FORMULATION, DEVELOPMENT AND STANDARDIZATION OF AN AYURVEDIC MUCO-ADHESIVE EXTENDED RELEASE VAGINAL TABLET
HTML Full TextFORMULATION, DEVELOPMENT AND STANDARDIZATION OF AN AYURVEDIC MUCO-ADHESIVE EXTENDED RELEASE VAGINAL TABLET
Mradu Gupta * 1, Simit Kumar 2, Benoy Brata Bhowmik 3, Nandita Karmakar 1 and Saswati Sasmal 1
Department of Dravyaguna 1, Institute of Post Graduate Ayurvedic Education and Research, A. P. C. Road, Kolkata - 700009, West Bengal, India.
Department of Microbiology 2, R. G. Kar Medical College and Hospital, Kolkata - 700004, West Bengal, India.
Dr. B. C. Roy College of Pharmacy and Allied Health Sciences 3, Durgapur - 713206, West Bengal, India.
ABSTRACT: The study aims to standardize a vaginal herbal formulation consisting of stem barks of Ficus glomerata Roxb. and Symplocos racemosa Roxb. Since they are used as Ayurvedic medicine and described in ancient text Charak Samhita as astringent, anti-inflammatory and treatment of vaginitis/leucorrhoea. This new herbal formulation is likely to exhibit significant antimicrobial action due to the presence of phenolic and flavonoid compounds. The extractive value was 1.64 and 1.70% w/w for aqueous and alcoholic extract. Total Phenol Content was 225.67 and 220.00 µg Gallic acid equivalent/mg for aqueous and alcoholic extract. Fifteen types of mucoadhesive tablets weighing 550, 700 or 900 mg each were prepared using combinations of research drug, Carbopol and Hydroxy-Propyl-Methyl Cellulose as polymers, talc as a glidant, magnesium stearate as a lubricant and Micro-Crystalline Cellulose and Lactose monohydrate as diluents. Similarly, five types of effervescent mucoadhesive tablets using sodium bicarbonate and citric acid in 3:1 ratio were prepared. Apart from weight, thickness, hardness, and friability of each group, the surface pH, swelling index and muco-adhesive force were determined. The formulation F-XII provides best physical parameters of hardness (6.0 ± 2.53 kg/sq.cm), pH value (4.80 ± 0.04), swelling index (69.39 ± 0.44% w/w) and muco-adhesive force (0.667 ± 0.02 Newton) which indicate its sustained slow releasing of anti-vaginitis drug delivery as an effective mucoadhesive vaginal tablet.
| Keywords: |
Muco-adhesive, Effervescent, Herbal, Vaginal tablet, Ficus glomerata, Symplocos racemosa
INTRODUCTION: Vaginitis is an inflammation of the vagina that can result in discharge, itching, and pain.
The cause is usually a change in the normal balance of vaginal bacteria or an infection. Similarly, leucorrhoea refers to the medical condition where excessive abnormal thick and sticky white or yellowish discharge occurs from the vagina accompanied with inflammation and associated with symptoms like itching, burning sensation and pain followed by body ache and tiredness. The vagina is a fibro-vascular tube connecting the uterus to the outer surface of the body.
The vaginal epithelium consists of a stratified squamous epithelium and lamina propia. In the allopathic system of medicine, vaginal tablets have been prescribed for patients suffering from leucorrhoea indicated through symptoms of excessive white, sticky, foul-smelling vaginal discharge, known as Swetapradar in Ayurveda. Traditional commercial preparations, such as creams, foams, gels, suppositories, irrigations, and tablets are known to reside in the vaginal cavity for a relatively short period owing to the self-cleaning action of the vaginal tract, and often require multiple daily doses to ensure the desired therapeutic effect. Therefore, the vaginal route appears to be highly appropriate for bio-adhesive drug delivery systems to retain drugs for treating largely local conditions 1. These drugs are transported across the vaginal membrane by the trans-cellular route, intra-cellular route or vesicular and receptor-mediated transport mechanisms. Bio-adhesives can control the rate of drug release and extend the residence time of vaginal formulations.
In this regard, mucosal drug delivery has received a great deal of attention over the past few decades. Further, the mechanism of extended drug release ensures that the active drug is released slowly over time from the tablet which has the advantage of taking medicine less often. Therefore, nowadays muco-adhesive extended release vaginal tablets are very useful for the treatment of Leucorrhoea where the tablets adhere on the mucous membrane of the inner vaginal wall and the active drug is released slowly within around 8 - 10 hours in vaginal fluid and inhibit the discharge by their anti-microbial activity 2 - 4. This has the advantage that tablets need not be taken repeatedly or orally; they can be just inserted in the vagina at bed-time only.
