MICROSPHERE FORMULATION OF ESSENTIAL OIL OF ACORUS CALAMUS L. CONTROLS ASPERGILLUS NIGER GROWTH IN FINISHED LEATHER AT AN EXTENDED PERIOD OF STORAGE: FEATURING EXTRACTION, FORMULATION AND EVALUATION
HTML Full TextMICROSPHERE FORMULATION OF ESSENTIAL OIL OF ACORUS CALAMUS L. CONTROLS ASPERGILLUS NIGER GROWTH IN FINISHED LEATHER AT AN EXTENDED PERIOD OF STORAGE: FEATURING EXTRACTION, FORMULATION AND EVALUATION
S. Iswarya 1, T. S. Subha 1, V. John Sundar 2 and A. Gnanamani * 1
Microbiology Division 1, Tannery Division 2, CSIR-CLRI (Central Leather Research Institute) Adyar, Chennai - 600020, Tamil Nadu, India.
ABSTRACT: Plant oils have been used for a variety of purposes for many thousands of years. In particular, the anti-microbial & anti-fungal activity of plant extracts and oils have formed the basis of many applications. Since, Aspergillus niger contamination in leather and leather apparels reduces the export value considerably, demand an appropriate solution. The present study emphasizes the anti-Aspergillus potential of an essential oil and microsphere formulation in combating the A. niger growth in finished leather. A. niger ATCC 16888 strain was chosen for the present study. Both well diffusion and broth dilution methods are used. Mode of action and microsphere formulation and its impact on leather studied using standard protocols. Essential oil (EO) of Acorus calamus L. exhibited appreciable Anti-Aspergillus potency compared to untreated control and standard drugs (Itraconazole & Voriconazole). The oil contains components in addition to asarone. The damage in the plasma membrane and reduction in ergosterol content authenticate the antifungal action EO. The new microsphere formulation of an essential oil appreciably prevent the growth of A. niger when tested in leather for more than 90 days compared to free oil wherein reoccurrence was evidenced within 60 days.
| Keywords: |
Acorus calamus L., Aspergillus niger, Asarone, Microsphere, Antifungal
INTRODUCTION: Acorus calamus Linn., commonly known as “Sweet flag” has a long history of traditional uses such as an appetizer, anaesthetic agent, bronchitis, nervous & vascular disorders and sedative 1. The rhizomes have also been considered for a laxative, expectorant, antispasmodic and anthelmintic according to Ayurvedic system of medicine 2. Thus, a description of Acorus calamus L. exists in the Theatrum Botanicum of Parkinson (1640), and it has been listed since then in some Pharmacopoeia and Codices.
Also, in the recent past, the antifungal activity of essential oil of A.calamus L. has been in reports 3, 4 and anti-aspergillus activity and anti-yeast activity of beta-asarone of A.calamus L., has been reported 5, 6. Acorus calamus L. (rhizome) contains an essential oil (2 to 9%), which consists of a mixture of phenylpropanoids 7. The essential oil of Acorus calamus L. also contains thermophilic (dimethoxy benzoquinone), which is known to be an anti-microbial compound 8.
Owing to the recent consumer perception of the use of different synthetic antimicrobials in health care results in safety and environmental risks and subsequent development of new physiological races of pathogens. Hence, there is a renewed interest in the application of botanical antimicrobials for health care. Moreover, biologically active plant-based chemicals are expected to be more advantageous over synthetic antimicrobials, as they are easily biodegradable, Eco-friendly and mostly without significant residual or health risk issues. Despite extensive research on botanicals, broad-spectrum plant-based antimicrobials are still explored for the primary health care of an individual. About Acorus calamus, in recent years, it has been reported for several important biological activities such as anti-inflammatory 9, immunomodulatory 10, antioxidant 11, 12, anticonvulsant 8, antidiabetic 13, anticellular 10, antibacterial, allelopathic, immunosuppressive 7, 14 and pesticidal property 15.
In rural areas, the paste of the Acorus calamus rhizome along with ghee, honey, water was applied on the tongue of the newborn to improve speech, memory power and visual power 16. Also, it also stimulates the nervous system and to get rid of depression. Several sleeping problems, drowsiness, and even sinusitis can be treated by inhaling the dried rhizome powder 17. The decoction of the rhizome powder is used to get relief from cough, fever, epilepsy and abdominal pain 18. Acorus calamus rhizome paste can be used as a topical agent to treat skin related problems 1.
About fungal contaminations in leather and leather apparels, a drastic decrease in export value necessitates the potential anti-mold agent. Moreover, Aspergillus species share the major percentage of contamination concerning leather-based products, research initiatives on anti-aspergillus agents need intensive research. In addition, since, the application methods, the concentration of the anti-fungal agents and the period of existence of antifungal agents play the major role in combating the growth of Aspergillus species, research inputs on new formulations to increase the efficacy and the sustainability of the agents and the release of the agents are required.
