HERBAL DRUGS – AN APPROACH FOR THE TREATMENT OF LIVER DISORDERS
HTML Full TextHERBAL DRUGS - AN APPROACH FOR THE TREATMENT OF LIVER DISORDERS
Swapnil Pandey 1 and Dindayal Darunde * 2
Department of Pharmacognosy, Varanasi College of Pharmacy, Varanasi - 221105, Uttar Pradesh, India.
Government College of Pharmacy, Amaravati - 444601, Maharashtra, India.
ABSTRACT: The liver is an amazingly complex organ which virtually affects every physiological process of the body. The liver is the largest glandular organ in the body and has more function than any other organs. Hepatic disorders which stem from a stressful lifestyle, inappropriate eating habits, and lack of exercise have become one of the major causes of morbidity and mortality in human being. The acute hepatic symptoms may be cured by some general prevention such as avoidance of constipation and balance between intake quantity of protein and disaccharides in normal food. An alternative and progressively increasing adaptation is the use of herbal extracts. Many of the plant-based drugs show an effective response to managing the hepatotoxicity and secondary symptoms of liver damage. While, the models used for conventional hepatotoxicity evaluation are still outdated, in the present review we have attempted to provide updated information of the molecular pathogenesis and aspects for the role of herbal pharmacotherapy in the alleviation of hepatic ailments. Furthermore, we have attempted to summarize the critical findings on hepatoprotective herbs over the last 20 years.
| Keywords: |
Hepatotoxicity, Hepatoprotective Herbs, Hepatic injury, Hepatic function test, Silybum marianum, Allium sativum
INTRODUCTION: Medicinal plants may serve as a vital source of potentially useful new compounds for the development of effective therapy to combat a variety of disease. Herbal medicine is used by about 80% of the world population, primarily in developing countries for primary health care. Ancient literature also mentions herbal therapy for age-related diseases, namely memory loss, osteoporosis, diabetic wounds, immune and liver disorder, etc. (Brower et al., 1998), 1998. Indian traditional medicine like Ayurveda, Siddha, and Unani are predominantly based on the use of plant materials.
Herbal drugs have gained importance and popularity in recent year because of their safety, efficacy, and cost-effectiveness 1. Recently, World Health Organization defined traditional medicine including herbal drugs as the therapeutic practice that has been in existence, often for hundreds of years, before the development and spread of modern medicine and is still in use today 2. The association of medical plants with other plants in their habitat also influences their medicinal value in some cases. One of the most important and well-documented uses of plant product is their use as medication. Hence, there is an ever-increasing need for safe medication 3.
Role of Liver in Human Physiology: Liver is crucial to life. Because it is responsible for so many vital functions, when the liver is damaged, our health is affected. The liver can do 500 functions - that's some multitasking! This is one powerful organ, the one organ in the body that is capable of regenerating itself. There is no organ that is more important to healthy metabolism than the liver- in many ways; it is as central to metabolisms, the heart is to the circulation of blood.
The liver plays a critical role in four key areas of metabolism: fuel management, nitrogen excretion, the regulation water distribution between the blood & tissues, and the detoxification of foreign substances. It also produces prothrombin and fibrinogen which helps in blood clotting and heparin, a mucopolysaccharide, sulphuric acid and ester that helps keeps blood from clotting within the circulatory system. The liver converts to sugar into glycogen. The liver plays a vital role in disease-free life. Because of
- Storage of vitamins, minerals, and sugars.
- Filter your blood and remove harmful substances.
- Store extra blood for emergencies. Being prepared can be a lifesaver!
- It keeps the electrolyte balance maintained. Electrolytes like calcium and potassium help the heart to keep beating!
- It helps to utilize fat-soluble vitamins like A (for eyesight), D (helps calcium to absorb), E (good for wound healing), F (essential fatty acids for normal growth and behavior), and K (helps the blood to clot).
- It helps use or eliminates excess hormones.
- It creates bile, which helps break down fats.
- It helps to manage blood sugar - helping to keep blood sugar stable. Without the liver functioning correctly, it can lead to diabetes or hypoglycemia, or reactive hypoglycemia (highs and lows).
- Processing digested food from the intestines.
