968: 6-10.
- Nadkarni KM: Indian Materia Medical. Bombay Popular Prakashan Pvt. Limited, Bombay 1976: 2: 489.
- Dash VB and Hashyap VL: Iatro Chemistry of Ayurveda. Concept Publishing Company, New Delhi 2002: 69-79.
- Srikantha Murthy KR and Bhavaprakasa of Bhavamisra: Krishnadas Academy, Varanasi 1998; 1: 479-484.
- Dash VB and Hashyap VL: Materia Medica of Ayurveda. Concept Publishing Company, New Delhi 1980.
- Shastri SVR: Asavakalpam, Vaidya Chandrika 1954; 16: 7-8.
- Shastri SVR: Asavakalpam, Vaidya Chandrika 1955; 17: 9.
- Vohra A and Satyanarayana T: Phytase production by the yeast Pichia anomala, Biotech Lett 2001; 23: 551-554.
- Vohra A and Satyanarayana T: A cost-effective cane molasses medium for enhanced cell bound phytase production by Pichia anomala. Journal of App Microbiology 2004; 97: 471-6.
- Dash VB and Hashyap VL: Materia Medica of Ayurveda. Concept Publishing Company, New Delhi 1987: 188-203.
- Kroes BH, Vanden Berg AJJ, Abeysekera AM, De Silva KTD and Labadie RP: Fermentation in traditional medicine: the impact of Woodfordia fruticosa flowers on the immunomodulatory activity and the alcohol and sugar contents of Nimba arishta.J Ethnopharmacol 1993; 40: 117-125.
- Bhardwaj S, Achliya GS, Meghre VS, Wadodkar SG and Dorle AK: In-vitro antibacterial activity of Takrarishta – an Ayurvedic formulation. Indian J Traditional Knowledge 2005; 4: 325-328.
- Santosh MK, Shaila D and Rao IS: Standardization of selected asavas and arishtas. Asian J Chem 2003; 15: 884-890.
- Ough CS: Fermentation rates of grape juice. III. Effect of initial alcohol, pH and fermentation temperature. Am J Enol Viticult 1966; 17: 74-81.
- Schneyder J: Einluss der verschiedenen Verarbeitungs verfahren und Lager ungsbedin- gungen auf die Entwicklung des Vitamin- gehaltes haltbar gemachter traubensafte. Mitt. (Klosterneuberg), Ser. A: Rebe Wein 1964; 14: 282-298.
- Fehling H: The quantitative determination of sugar and starch by means of copper sulphate. Eur J Org Chem 1849; 72: 106-113.
- Moore S and Stein WH: In: Methods in Enzymol. Academic Press, New York 1948; 3: 468.
- Robert EB: Method for estimation of tannin in grain sorghum. Agro J 1971: 511-512.
- Borzani W, Garab A, Pires MH, Piplovic R and De Ia Higuera GA: Batch ethanol fermentation of molasses: a correlation between the time necessary to complete the fermentation and the initial concentrations of sugar and yeast cells. World J Microbiol Biotechnol 1993: 265-68.
- The Indian pharmacopeia commission, Ghaziabad 2007: 2.
How to cite this article:
Thirukannan S, Krithivel MK, Ilansuriyan P and Marimuthu M: Physico-chemical changes that occurs during fermentation of Arishtam (a hydro alcoholic class of drug of Ayurveda). Int J Pharmacognosy 2015; 2(8): 391-99. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.2(8).391-99.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
3
391-399
818
1620
English
IJP
S. Thirukannan, M. K. Krithivel, P. Ilansuriyan and M. Marimuthu *
Nutraceutical Chemistry Lab, Department of Food Process Engineering, School of Bioengineering, SRM University, Kattankulathur, Tamil Nadu, India.
marimtu@gmail.com
15 June 2015
15 July 2015
14 August 2015
10.13040/IJPSR.0975-8232.IJP.2(8).391-399
31 August 2015