Therefore, by keeping in view the numerous use of Acorus calamus L. in various traditional systems of medicine, it was desired to explore the anti- Aspergillus potential of essential oil of Acorus calamus L. rhizome. Followed by extraction, chemical characterization and the mode of action studies were carried out, in addition, to increasing the efficacy of the essential oil through new formulation and evaluated the anti-aspergillus efficacy of the new formulation in finished leathers.
MATERIALS AND METHODS: Aspergillus niger ATCC16888 strain obtained from the American Type culture collection was used for the present study as a representative organism. Czapexdoxagar, voriconazole, itraconazole, triton X 100, Yeast extract and Sucrose were procured from Himedia, Mumbai, Maharashtra, India. Sodium tripolyphosphate, XTT (2,3-bis- (2- methoxy- 4-nitro- 5-sulfophenyl)-2H-tetrazolium-5-carboxanilide) and sebacic acid were procured from Sigma-Aldrich, USA. All other chemicals (Laboratory grade) were obtained from S.D. Fine chemicals, Mumbai, Maharashtra State, India.
Acorus calamus L. (rhizomes) was procured from M/s JK Herbal farm, Veera Kaur, Perambalur district, Tamil Nadu, India and authenticated by Dr. T. Anandhan, Assistant Director (Retd.), Siddha Central Research Institute, Chennai. A voucher sample of the identified plant was deposited in National Institute of Herbal Science-Plant Academy Research Centre, Chennai for further authentication.
Extraction of Essential Oil: Acorus calamus L. (rhizomes) was shade dried and then pulverized to a fine powder and subjected to hydrodistillation process in a Clevenger type apparatus for 4 h. The essential oil obtained was concentrated using a rotary vacuum evaporator (Buchi, Japan) at about 40-60 ºC along with sodium sulfate and stored in sealed amber glass vials at 4 ºC until use.
Characterization Studies on Essential Oil (EO):
Instrumental Analyses:
UV-visible Spectrum: UV-visible spectrum of EO was measured using Shimadzu spectrophotometer UV 2450(Japan), in the range of 200-800 nm at 1 nm resolution.
Fourier Transform Infra-Red (FT-IR) Spectrum: Fourier Transform Infrared (FT-IR) spectrum was recorded in Spectrum-I FT-IR spectrophotometer (Perkin-Elmer Co, USA.). A 20μl of EO was mixed thoroughly with 200μg of Potassium Bromide (Sigma, USA) and pelletized using a hydraulic press. FT-IR spectrum was measured in the range of 4000-400cm-1 at a resolution of 4 cm-1. All the measurements were of 20 scans with potassium bromide as background reference.
Gas Chromatography-Mass Spectroscopy: GC-MS analysis was performed in Shimadzu Model QP2010 with head space analyzer, quadrupole detector using DB-5 MS column (30m length X 0.25mm ID X 0.25μm) operated under following conditions viz., manual injection; initial oven temperature as 40 ºC; injector temperature as 200 ºC; Ion source temperature as 200ºC; interface temperature as 240 ºC. Helium (99.999%) was used as a carrier gas at the flow rate of 1.36 ml/min. Eluent from GC column was directly introduced into the source of mass spectra and spectra obtained compared with that of peaks with Wiley and NIST libraries and with published data.
1H and 13C-NMR spectral analysis: 1H and 13C NMR of the sample was recorded using JEOL ECA 500 MHz spectrophotometer equipped with Delta version; irix platform operating at 500 MHz (1H) and 125 (13C). Sample concentration was maintained as 0.5mM per ml in 0.6ml CDCl3 solution (Deuterated, Sigma, USA, 99.99%) in a 5 mm OD wilmad precision NMR tube. 1H and 13C chemical shifts of solvent (7.27 and 77.0 ppm) used as a secondary reference and referred to TMS signal using usual relationships. Proton NMR (1H/pNMR) spectra were collected as 16K points over an x-sweep till 20 ppm, spectral width using a 90° pulse with a relaxation delay of 5 sec. The 13C NMR spectrum was collected with 30° pulse as 32K points with a relaxation delay of 2 sec.
Formulation of EO: The essential oil obtained from the above said procedure was formulated into microspheres for effective usage and applications. The formulation made according to the protocol summarized 19 with slight modifications wherein, chitosan polymer used for the formulation was dissolved and stabilized with sebacic acid instead of acetic acid and glutaraldehyde 20. The modified procedure followed in the present study completely avoided the use of glutaraldehyde for the stabilization of the spheres. In detail, to the homogenized solution of 2% (w/v) of chitosan dissolved in 1% (w/v) sebacic acid, 1ml of essential oil was added dropwise and again homogenized at 6000 rpm for 2-4h for uniform mixing to facilitate the proper cross-linking and stabilization Scheme. The obtained chitosan microspheres were washed several times with acetone to remove the oil globules that are adhered on the surface of the spheres and then freeze-dried and stored at 4 ºC until further applications.