Many herbal plants and their combinations like Ayurvedic drugs have been prescribed for oral administration and external application in the Ayurvedic text for the treatment of vaginitis or leucorrhoea. The aim of the present study was to standardize the new vaginal herbal formulation by mixing the equal parts of stem barks of Ficus glomerata Roxb. and Symplocos racemosa Roxb. because both these plants have been used since ancient times in the Ayurvedic system of medicine and elaborated in ancient texts such as Charak Samhita (Chikitsa Sthanam) as an astringent, anti-inflammatory and hemostatic and useful for arresting excessive abnormal vaginal discharge 5 - 7. This is a new herbal formulation which has not been evaluated till now although it is likely to exhibit sustained and significant antimicrobial action due to the synergetic effect of the phenolic and flavonoid compounds present in this research drug and the pharmacological properties of its constituent herbs.
This research formulation contains the plant Ficus glomerata Roxb. or Cluster Fig which belongs to the Moraceae family. It is a moderate sized spreading lactiferous tree without much prominent aerial roots found throughout India whose fruits are eaten by villagers. Its leaves are dark green, ovate or elliptical while the fruits contain 2 - 5 cm diameter sub-globose and smooth receptacles. The fruits are orange and dull reddish when ripe and having a pleasant smell. The stem bark is 0.5 -1.8 cm. thick, grayish-green in color and has an uneven soft surface 6, 7. On rubbing it, white papery flakes come out from the outer surface; the inner surface is light brown, fracture fibrous and mucilaginous taste.
The stem bark, fruits, leaves and latex of this plant have been used since ancient times as mentioned in the Ayurvedic text book for the treatment of dysentery, diarrhea, toothache, stomach-ache, vaginal disorders, menorrhagia, hemoptysis, diabetes, piles, and glandular swelling, etc. The roots of the plant are used in dysentery, pectoral complications, and diabetes, and also applied in inflammatory glandular enlargement, mumps, and hydrophobia. The latex is externally applied on wounds to decrease inflammation, pain, and edema and to promote healing.
The Phytochemical compounds isolated from the stem bark are leucocyanidin-3-o-B-glucopyranoside, leucopelargonidin 3-O-a- L-rhamnopyranoside, B- sitosterol, stigmasterol, tetracyclic triterpene- gluanol acetate and tiglic acid. The reported pharmacological properties of the different plant parts are hypoglycaemic, antiulcer, antioxidant, wound-healing, anti-inflammatory, anti-diarrhoeal, anti-bacterial, anti-fungal, antipyretic and anti-diuretic 8.
Symplocos racemosa Roxb. known as Lodhra belonging to the Symplocaceae family is found distributed throughout North Eastern India up to 2,500 ft. elevation. It is a small evergreen tree with stem up to 6 m in height and 15 cm in diameter. Its stem bark is useful in bowel complaints such as diarrhea and dysentery, in dropsy, eye disease, liver complaints, wound healing, excessive vaginal discharge, menstrual problems, fevers, ulcers, scorpion-string, etc. The bark is often employed in the preparation of plasters and is reported to promote maturation or resolution of boils, stagnant tumors and other malignant growths. A decoction of the bark or wood is used as a gargle for giving firmness to spongy and bleeding gums and relaxed uvula.
The phytochemical investigation of the n-butanol soluble fraction of the bark of stem of Symplocos racemosa Roxb. yielded two phenolic glycosides of salirepin series namely symplocuronic acid and sympocemoside while salirepin has also been isolated from this plant 9. The alcohol extract of stem bark indicated the presence of carbohydrates, glycosides, saponins, terpenoids, and alkaloids while its ether extract indicated the presence of glycosides, phytosterol, and steroids. The prominent pharmacological activities of its stem bark are antibacterial, anti-inflammatory, antiulcer, anti-tumor, antimicrobial and antioxidant 10.
MATERIALS AND METHODS:
Plant Materials: The stem barks of Symplocos racemosa Roxb. and Ficus glomerata Roxb. were purchased from crude drug supplier of Katwa Chowrasta, Burdwan district for the preparation of herbal vaginal tablet and the plant samples were authenticated by the Research Officer, Botanical Survey of India, Howrah, India (Ref. No. BSI/CNH/SF/Tech./2016).