- Whatever wastes that the kidney does not remove from circulation, the liver removes from circulation.
- Clearing bacterial infection and combating infections in general. An impaired liver means an impaired ability to fight infections.
- Neutralizing toxins and drugs.
A Few Signs that the Liver Needs to be Cleansed could be (If Other Things are not Causing the Symptoms): If you experience any of the following symptoms, you may be experiencing autointoxication (a process whereby you are poisoned by substances produced by your own body as a result of inadequate digestion and elimination),
- Breaking out in acne - which, of course, is hard on the self-esteem,
- Hair breakage,
- Nightmares - bad dreams,
- Insomnia,
- Exhaustion,
- Flu-like feelings,
- Difficulty thinking or focusing,
- Pain under right rib,
- Blood sugar imbalance.
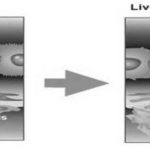
Cause of Liver Disorders: Liver diseases have become the major cause of morbidity and mortality in human being. Among the many diseases that can affect the liver, the most common is viral hepatitis Fig. 1. Hepatitis can be caused by drugs, viruses, bacteria, and parasites like amoebiasis and giardiasis. The use of natural remedies for the treatment of liver diseases has a long history, and medicinal plants and their derivatives are still used all over the world in the form or the older for this purpose 4. The protective liver plants contain a wide variety of chemical constituent phenols, coumarins, monoterpenes, carotenoids, glycoside, and polyphones. The main cause of hepatotoxicity is yet unknown. It appears to involve two pathways- direct hepatotoxicity and adverse immune reaction 5.
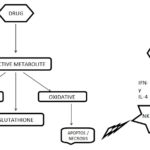
The most common hepatotoxicity induced by the bioactivation of drugs to the active metabolites, which can react interact with cellular macromolecules such as proteins, lipids and nucleic acid leading to protein dysfunction, lipid peroxidation, DNA damage and oxidative stress Fig. 2. The reactive metabolites may induce disruption of ionic gradients and intracellular calcium stores, resulting in mitochondrial dysfunction and loss of energy production.
FIG. 1: DIFFERENCE BETWEEN NORMAL LIVER CELLS AND INFECTED LIVER CELLS
FIG. 2: ROUTE OF HEPATIC INJURY
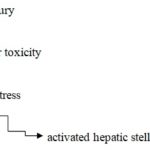
Its dysfunction release an excessive amount of oxidants, which in turn causes injury to hepatic cells. Activation of some enzymes in the cytochrome P-450 system such as CYP2E1 also leads to oxidative stress Fig. 3. Injury to hepatocyte and bile duct cells leads to accumulation of bile acid inside the liver. This promotes further liver damage. This impairment of cellular function can culminate in cell death and possible liver failure. Thus it is the delicate balance of inflammatory and hepatoprotective mediators produce after the activation of the innate system that determines an individual’s susceptibility and adaptation to hepatic injury 6.
FIG. 3: MECHANISM OF LIVER INJURY
Management of Hepatic Encephalopathy: Some most common factors which helps to manage the hepatic injury.
- Avoid constipation
- Avoid other precipitating factors
- Maintain adequate protein and energy intakes
- Non-absorbable disaccharides- lactulose 20-40 ml daily.
Pharmacological Evaluation of Hepato-protective Plants: To investigate hepatotoxic substances, it is customary to subject animals to a range of toxic substances.
These include carbon tetrachloride, alpha amanitine, and phalloidin, Paracetamol, liquid paraffin, thioacetamide, etc, which induce animals liver damage, and changes in serum ALT and AST and further histological observation are evaluated.
In-vivo Models: The various in-vivo models are studied for the evaluation of hepatic protective drugs. The models are listed below-
Paracetamol Induced Hepatotoxicity: The method of acetaminophen-induced acute hepatotoxicity can be used most widely. Albino rats are used for the pharmacological evaluation of the test drug. The standard and test drug of various concentrations are given to the rats, and different parameters like an evaluation of serum bilirubin, SGPT, SGOT are tested.
Carbon Tetra Chloride Induced Hepatotoxicity: The carbon tetrachloride is used for the induced hepatotoxicity into the animal. The animal shows fatty changes, gross necrosis, broad inflammation of the lymphocytes, and kupffer cells around the central vein. SGPT, SGOT, ALP serum bilirubin are a most sensitive test which is considered as an index to estimate the liver disease.