Characterization of Microspheres: The spheres obtained were subjected to characterization as described in the following paragraphs.
Percentage Yield: The yield of microspheres was calculated based on the quantity of polymer and oil used in microsphere preparation.
% Yield = Practical value / Theoretical value × 100
Swelling Property: Swelling index of the microspheres was assessed according to the method summarized 21. In brief, a known amount of pre-weighed microspheres was soaked in phosphate buffered saline for 24h and the increase in weight after the scheduled period was measured. The degree of swelling was calculated according to the equation summarized below
The degree of swelling = [(Wt – Wo)/Wo] × 100
Where, Wt is the weight of the microspheres at time t, Wo is the initial weight of the microspheres.
Incorporation Efficiency: Essential oil incorporation efficiency of the microspheres was calculated according to the procedures summarized 22. A known amount of microspheres were soaked in DMSO for 24hr and then centrifuged at 5000 rpm. The entrapped amount of oil in the supernatant was measured spectrophotometrically.
Incorporation Efficiency = Mactual / Mtheoretical × 100
Mactual = Actual oil content in the weighed quantity of microspheres, MTheoretical = Amount of oil added during the formulation process.
In-vitro Release of EO: In-vitro, EO release studies were carried out as per the protocol summarized 23. In detail, a known amount of microspheres were suspended in 10ml of PBS (pH 7.4) and kept under stirring condition (100 RPM). For every 1hr, 1.0 ml of sample was removed, and it was replaced with 1.0 ml of fresh PBS to maintain the volume. The sample removed was centrifuged at 10,000 rpm for 5-10 min, and the supernatant was analyzed spectrophotometrically (300 nm). Essential oil release kinetics determined using kinetic models.
Anti- Aspergillus Activity of Free Oil and the Oil in the form of the Microsphere:
Well Diffusion method: Initial screening on Anti-Aspergillus activity was carried out using the standard procedure as summarized in CLSI (1990). In brief, pre-sterilized Czapexdox agar (CPD agar) was poured aseptically in a petri dish and allowed to solidify. A 5 days old A.niger ATCC 16888 4×105spores/ml suspended in Triton X100 (1%) were swabbed or spread using L-rod. Wells was created using well borers of size 10mm and to that essential oil (free form) of Acorus calamus L. at two different concentrations (5 and 10µg) was introduced and the plates were incubated at 37ºC for 7 days. In the case of microsphere formulation, 20 mg of microspheres dissolved in DMSO was used. At the end of the incubation period, the zone of inhibition was measured in millimeter (mm). Voriconazole (30 mg) and Itraconazole (1 mg) antibiotic discs were used as references.
Determination of Minimum Inhibitory Concentration (MIC): Minimum Inhibitory Concentration (MIC) was determined using a standard protocol (CLSI 1990). In brief, to each test tube add 3 ml of yeast extract sucrose broth (YES Broth) with 4 × 105 of Aspergillus niger spores. Different concentration of essential oil of Acorus calamus L. (0.5, 1.0, 2.0, 5.0, 10.0, 20.0 µg /ml) were added and incubated at 37 ºC for four days. The broth without oil served as a positive control. The concentration in which no visible fungal growth compared to that of neat control was recorded as the MIC value. Voriconazole and Itraconazole antibiotics were used as reference compounds. The experiments were done as triplicates, and the results were expressed as mean ± SD values.
Mode of Action of Essential Oil against A. niger: Mode of action of EO of Acorus calamus L. determined in terms of determining lesion of the plasma membrane, ergosterol quantification and mitochondrial dehydrogenase activity assay 24. With respect to plasma membrane lesions determination, spore suspension of A. niger ATCC 16888 containing 4 × 105 spores/ml was taken in a test tube, and the requisite amount of essential oil was added in such a way to get the final concentration of 0, 0.2, 0.4, 0.6, 0.8, 1.0 µg/ml respectively and incubated for the period of 12h at 28 ±2 ºC in an incubator under shaking condition (100 RPM). Colony forming units (CFUs) for each concentration was assessed by spread plate method. For the quantification of ergosterol content upon treatment of EO, a requisite amount of A. niger ATCC 16888 spore suspension (4×105spores/ml) was inoculated in Czapex Dox Broth (CPD) medium containing 1.0 µg/ml of essential oil of Acorus calamus L. and incubated for four days at 28 ± 2 °C. Tubes without oil were served as a control. Followed by incubation, mycelial growth was harvested and washed twice with distilled water. Then re-suspended in 5 ml of 25% alcoholic potassium hydroxide and vortex the mixture for 2min and then incubated at 85 ºC for 4h. Sterols were extracted from each sample by adding a mixture of 2 ml sterile distilled water and 5ml n-heptane at room temperature. The non-aqueous layer was separated and subjected to UV-visible spectral analysis in the spectral range of 230-300 nm and the ergosterol was quantified as determined.