Chemicals used for Preparation of Tablets: Di-calcium phosphate, Gum acacia, Lactose monohydrate, Sodium carboxy-methyl-cellulose, Sodium starch glycolate, Starch (maize), Ferric Chloride (FeCl3), Magnesium stearate (IP grade), Microcrystalline cellulose (IP grade), Talc (IP grade), Folin - Ciocalteu’s reagent, Sodium carbonate and Sulphuric acid were obtained from M/s Merck Specialties Pvt. Ltd., Mumbai. Carbopol 934P and Hydroxy-propyl-methyl-cellulose K4M were purchased from reputed company M/s Hi-Media Laboratories Pvt. Ltd., while Citric acid monohydrate was procured from M/s B.D. Pharmaceutical works Pvt. Ltd. and Sodium bi-carbonate (IP grade) from M/s Indian Drug House.
Preparation of Extracts: The stem barks of Symplocos racemosa Roxb. and Ficus glomerata Roxb. were taken in equal quantity by weight, washed, sun-dried and crushed to a particle size of 40 mesh. This coarse powder was sequentially extracted with petroleum ether (60 °C - 80 °C), chloroform, acetone, ethanol, and water using soxhlet apparatus. These extracts were filtered using a Buckner funnel and Whatman No. 1 filter paper at room temperature and concentrated at reduced temperature and pressure using rotary evaporator. All obtained extracts were stored in a refrigerator below 10 °C for subsequent experiments. The aqueous extract of the research formulation was used in study 11, 12.
Analysis of Phytochemical Constituents: The extract was standardized by using the standard methods for ascertaining the presence of different phytochemical constituents such as alkaloids, amino-acids, reducing sugars, tannins, saponins, anthraquinones, steroids, terpenoids, flavonoids, and salicylates.
Determination of Total Phenol Content: The total phenol content was determined using the Folin- Ciocalteu reagent. To 0.5 ml aliquot of dried aqueous extract, 2.5 ml of Folin- Ciocalteu’s reagent (10%) and 2 ml of 7.5% sodium carbonate were added. The absorbance was read after 30 min incubation period at room temperature at 760 nm colorimetrically, and calculation was done based on standard calibration plot using known different concentrations of Gallic acid and Total phenolic content was expressed as mg Gallic Acid Equivalents (GAE) 13, 14.
Formulation of Drug-Free Tablets (i.e., Placebo): The drug-free (placebo) tablets were prepared using a mixture of polymers and microcrystalline cellulose with or without effervescent. Carbopol and Hydroxy-Propyl-Methyl Cellulose (HPMC) were used as polymers. Tablets loaded with effervescent agent consisted of sodium bicarbonate and citric acid in 3:1 molecular ratio and produced gas bubbles in liquid media. All used chemicals were of analytical reagent grade. The composition of the polymers and effervescent agents used in various placebo tablets is shown in Table 1. For the various drug-free formulations, the effect of the effervescent agent on polymers’ bio-adhesive characteristics was investigated. The performances of these bio-adhesive polymers were evaluated, especially three main parameters pH value, swelling index, and the bio-adhesive strength. Based on these data, suitable polymers were selected to prepare the bio-adhesive effervescent vaginal tablets of active research drug.
TABLE 1: COMPOSITIONS OF POLYMERS USED IN PLACEBO TABLETS
| S. no. | Polymer used | Polymer weight (mg) | Effervescent Mixture (mg) |
| S1a | Carbopol 934P | 150 | 0 |
| S1b | Carbopol 934P | 150 | 100 |
| S2a | HPMC K4M | 150 | 0 |
| S2b | HPMC K4M | 150 | 100 |
| S3a | Sodium CMC | 150 | 0 |
| S3b | Sodium CMC | 150 | 100 |
| S4a | Carbopol + Sod. CMC | 75+75 | 0 |
| S4b | Carbopol + Sod. CMC | 75+75 | 100 |
| S5a | Sod.CMC + HPMC K4M | 75+75 | 0 |
| S5b | Sod.CMC + HPMC K4M | 75+75 | 100 |
Preparation of Muco-adhesive Vaginal Tablets: The various types of bio-adhesive vaginal herbal tablets were prepared using the dry compression technique of tablet preparation. During this study, 15 types of muco-adhesive herbal vaginal formulations and 5 types of muco-adhesive effervescent vaginal formulations were prepared for testing. All these formulations were prepared by mixing the same amount of the active research drug extract powder (500 mg) with different amounts of excipients, binders and developers. The polymers Carbopol and Hydroxy-Propyl-Methyl Cellulose were used as excipients, while talc and magnesium stearate were added as glidant and lubricant respectively. Micro-Crystalline Cellulose and Lactose monohydrate were used as diluents 15. The binder hydroxyl-propyl-methyl-cellulose was used to form a sustained-release matrix with the polymer carbopol, which swells to form hydrogel-like matrices through which drug molecules could be released at a controlled rate 16. Then all ingredients were passed through a 44# mesh sieve and finally, the mixture was compressed into tablet-form using single punch tablet compression machine. The ingredients in each type of mucoadhesive tablet are shown in Table 2.