Thioacetamide Induced Hepatotoxicity: Adult female Wistar rats weighing 180-200 g are kept in wire-bottomed cages at control temperature with 12 h. The thioacetamide and test group received the saline from the rats and evaluated different parameters like SGPT, SGOT, ALP, and AST.
Alcohol and Carbon Tetra Chloride Induced Hepatotoxicity:
Carbon Tetra Chloride and Liquid Paraffin Induced Hepatotoxicity: 7
In-vitro Models: Developed in the past years. Next, to their use in drug development, they can also be applied to study environmental toxins and their hepatotoxicity. The 3 main approaches are ex-vivo isolated and perfused organ models, precision-cut liver slice, and cell culture models. Although the advantage of whole organ perfusions is based on the assessment of physiologic parameters such as bile production and morphologic parameters such as tissue histology, cell culture models can be efficiently used to assess cellular metabolism, cytotoxicity, and genotoxicity. The advantage of precision-cut liver slices is based on the juxtaposition of cellular assays and tissue morphology.
Hepatoprotective Herbs:
Silybum marianum: Milk thistle (Silybum marianum) has a long and important history in herbal medicine dating back over 2,000 years in European herbal traditions.
The root, leaf, and steam have medicinal use. But flavonolignans most widely used nowadays. The extracts were injected to the rats, at a dose of 25 mg kg-1 body weight together with thioacetamide at a dose of 50 mg kg body weight. Significant decrease in the activity of aminotransferases, alkaline phosphatase and bilirubin were observed in the groups treated with extracts and compared with the group that was treated only with thioacetamide 8.
Other plants dugs which are reported as hepatoprotective is given below:
TABLE 1: LIST OF VARIOUS HEPATOPROTECTIVE HERBS WITH PROPER BIOLOGICAL SCREENING METHOD
| S.
no. |
Plant
name |
Family | Part used | Model
used |
Animal
used |
Tested
parameters |
Dose/ route of administration |
| 1 | Andrographis
paniculata |
Acanthaceae | Leaves | Paracetamol-induced | Healthy adult male Wistar albino rats (weighing between 120-250g) | AST, ALT, GGT, ALP, LDH and bilirubin | 500 mg/kg b.w, p.o. 9 |
| 2 | Allium
sativum |
Liliaceae | Bulb | Isoniazid-induced | Wistar
albino rats |
ALT, AST,
ALP and Total Bilirubin |
0.25 g/kg/day, oral route 10,11 |
| 3 | Azadirachta
indica |
Meliaceae | Aerial parts | Paracetamol-induced | Male albino Wistar rats (100-150 g; 4-6 weeks old) | GST, GPX, SOD | 500 mg/kg,
p.o. 12, 13 |
| 4 | Boerrhavia
diffusa |
Nyctaginaceae | Whole plant | Thioacetamide | Albino Rat | GOT, GPT, ACP and ALP ,TG and ALT | 100mg/gmbw
14, 15 |
| 5 | Curcuma
longa |
Zingiberaceae | Rhizome | Paracetamol induced | Male albino Wistar rats (100-150 g) | ALT, AST, ALP | 600mg/kg oral route 16, 17 |
| 6 | Wedelia
calendulaceae |
Asteraceae | Leaves | Thioacetamide induced | Albino
rat |
SGOT, SGPT, ALP LP, GSH | (100, 200 and 400mg/kg B.W.) oral route 18 |
| 7 | Camellia
sinensis |
Theaceae | Leaves | Chloroform indcued | Male albino
rat |
CAT, SOD, GSH | 100mg/kg and 200 mg/kg 19 |
| 8 | Capparis
spinosa |
Capparidaceae | Leaves | Carbon tetra chloride induced | Male albino
rat |
ALT, AST | 100mg/kg, oral route 20 |
| 9 | Cassia
tora |