Concerning mitochondrial dehydrogenases activity assessment, 2×106 spores/ml of A. niger ATCC 16888 was added to each well of 96 well plates and then incubated with different concentrations of essential oil (0, 0.2, 0.4, 0.6, 0.8, 1.0 µg/ml). Wells without oil served as a control. The plates were incubated for 24h at 28°C. Followed by incubation, a 50µl solution containing 50µg/ml XTT and 25µM Menadione was added, and the incubation was continued for the next 2 h. The optical density of the resulting solution was measured at 450 nm using plate reader supplied by BioTek Instruments, India.
Application Studies in Finished Leather: Application and testing studies were carried out according to standard ASTM methods (ASTM 4576-01). Two different procedures were followed in the present study. In the first procedure, a 5 × 5 m2 finished leather obtained from Tannery Division of CLRI premises has been exposed to the formulation by a hand spray technique. The microsphere loaded leather samples were then exposed to A. niger spores according to the standard procedure followed in ASTM and kept under humid condition (humidity: 80%, temperature 25 °C) for 7 days. For comparison, the leather sample exposed to EO alone was also made and exposed to fungal spores and incubated under humid condition. In the second procedure, the leather sample was pre-exposed to 107-109 spores of Aspergillus niger ATCC 16888 strain and then treated with microsphere formulation by hand spray technique and incubated for 7 days. Leather sample without microsphere formulation was considered as a control.
RESULTS AND DISCUSSION: Leather industry, one of the major revenue generating industry in India, often facing problems during export due to mold growth or fungal contaminations, despite, the incorporation of effective preservatives during finishing operations, which poses a big challenge to the leather sector. Compared to chrome tanned leathers, vegetable tanned leathers (the high demand at an international market) are highly prone to mold growth. Among the various mold showing growth, the predominant one was Aspergillus niger. Hence, efforts have been taken to combat A. niger growth in vegetable tanned leather. The permissible level of synthetic biocides currently in use could not be able to control the growth and reoccurrence of mold growth in the finished leather. The possibility of the use of a higher concentration of biocides reflected as pollution load in the form of high chemical oxygen demand in wastewater generated is not an acceptable solution. Suggestions to use non-polluting agents as fungicides, i.e., in the form of natural organics or biodegradable agents have been the most suitable solution. But, the issue on availability, quantity and nil issue on reoccurrence are the points that need to be considered before application.In India, numerous reports suggested that a number of traditional medicinal plants demonstrate anti-fungal activity. But, concerning A. niger, only a very few plants showed the efficacy that too only at higher concentrations. Several previous and recent studies have described many important biological activities, especially, the antimicrobial property of Acorus calamus L. roots, rhizome, and essential oils 6. It has been observed that at lower concentrations reoccurrence was there.
In general, compared to a crude extract of whole plants/ flowers alone/leave alone/rhizome alone, etc., the essential oils displayed appreciable antifungal activities because of the presence of low molecular weight mono and sesquiterpene hydrocarbons and their oxygen analogs and phenol derivatives. Small size molecules of essential oils easily penetrate through the cell wall and affect the various biochemical processes 25 of the microorganisms. Essential oil of clove 26, cinnamon 27, ginger, turmeric 28 for anti-fungal activity were in reports, and few papers studied the mode of action of essential oil on the destruction of fungal spores. All the said essential oils were studied for anti-aspergillus activity under in-vitro conditions and not much explored for new formulations and applications studies in leather and leather products. In the present study, essential oil of Acorus calamus L. after extraction has been subjected to characterization using various instrumental analyses and formulated into microspheres as summarized above in materials and methods. The yield of the essential oil in the present study was estimated at 6 %, whereas it was around 3.6-5.5 % 29. The oil obtained was reddish brown with a pleasant odor Table 1.
TABLE 1: PHYSICAL CHARACTERISTIC FEATURES OF CRUDE ESSENTIAL OIL OF ACORUS CALAMUS L.
| S. no | Physical properties | Essential oil of Acorus calamus L. |
| 1. | Color | Reddish brown |
| 2. | Odor | Pleasant |
| 3. | pH (at 30ºC) | 6.5 ± 0.2 |
| 4. | Specific gravity (at 30ºC) | 0.95 ± 0.02 |
| 5.
|
Physical appearance
|
 |
UV-Visible spectral analysis of the essential oil of Acorus calamus L. showed two absorption peaks at UV region Fig. 1A, and the peak at 282 nm suggested the presence of Asarone 30. FT-IR spectral information Table 2 of essential oil shown in Fig. 1B suggests the presence of aromatic and phenolic moieties, which matches with the structure of asarone 29, 30. About differential scanning calorimeter (DSC), three endothermic peaks at 125.18 and 161.31 and 204.74º C Fig. 1C were observed. These peaks suggest the presence of constituents other than asarone. Thermogravimetric analysis Fig. 1D reveals 50% weight loss at 242 ºC and infers the presence of both volatile and thermal stable components in the mixture. Gas-liquid chromatography and mass spectroscopy (GC-MS) analysis Fig. 1E suggested that other than asarone, the presence of long-chain unsaturated fatty acids was evidenced 31, 32.
FIG. 1: INSTRUMENTAL ANALYSIS (UV-VISIBLE, FTIR, GC–MS (CHROMATOGRAM AND MASS FRAGMENTATION), 1H AND 13C NMR SPECTRUM) OF ESSENTIAL OIL (EO) OF ACORUSCALAMUS L.
TABLE 2: PEAK DETAILS OF ESSENTIAL OIL OF A. CALAMUS L. OBTAINED FROM FTIR ANALYSIS
| S.
no |
Wave number (cm-1) | Assignment |
| 1 | 2996.2 | Methyl group |
| 2 | 2930.8 | Methylene group |
| 3 | 2850 | Symmetric vibration of the methylene group |
| 4 | 1738.5 | Ester group |
| 5 | 1611.5 | Carboxylate group |
| 6 | 1511.5 | Aromatic ring structure |
| 7 | 1319.2 | Phenol or tertiary hydroxyl (OH) group |
| 8 | 1211.5 | Tertiary amine |
| 9 | 1034.6 | Sulphonamides or phosphate ion |
| 10 | 861.54 | Aromatic compound |
| 11 | 761.5 | 1, 3 disubstitution meta group. |
The essential oil of Acorus calamus L. was formulated into microspheres by emulsification technique. About the characterization of microspheres, the protocol followed in the present study using chitosan and sebacic acid yielded 60.1% microspheres for the volume of 1grams of chitosan and 0.1g sebacic acid. Swelling index or drug loading efficiency and drug incorporation efficiency were calculated as 80% and 75% respectively.
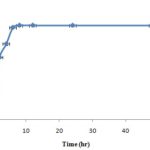
In general, the formulation was intended for the extended release of drug and time required for the maximum percentage of drug release has been considered as the important response variable in the formulation. There are many factors that play a major role in the extended release of drug-like a drug to polymer or polymer cross-linker ratio and agitation time 33. Chitosan microspheres dissolve faster in acidic media than the alkaline media. Therefore, the study was carried out in Phosphate buffer (pH 7.4), and the EO release profile was shown in Fig. 2. It has been found that there was no rapid release of EO as expected with other drugs.
FIG. 2: RELEASE PROFILE OF EO FROM MICROSPHERES
The release was slow and stabilized after 4 h. The essential oil in the interior of the microspheres was trapped by the chitosan matrix and not released within the short term until the microspheres degraded by enzymes in the biological environment 34. The chitosan microspheres showed marked retardation in the release profiles due to effective cross-linking density. Thus, the sebacic acid cross-linked chitosan microspheres were potentially useful for controlled release of essential oil. To investigate the mechanism of drug release, the release data were analyzed using various models. Release kinetics of the microsphere matrix was depended on the various factors, i.e. type of polymer used, the concentration of polymer, a drug to polymer ratio, the solubility of the drug and dispersed phase to continuous phase ratio. These variables directly affect the loading efficiency of the microspheres 35. Release kinetics of EO loaded chitosan microspheres showed linearity with R2 value near to unity (0.950) indicating the possibility of zero order kinetics.
FIG. 3: ANTIFUNGAL ACTIVITY OF ACORUSCALAMUS L. ESSENTIAL OIL AGAINST A. NIGER ATCC 16888 AT 5µG (a) AND 10µG (b) CONCENTRATION COMPARED WITH STANDARD DRUG ITRACONAZOLE (c) AND VORICONAZOLE (d) AGAINST A. NIGER ATCC 16888 AND UNTREATED PLATE SHOWING NORMAL GROWTH
About the results on the anti-Aspergillus activity of essential oil of Acorus calamus L. against A. niger ATCC 16888, the zone of inhibition was concentration dependent, and the maximum zone of inhibition (31±1mm diameter) Fig. 3 was observed with 10µg of essential oil, whereas, complete growth was observed in control plates. The zone of inhibition for standard drugs observed was of 20±1 and 37 ± 1 mm respective to Itraconazole and Voriconazole 36. Results of the present study corroborate well with reports 5, 6.
The Leaf extract of Acorus calamus was fractioned and purified the active constituent asarone and reported that 80µg/ml of concentration completely inhibit the growth of Aspergillus species which is very high compared with the inhibition concentration observed in the present study 37. It was studied, that the anti-Aspergillus activity of the essential oil of Acorus calamus and observed that 0.4µl/ml was sufficient to inhibit the fungal growth and in addition, 0.25 µl/ml inhibit the aflatoxin production, which was lower than the reference compound nystatin (≥2.5 µl/ml) 38. Ethanol and Ethyl acetate extract of Acorus calamus posses highest antifungal activity with a maximum zone of 20mm which is very less when compared with the observation made in the present study 39.