Leguminosae | Leaves | Galactosamine induced | Albino
rats |
ALT, AST, SGOT, SGPT, Bilirubin, | 100 and 250mg/kg i.p. 21 |
| 10 | Cichorium
intybus |
Asteraceae | Leaves | Chlorpromazine induced | Adult albino
rat |
Leukocytes | 300mg/kg 22 |
| 11 | Glycyrrhiza
glabra |
Leguminosae | Leaves | Carbon tetra chloride induced | Male albino
rat |
ALT, GST, GSH, SOD | 500 mg/kg, subcutaneous 23 |
| 12 | Ginkgo
biloba |
Ginkgoaceae | Whole
plant |
Carbon tetra chloride induced | Male albino
rat |
ALT, AST, ALP | 0.5 g/kg body weight per day, subcutaneous injection 24, 25 |
| 13 | Ocimum
sanctum |
Labiatae | Leaves | Paracetamol and carbon tetra chloride | Albino rats
(150–200 g) |
Total serum protein, albumin-globulin ratio, ALP, AST and ALT | 100 mg/kg BW/day, p.o. 26 |
| 14 | Phylanthus
niruri |
Euphorbiaceae | Leaves | Carbon tetra chloride induced | Mice | SGOT | 5 mg/kg body weight, orally & intraperitoneally27 |
| 15 | Rheum
emodi |
Polygonaceae | Root | Paracetamol-induced | Albino Rats
(150–200 g) |
ALT, AST, ALP, albumin and bilirubin (total and direct) levels | 2 g/kg, orally28 |
| 16 | Taraxacum
officinale |
Asteraceae | Whole plant | Galactosamine induced | Sprague-Dawley rats | Dandelion | 3% DWE diet, i.p. 29, 30 |
| 17 | Vitis
vinifera |
Vitaceae | Leaves | Carbon tetra chloride induced | Male albino
rat |
ALT, AST | 125 mg/kg dose (per os) 31 |
| 18 | Tephrosia
purpura |
Fabaceae | Roots & leaves | Galactosamine,
carbon tetrachloride induced |
Male albino
rat |
SGOT, SGPT, bilirubin level | 500mg/kg,
orally 32 |
| 19 | Tinospora
cordifolia |
Menispermaceae | Whole plant | Carbon tetra chloride induced | Adult male
albino rat |
SGOT, SGPT and ALP | 100mg/kg/d.
i.p. 33 |
| 20 | Zingiber
officinale |
Zingiberaceae | Rhizome | Ferric chloride induced | Sprague Dawley rats (150-170 g) | ALP, SGPT, SGOT, ALT, AST | 500 mg/kg,
orally 34 |
| 21 | Eclipta
alba |
Composite | Leaves | Paracetamol, carbon tetra chloride induced | Albino Wistar
rats |
Sleep time, zoxazolamine paralysis time, bromosulphaline clearance, serum trans-aminases, and serum bilirubin | 10-80 mg/kg,
p.o. 35 |
| 22 | Foeniculum
vulgare |
Umbelliferae | Fruit | Carbon tetra chloride induced | Sprague-Dawley rats weighing
180- 200 g |
Serum aspartate
aminotransferase, alanine amino-transferase, alkaline phosphatase |
0.3 ml/kg i.p. 36 |
| 23 | Trigonella
foenumgraecum |
Leguminosae | Fruit
|
Thiobarbituric-acid induced | Albino wistar
rats |
ALT, AST serum bilirubin | 200 mg kg−1 day−1, orally 37 |
| 24 | Ficus
carica |
Moraceae | Leaves | Carbon tetra chloride induced | Albino wistar
rats |
ALT, AST serum bilirubin | 500mg/kg 38 |
| 25 | Annona
squamosal |
Annonaceae | Leaves | Isoniazid+rifam-
picin induced |
Albino
rats |
Decreased ALT,AST, | 300mg/kg b.w, i.p. 39 |
| 26 | Lepidium
sativum |
Brassicaceae | Leaves | Carbon tetra chloride | Albino wistar
rats |
AST, ALT, ALP levels and bilirubin | 200 and 400 mg/kg body weight, i.p. 40 |
| 27 | Sargassum
polycystum |
Sargassaceae | Leaves | Galactosamine induced | Wistar strain male albino rats | ALT,AST | 125mg/kg b.w, orally 41 |
| 28 | Prostecheami-chuacana | Orchidaceae | Leaves | Carbon tetra chloride induced | Albino
rats |
Blood biochemical profile | 200-600 mg/kg b.w, orally 42 |
| 29 | Phyllanthus