In the present study, Minimum inhibitory concentration (MIC) of essential oil (EO) was in the range of 1µg concentration. This concentration completely inhibits mycelial growth and the vegetative phase Fig. 4. The mode of action of EO on A.niger assessed using lesion of the plasma membrane of A.niger spores upon exposure to EO followed by growth suggested that appreciable decrease in the number of spores as shown in Fig. 5a 24. It was observed the lesion of the plasma membrane of A.flavus upon treatment with dill oil. Fig. 5b depicted the ergosterol content of the plasma membrane upon exposure to essential oil and suggested that ergosterol biosynthesis was reduced significantly 24. The reduction percentage of ergosterol content in the plasma membrane compared with the control was found to be 71.1% for 1µg concentration of EO.
It was observed a 79.4 % reduction in ergosterol content upon treatment with Dill oil (Anethum graveolens). Further, assessment of mitochondrial dehydrogenase profile study using XTT suggested the reduction in dehydrogenase activity and it was dose-dependent as shown in Fig. 5c 24.
FIG. 4: MIC ASSAY - ESSENTIAL OIL OF ACORUS CALAMUS L. TREATED TUBES WITH VARIED CONCENTRATIONS OF TEST COMPOUNDS (0.5-20µg/ml). RESULT WAS COMPARED WITH THAT OF UNTREATED (CONTROL TUBE SHOWING NORMAL GROWTH OF A. NIGER ATCC 16888).
FIG. 5: MODE OF ACTION ASSESSMENT: a) LESION OF PLASMA MEMBRANE. b) ERGOSTEROL BIOSYNTHESIS- UNTREATED AND TREATED WITH 1µl/ml OF ACORUSCALAMUS L. OIL. c) MITOCHONDRIAL DEHYDROGENASE ACTIVITY SHOWED REDUCTION IN THE MITOCHONDRIAL IN DOSE DEPENDENT MANNER COMPARED TO THAT OF UNTREATED CONTROL (0% CELL DEATH)
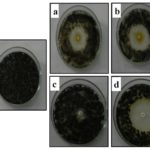
Essential oil encapsulated microspheres received from the present method suggested that oil was well encapsulated in the spheres. The anti-Aspergillus efficacy of microspheres was tested by diffusion methods and the results shown in Fig. 6 implied, compared to the virgin essential oil of Acorus calamus L., the formulations demonstrated higher efficacy by completely inhibiting the growth and the inhibition zone of about 41± 1mm in the case of microspheres was observed. This could be due to the increased bioavailability of drugs owing to high surface to volume ratio and also due to specific targeting of drugs to the absorption site. This also proves the potential of formulations for the treatment of infections caused by such microorganisms.
FIG. 6: ANTIFUNGAL ACTIVITY OF MICROSPHERES OF ACORUS CALAMUS L. ESSENTIAL OIL AGAINST A. NIGER ATCC 16888 a) CONTROL b) MICROSPHERES (20 mg)
FIG. 7: LEATHER APPLICATION STUDY a) CONTROL b) EO TREATED c) MICROSPHERE POST-TREATMENT d) MICROSPHERE PRE-TREATMENT. Fungal recurrence after 50 days, dead fungal spores
Further, the two different procedures followed on the application studies revealed interesting observations Fig. 7. Finished leather with precoated microspheres does not allow the applied A. niger spores to sporulate for more than 90 days. Whereas, application of free essential oil lost its activity within 50 days and slow growth of A. niger spores was visualized from day 60 onwards. Concerning the application of microspheres to the already grown A. niger in finished leather showed the death of spores within 14 days, but further incubation under humid condition reoccurrence of A. niger growth was observed. These observations suggested that microsphere encapsulated with essential oil is applied at the time of finishing appreciably control the growth of mould in the form of A. niger, whereas, for the already contaminated leather, the control was partial, and the re-growth of the fungal species suggested that the spores may be intact with the fibrillar structures, and the penetrability of microspheres was not achieved and thus exhibited the reduction in efficacy.
CONCLUSION: In conclusion, the observations made in the present study revealed that the essential oil of Acorus calamus L., exhibited statistically significant antifungal activity. The mode of action studies indicates that the essential oil of Acorus calamus L. interact with the plasma membrane of A. niger ATCC 16888 and reduces the ergosterol content substantially. Also, essential oil of Acorus calamus L. also inhibit the acidification of external medium and mitochondrial dehydrogenases activity of A. niger ATCC 16888. Further, the new formulation method followed in the present study suggested the effective use of EO in controlling the mold growth. The study carried out with finished leather gave an interesting observation and the application of microsphere entangled EO effectively control the growth when it has been applied during the manufacturing process. However, for the already grown mold, the new formulation can able to kill the spores, but, the reoccurrence was unavoidable due to the intact spores present in between the fibrillar structure of the material.
ACKNOWLEDGEMENT: The authors greatly acknowledge the Council of Scientific and Industrial Research, New Delhi for the financial assistance provided in the form of CSC0201.
CONFLICT OF INTEREST: There is no conflict of interest in this study.
REFERENCES:
- Kirtikar KR and Basu BD: Indian Medicinal plants, Mahendrapal Singh Publishers, Edition 4th, 1987.
- Anonymous: The Wealth of India: A dictionary of Indian Raw materials and Industrial products, Edition 1st, 2001. India.
- Thirach S, Tragoolpua K, Punjaisee S, Khamwan C, Jatisatienr C and Kunyanone N: Antifungal activity of some medicinal plant extracts against Candida albicans and Cryptococcus neoformans. Acta Horticulturae 2003; 597: 217-221.
- Rajput SB and Karuppayil SM: β-Asarone, an active principle of calamus rhizome, inhibits morphogenesis, biofilm formation and ergosterol biosynthesis in Candida albicans. Phytomedicine 2013; 20: 139-142.
- Phongpaichit S, Pujenjob N, Rukachaisirikul V and Ongasakul M: Antimicrobial activities of the crude extracts of Acorus calamus Journal of Science and Technology 2005; 27: 517-523.
- Asha Devi S and Ganjewala D: Antimicrobial activity of Acorus calamus (L.) rhizome and leaf extract. Acta Biologica Szegediensis 2009; 53: 45-49.
- Balakumbahan R, Rajamani K and Kumanan K: Acorus calamus: An overview. Journal of Medicinal Plants Research 2010; 4: 2740-2745.
- Jayaraman R, Anitha T and Joshi VD: Analgesic and Anticonvulsant effects of Acorus calamus roots in mice. International Journal of Pharmaceutical Technology and Research 2010; 2: 552-555.
- Vohra SB, Shah SA, Sharma K, Naqvi SAH and Dandiya PC: Antibacterial, antipyretic, analgesic and anti-inflammatory studies on Acorus calamus, Annals of Natural and Academic Sciences 1989; 25: 13-20.
- Mehrotra S, Mishra KP, Maurya P, Srimaln CC, Yadav VS, Pandey R and Singh VK: Anti-cellular and immuno-suppressive properties of ethanolic extracts of Acorus calamus International Journal of Immunology and Pharmacology 2003; 3: 53-61.
- Acuna VM, Atha DE, Ma J, Nee MH and Kennelly J: Antioxidant capacities of ten edible Northa American plants. Phytotherapy Research 2002; 16: 63-65.
- Manikandan S and Devi RS: Antioxidant property of α-Asarone against noise stress-induced changes in different regions of rat brain. Pharmacology Research 2005; 52: 467-474.
- Lee MH, Chen YY, Wang SC, Watanobe T and Tsai YC: Inhibitory effect of β-Asarone a component of Acorus calamus essential oil on inhibition of adipogenesis in 3T3-L cells. Food Chemistry 2011; 126: 1-7.
- Paithankar VV, Belsare SL, Charde RM and Vyas JV: Acorus calamus: An Overview. International Journal of Biomedical Research 2011; 2: 518-529.
- Tariq RM, Naqvi SMH, Chaudhary MI and Abbas A: Importance and Implementation of essential oil of Pakistanian Acorus calamus As a Biopesticide. Pakistan Journal of Botany 2010; 42: 2043-2050.
- Meena AK, Rao MM, Singh A and Kumar S: Physiochemical and preliminary phytochemical studies on rhizome of Acorus calamus. International Journal of Pharmacy & Pharmaceutical Sciences 2010; 2: 130-131.
- Jadhav T: Sushruta Samhita, Chaukhambhasurbharthi academy, India, Edition 1st, 1994.
- Chellaiah M, Muniappan A, Nagappan R and SavrimuthuI: Medicinal plants used by traditional healers in Kanchipuram District of Tamil Nadu, Indian Journal of Ethnobiology & Ethnomedicine 2006; 2: 43.
- Roy S, Panpalia SG, Nandy BC, Rai VK, Tyagi LK, Dey S and Meena KC: Effect of method of preparation on chitosan microspheres of Mefenamic acid. International Journal of Pharmaceutical Science and Drug Research 2009; 1: 36-42.
- Sailakshmi G, Mitra T and Gnanamani A: Engineering chitosan and collagen macromolecules using sebacic acid for clinical applications. Progress in Biomaterials 2013; 2: 1-12.
- Chandrakala V and Mary Saral A: Optimisation and characterization of nasal microparticles for levadopa delivery to CNS. Research Journal of Pharmaceutical Biological Chemical Sciences 2013; 4: 882-897.
- Shanmugarathinam A and Puratchikody A: Formulation and characterization of ritonavir loaded ethyl cellulose buoyant microspheres. Journal of Pharmaceutical Sciences & Research 2014; 6: 274-277.