amarus |
Euphorbiaceae | Leaves | Carbon tetra chloride induced | Albino
rats |
AST, ALT, SGOT | 25, 50 and 75 mg/kg, p.o. 43 |
| 30 | Fumaria
indica |
Fumariceae | Leaves | Paracetamol and carbon tetra chloride induced | Albino
rats |
Serum biochemical parameters | 10–20 mg p.o. 44 |
| 31 | Silybum
marianum |
Asteraceae | Leaves | Thioacetamide induced | Albino wistar
rats |
Aminotransferases, bilirubin, alkaline phosphates | 25mg/kg b.w,
p.o. 45 |
| 32 | Cassia
roxburghii |
Caesalpiniaceae | Seeds | Carbon tetra chloride +ethanol
induced |
Albino wistar
rats |
AST, ALT, SGOT | 250-500 mg/kg46 |
| 33 | Coccinia
grandis |
Cucurbitaceae | Leaves | Carbon tetra chloride induced | Albino Wistar
rats |
ALT, AST, amino transferases | 250 mg/kg 47 |
| 34 | Solanum
nigrum |
Solanaceae | Fruits | Carbon tetra chloride induced | Male albino
rats |
AST, ALT, ALP and total bilirubin | 250 mg/kg, p.o. 48 |
| 35 | Orthosiphon
stamineus |
Lamiaceae | Leaves | Paracetamol-induced | Male albino
rats |
Decreased in the level of ALT AST | 200 mg/kg 49 |
(s.c- sub cutaneous i.p.-intraperitoneal, b.w- body weight, AST- Aspartate Aminotransferases, ALT- Alanine Aminotransferases, ALP- Alkaline Phosphates, SGOT- Serum Glutamic Aminotransferases, SGPT- Serum Glutamic Phosphotransferases)
TABLE 2: LIST OF VARIOUS HEPATIC FUNCTIONS TESTS WITH THEIR INTERPRETATIONS 50
| Hepatic function test parameters | Abbreviations | Reference range | Interpretations |
| Albumin | Albumin | 3.5 to 5.3 g/dL | To assess the severity of liver injury (HIV infection and malnutrition may confound this) |
| Alkaline phosphatase | ALP | 30 to 120 IU/L | To diagnose cholestasis and infiltrative disease |
| Alanine transaminase | ALT | 7 to 56 IU/L | To diagnoses liver dysfunction |
| Anti-mitochondrial antibody | AMA | < 0.1 units | To diagnose primary biliary cirrhosis |
| Aspartate transaminase | AST | 6-40 IU/L | Elevated AST levels are not specific for liver damage, and AST has also been used as a cardiac marker |
| Bilirubin
(unconjugated) |
Bilirubin (unconjugated) | 0.1–0.4 mg/dL | To assess for
hemolysis |
| Bilirubin (total) | Bilirubin (total) | 0.1–1.0 mg/ dL | To diagnose jaundice and assess severity |
| Gamma-glutamyl transpeptidase | GGT | 0 to 42 IU/L | GGT is raised in chronic alcohol toxicity |
| Serum glutamic oxaloacetic transaminase | SGOT | 5 to 40 units per L | To diagnose hepatocellular disease and assess the progression of the disease |
| Serum glutamate pyruvate transaminase | SGPT | 7 to 56 units per L | ALT relatively lower than AST in persons with alcoholism |
DISCUSSION: Hepatoprotective disorder is the most common disorder and affects normal physiology of the liver. This review discusses the plant drugs which have shown the significant result as the hepatoprotective agent even in some cases with good potency. There is an increasing demand by the patient to use the natural product with hepatoprotective activity. A large number of herbal species has been used traditionally as a medicine against hepatotoxicity ailments. Many of them have been studied scientifically and proved to be beneficial for the liver as a hepatoprotective. The success has been attained to isolate various single chemical entities responsible for hepatoprotective activity 51.
Most of the plant extract is water soluble so the achievement of successful bioavailability is tough task. To overcome these problems different kinds of targeted formulation have been developed. In this aspect phytosomes and liposomes have emerged as prospective tools for delivery of bioactive to hepatic tissues. The different kind of marketed formulation such as silyphos phytosomes, ginkgo liposomes, quercetin phytosomes is available in the market which achieves maximum bioavailability 52.
CONCLUSION: Plants have played a remarkable role in health care since ancient time. Traditionally plant-based medicine exerts a great deal of importance to people living in developing countries and also lead to the discovery of new drugs. Herbal medicines make an enormous contribution to primary health development. In recent days, hepatotoxicity is a major cause for the human being, so this review includes all the study of plants drug which gives significant responsibility for the treatment of hepatotoxicity. The research of botanical medicines shows the different result for the treatment of liver dysfunction. The different herbal remedies such as a green tree, ginger, and curcumin are the well-known drugs for the treatment of acute liver toxicity.
In another way, many hepatic trails are done by the scientist today and much research yet to be done, but a list of these plants has an appreciable response for the treatment of viral hepatitis, cirrhosis of liver and liver toxicity. The single drug cannot show significant response; the combination of two or more plant extract may prove very effective treatment of liver disorders caused by over drinking of alcohol, toxic elements and different viral infections.
ACKNOWLEDGEMENT: I have taken efforts in this review paper. However, it would not have been possible without the kind support and help of Library Personals of organizations, thus I would like to extendmy sincere thanks to all of them.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Agarwal SS: Development of hepatoprotective formulations from plant sources: In Pharmacology and Therapeutics in the New Millennium. New Delhi, 2001: 357-58.
- Handa SS: Plants as drugs: The Eastern Pharmacist 1999; 79-85.
- Thyagarajan SP, Jayaram S, Gopalakrishnan V, Hari R, Jeyakumar P and Sripathi MS: Herbal medicines for liver diseases in India. J Gastroenterol Hepatol 2002; 370-6.
- Nadeem MPC, Dandiya PC, Pasha M, Imran D, Balani K and Vohora SB: Fitoterapia 1997; 68: 245-51.
- Lewis HW and Elvin-Lewis MPH: Medical Botany: Plants Affecting Man’s Health, John Wiley and Sons, New York: 1977: 217.
- Lynch T and Price A: The effect of cytochrome P-450 metabolism on drug response, interactions and adverse effects 391-396.
- Mukharjeek Pulok K: Evaluation of herbal drugs an approach to the evaluation of botanicals. Horizons Pharmaceutical 2012; 519-21.
- Madani H, Talebolhosseini M, Asgary S and Nader GH: Hepatoprotective activity of Silybum marianum and Cichorium intybus against thioacetamide in rat. Pak J Nutrition 2008; 172-76.
- Rajalakshmi GK, Jothi A, Venkatesa RS, Venkatesa and Jegatheesan K: Hepatoprotective activity of Andrographis paniculata on paracetamol-induced liver damage in rats. Journal of Pharmacy Research 2012; 5(6): 2983-98.
- Chattopadhyay RR and Bandyopadhyay M: Possible mechanism of hepatoprotective activity of Azadirachta indica leaf extract against paracetamol-induced hepatic damage in rats. 2006; 123-30.
- Nasimilyas, Sadiq M and Jahangir A: Hepatoprotective effect of garlic (Allium sativum) and milk thistle (silymarin) in isoniazid-induced hepatotoxicity in rats. 2011; 213-18.
- Jeong HG, You HJ, Park SJ, Moon AR, Chung YC, Kang SK and Chun HK: Hepatoprotective effects of 18-glycyrrhetinic acid on carbon tetrachloride-induced liver injury inhibition of cytochrome P450 2E1. 2002; 46.
- Chattopadhyay RR and Bandyopadhyay M: Possible mechanism of hepatoprotective activity of Azadirachta indica leaf extract against paracetamol-induced hepatic damage in rats. 2006; 123-30.
- Aghela N, Rashidib I and Mombeinia A: Hepatoprotective activity of Capparis spinosa root bark against CCl4 induced hepatic damage in mice. Iran J Res 2007; 6(4): 285-90.