- Ibezim EC, Andrade CT, Marcia C, Barretto B, Odimegwu DC and De Lima FF: Ionically crosslinked chitosan / tripolyphosphate microparticles for the controlled delivery of pyrimethamine. Ibnosina Journal of Medicine and Biomedical Sciences 2011; 3: 77-88
- Tian J, Ban X, Zeng H, He J, Chen Y and Wang Y: The mechanism of antifungal action of essential oil from dil (Anethum graveolens) on Aspergillus flavus. Plos One 2012; 7: 1-10.
- Justasirvaityte, Siugzdaite J, Valeika V and Dambrauskiene E: Application of essential oils of thyme as a natural preservative in leather tanning. Proceedings of the Estonians Academy of Sciences 2012; 61: 220-227.
- Pinto E, Vale-Silva L, Cavaleiro C and Salgueiro L: Antifungal activity of the clove essential oil from Syzygium aromanticum on Candida, Aspergillus and Dermatophyte species. Journal of Medical Microbiology 2009; 58: 1454-1462.
- Sukatta U, Haruthaithanasan V, Chantarapanont W, Dilokkunananat U and Suppakul P: Antifungal activity of clove and cinnamon oil and their synergistic against postharvest decay fungi of grape in-vitro. Kasetsart Journal of Natural Sciences 2008; 42: 169-174.
- Norajit K, Nattalaohakunjit and Kerdchoechuen O: Antibacterial effects of five Zingiberaceae essential oils. Molecules 2007; 12: 2047-2060.
- Kasture A, Patel S, Chauhan J and Krishnamurthy R: In- vitro antimicrobial effect of Essential oil from leaf and rhizome of various accessions of Acorus calamus, and its Phytochemical Screening. European Journal of Medicinal Plants 2015; 9: 1-13.
- Saxena M and Saxena J: Evaluation of phytoconstituents of Acorus calamus by FTIR and UV-VIS Spectroscopic Analysis. International Journal of Biological and Pharmaceutical Research 2012; 8: 498.
- Gyawali R and Kim KS: Volatile organic compounds of medicinal values from Nepalese Acorus calamus. Kathmandu Univerity Journal of Science, Engineering and Technology 2009; 5: 51-65.
- Dissanayake MLMC, Shin Itci Ito and Yoshihiko Akakabe: TLC Bioautography guided Detection and biological activity of antifungal components from medicinal plant Acorus calamus Asian Journal of Plant Pathology 2015; 9: 16-26.
- Hemant KSY, Mangla N and Singh and Shivakumar HG: Chitosan / sodium tripolyphosphate cross-linked microsphere for the treatment of gastric ulcer. Der Pharmacia Lettre 2010; 2: 106-113.
- Yeo Y, Back N and Park K: Microencapsulation methods for delivery of protein drugs. Biotechnology and Bioprocess Engineering. 2001; 6: 213-230.
- Dhakar RC, Dattamaurya S, Sadar BPS, Bhagat S, Prajapathi SK and Jain CP: Variables influencing the drug entrapment efficiency of microspheres: A Pharmaceutical review. Der Pharmacia Letter 2010; 2: 102-116.
- Manavathu EK, Cutright JL and Chandrasekar PH: Organism dependent fungicidal activities of azoles. Antimicrobial Agents Chemotherapy 1998; 42: 3018-3021.
- Tiwari N, Chaudhary A, Mishra A and Bhatt G: Antimicrobial activity of β- asarone from Acorus calamus International Journal of Chemical & Analytical Sciences 2009; 1:211-213.
- Shukla R, Singh P, Prakash B and Dubey NK: Efficacy of Acorus calamus essential oil as a safe plant-based antioxidants aflatoxin B1 suppressor and broad-spectrum antimicrobial against food interesting fungi. International Journal of Food Science and Technology 2013; 48: 128-135.
- Kumar V, Singh R and Joshi V: Antimicrobial activity of rhizome extract of Acorus calamus against different microorganisms. Octa Journal of Biosciences 2014; 2: 59-63.
How to cite this article:
Iswarya S, Subha TS, Sundar VJ and Gnanamani A: Microsphere formulation of essential oil of Acorus calamus L. controls Aspergillus niger growth in finished leather at an extended period of storage: featuring extraction, formulation and evaluation. Int J Pharmacognosy 2016; 3(7): 295-05. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.3(7).295-05.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
2
295-305
842
1195
English
IJP
S. Iswarya, T. S. Subha, V. J. Sundar and A. Gnanamani *
Tannery Division, CSIR-CLRI (Central Leather Research Institute) Adyar, Chennai, Tamil Nadu, India
gnanamani3@gmail.com
20 June 2016
17 July 2016
29 July 2016
10.13040/IJPSR.0975-8232.IJP.3(7).295-305
31 July 2016