- Rawat AKS, Tripathi SK and Showme U: Hepato-protective activity of Boerhaavia diffusa roots: A popular Indian ethnomedicine J Ethnopharmacology 1997; 56: 61-66.
- Somchit MN, Sulaiman MR, Noratunlina R and Ahmad Z: Hepatoprotective effects of Curcuma longa rhizomes in paracetamol induced liver damage in rats. 2002; 698-702.
- Kumar Mu, Ahuja M and Sharma S: Hepatoprotective study of curcumin- soya lecithin complex 2008; 76: 761-74.
- Haldar KP, Gupta M, Mazumder KU, Kandar CC and Manikandan L: Hepatoprotective Effect of Wedelia calendulaceae against thioacetamide induced liver damage in Rats. 2007; 3: 414-21.
- Singaravel S, Srinivasan D, Jothivel N, Rsilingam D and Mani V: Hepatoprotective activity of Camellia sinensis and its possible mechanism of action. Pharma Thera 7; 9-14.
- Aghela N, Rashidib I and Mombeinia A: Hepatoprotective activity of Capparis spinosa root bark against CCl4 induced hepatic damage in mice. J Res 2007; 285-90.
- Vetrivel R, Shanmugavalli N, Greety S and Umashankar V: Hepatoprotective effects of Cassia tora on CCl4 induced liver damage in albino rats. J Sci Tech 2009; 2(3): 41-44.
- Sadeghi H, Nikbakht M, Ghaitasi I and Sabzali S: Hepatoprotective effect of Cichorium intybus on CCl4- induced liver damage in rats. J Biochem 2008; 2(6): 141- 44.
- Jeong HG, You HJ, Park SJ, Moon AR, Chung YC, Kang SK and Chun HK: Hepatoprotective effects of 18-glycyrrhetinic acid on carbon tetrachloride induced liver injury inhibition of cytochrome P450 2E1. 2002; 46.
- Shenoy KA, Somayaji SN and Bairy KL: Hepatoprotective effects of Ginkgo biloba against carbon tetra chloride induced hepatic injury in rats. J Pharmacol 2001; 33: 260-66.
- He SX, Luo JY, Wang YP, Wang YL, Fu H, Xu JL, Zhao G and Liu EQ.: Effects of extract from Ginkgo biloba on carbon tetrachloride-induced liver injury in rats. World J Gastroenterol 2006; 12(24): 3924-8.
- Lahon K and Das S: Hepatoprotective activity of Ocimum sanctum alcoholic leaf extract against paracetamol-induced liver damage in Albino rats. J Pharmacognosy 2011; 3(1): 13-18.
- Bhattacharjee R and Parames CS: Protein isolate from the herb Phyllanthus niruri plays a hepatoprotective role against carbon tetrachloride-induced liver damage via its antioxidant properties. J Food and Clinical Toxicology 2007; 817-26.
- Akhtar MS, Amin M and Ahmad and AM: Hepatoprotective effect of emodi roots against paracetamol-induced hepatotoxicity in rats. J Ethnobotanical Leaflets 2009; 13: 310-15.
- Singh A, Malhotra S and Subban R: Taraxacum officinale hepatoprotective herb with therapeutic potential. J Pharmacog 2008; 2(3): 163-67.
- Park JY, Park CM and Song YS: Hepatoprotective activity of dandelion Taraxacum officinale water extract against D- Galactosamine-induced hepatitis in rats 2008; 37(2): 177-83.
- Orhan DD, Orhan N, Ergun E and Ergun F. Hepatoprotective effect of Vitis vinifera leaves on carbon tetrachloride-induced acute liver damage in rats. J Ethnopharmacol 2007; (1): 145-51.
- Bishayi B, Roychowdhury S and Ghosh S: Hepato-protective activity and immunomodulatory properties of Tinosporia cordifolia in CCl4 in toxicatmature albino rats. J Toxicol Sci 2002; 27(3): 139-46.
- Murthy SR and Srinivasn M: Hepatoprotective effects of Tephrosia purpurea experimental animals. Journal Pharmaco 1993; 25-34.
- Elkoy TA, Mouneir SM, Kamel G, Alwabel NA and Zaher S: hepatoprotective effect of methanol extracts of Zingiber officinale and Cichorium intybus. Indian J Pharm Sci 2010; 72(5): 564-70.
- Singh B, Saxena AK, Chandan BK, Agarwal SG and Anand KK: In-vivo hepatoprotective activity of active fraction from ethanolic extract of Eclipta alba J Physiology and Pharmacology 2001; 45(4): 435-41.
- Hanefiozbek, Ugras S, Bayram I, Uygan I, Erdogan E, Ozturk A and Huyut Z: Hepatoprotective effect of Foeniculum vulgare essential oil, a carbon-tetrachloride induced liver fibrosis model in rats. J Lab Anim Sci 2004; 1: 31.
- Kaviarasan S, Viswanathan P and Anuradha CV: Fenugreek seed Trigonella foenumgraecum polyphenols inhibit ethanol-induced collagen and lipid accumulation in rat liver. 2007; 23(6): 373-83.
- Mohan GK, Pallvi E, Ravi Kumar B, Rajesh M and Venkatesh S: Hepatoprotective activity of Ficus carica leaf extract against carbon tetrachloride-induced hepatotoxicity in rats 2007; 162-66.
- Mohamed T, Saleem S, Christina AJM, Chidambaranthan N, Ravi V and Gauthaman K: Hepatoprotective activity of Annona squamosa J Applied Research in Natural Products 2008; 1(3): 1-7.
- Abuelgasim AI, Nuha HS and Mohammed AH: Hepatoprotective Effect of Lepidium sativum against carbon tetrachloride-induced damage in rats. J Animal and Veterinary Sciences 2008; 20-23.
- Meena B, Ezhilan AR, Rajesh R, Hussain SA, Ganesan B and Anandan R: Anti-hepatotoxic potential of Sargassum polycystum on antioxidant defense status in D-galactosamine induced hepatitis in rats. J Biochemistry Research 2008; 2(2): 051-055.
- Rosa MP, Gutiérrez and Rosario VS: Hepatoprotective and inhibition of oxidative stress of Prostecheamichuacana. 2009; 46-51.
- Pramyothin P, Ngamtin C, Poungshompoo S and Chaichantipyuth C: Hepatoprotective activity of Phyllanthus amarus extract in ethanol-treated rats. J Ethnopharmacol 142(2): 169-73.
- Rao KS and Mishra SH: Indian Journal of Pharmaceutical Science 1997; 59(4): 165-70.
- Madani H, Talebolhosseini M, Asgary S and Nader GH: Hepatoprotective activity of Silybum marianum and Cichorium intybus against thioacetamide in rat. J Nutrition 172-76.
- Arul Kumaran KS, Rajasekaran A, Ramasamy A, Jegadeesan M, Kavimani S and Somasundaram A: Cassia roxburghii seeds protect liver against toxic effects of ethanol and carbon tetrachloride in rats. J Pharm Tech Res 2009; 1(2): 273-46.
- Vadivu R, Krithika A, Biplab C, Dedeepya P, Shoeb N and Lakshmi KS: Evaluation of hepatoprotective activity of the fruits of Coccinia grandis J Health Res 2008; 1(3): 163-68.
- Sultana S, Perwaiz S, Iqbal M and Athar M: Crude extracts of hepatoprotective plants, Solanum nigrum and Cichorium intybus inhibit free radical-mediated DNA damage. J Ethnopharmacol 1995; 45: 189-92.
- Maheswari C, Maryammal R and Venkatanarayanan R: Hepatoprotective activity of ortho siphonstamineus on liver damage caused by paracetamol in rats. J Jordan Biological Sciences 2008; 1(3): 105-08.
- Kashaw V, Nemalkumar A and Agarwal A: Hepato-protective prospective of herbal drugs and their vesicular carriers. International Journal of Research in Pharmaceutical and Biomedical Sciences 2011; 10.
- Stickel F and Schuppan D: Herbal medicine in the treatment of liver diseases. 2007; 39: 293-04.
- Murray: Phytosomes- Increase the absorption of herbal extract. www.doctormurray.com articles/ silybin.htm.
How to cite this article:
Pandey S and Darunde D: Herbal drugs - An approach for the treatment of liver disorders. Int J Pharmacognosy 2019; 6(5): 164-71. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.6(5).164-71.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.