TABLE 2: INGREDIENTS OF VARIOUS MUCO-ADHESIVE TABLETS
| S. no. | Ingredients
(mg) |
F-
I |
F-
II |
F-III | F-IV | F-
V |
F-VI | F-VII | F-VIII | F-IX | F-
X |
F-XI | F-XII | F-XIII | F-XIV | F-XV |
| 1 | Active Research Drug (mg) | 500 | 500 | 500 | 500 | 500 | 500 | 500 | 500 | 500 | 500 | 500 | 500 | 500 | 500 | 500 |
| 2 | Carbopol 934P | 25 | 12.5 | 10 | - | - | - | - | 25 | 20 | 35 | 35 | 15 | 45 | 25 | 15 |
| 3 | Sodium Starch Glycolate | 25 | 37.5 | 40 | 3 | 3 | 8 | - | - | - | - | - | - | - | - | - |
| 4 | HPMC K4M | - | - | - | 10 | 25 | 35 | 95 | 75 | 50 | 95 | 150 | 90 | 90 | 110 | 110 |
| 5 | Starch (maize) | - | - | - | 80 | 65 | 55 | - | - | - | - | - | - | - | - | - |
| 6 | Gum Acacia | - | - | - | - | - | 10 | - | - | - | - | - | - | - | - | - |
| 7 | Lactose monohydrate | - | - | - | 100 | 100 | 85 | 33 | 65 | 65 | 132 | 100 | 100 | 100 | 100 | 100 |
| 8 | Di-Calcium Phosphate | - | - | - | - | - | - | 65 | 28 | 58 | 128 | - | - | - | - | - |
| 9 | Microcrystalline cellulose | - | - | - | - | - | - | - | - | - | - | 95 | 175 | 145 | 145 | 155 |
| 10 | Talc | - | - | - | 7 | 7 | 7 | 7 | 7 | 7 | 10 | 10 | 10 | 10 | 10 | 10 |
| 11 | Magnesium Stearate | - | - | - | - | - | - | - | - | - | - | 10 | 10 | 10 | 10 | 10 |
| Total ingredients (mg) | 550 | 550 | 550 | 700 | 700 | 700 | 700 | 700 | 700 | 900 | 900 | 900 | 900 | 900 | 900 | |
Formulation of Effervescent Muco-adhesive Vaginal Tablets: The effervescent mucoadhesive vaginal tablets were also prepared by direct mixing of required quantity of 500 mg active research drug, effervescent (consisting of sodium bicarbonate and citric acid in the ratio of 3:1), polymers (i.e., carbopol, hydroxy-propyl-methyl cellulose and sodium carboxy-methylcellulose), microcrystalline cellulose, magnesium stearate, and talc. Talc and magnesium stearate were added as glidant and lubricant respectively in the ratio of 2:1. Finally, the mixture was compressed into tablets using a single punch tablet compression machine. Each tablet of different effervescent formulation, i.e., T-1 to T-5, contained 500 mg of active research drug and has an approximate weight of 900 mg as detailed in Table 3.
TABLE 3: COMPOSITION OF VARIOUS MUCO-ADHESIVE EFFERVESCENT VAGINAL TABLETS
| S. no. | Ingredients | T-1 | T-2 | T-3 | T-4 | T-5 |
| 1 | Active Research Drug | 500 | 500 | 500 | 500 | 500 |
| 2 | Carbopol 934P | 75 | – | 75 | 60 | – |
| 3 | HPMC K4 M | 75 | 75 | – | 60 | 60 |
| 4 | Sodium CMC | – | 75 | 75 | – | 60 |
| 5 | Sodium bi-carbonate | 75 | 75 | 75 | 97.5 | 97.5 |
| 6 | Citric acid | 25 | 25 | 25 | 32.5 | 32.5 |
| 7 | Microcrystalline cellulose | 127.5 | 127.5 | 127.5 | 127.5 | 127.5 |
| 8 | Talc | 15 | 15 | 15 | 15 | 15 |
| 9 | Magnesium Stearate | 7.5 | 7.5 | 7.5 | 7.5 | 7.5 |
| Total (mg) | 900 | 900 | 900 | 900 | 900 |
Evaluation of Muco-adhesive Vaginal Tablets: All types of mucoadhesive vaginal tablets were tested and evaluated in respect of the following standard testing parameters:
Weight Variation: Ten tablets from each formulation were weighed using an electronic balance, and the average weight was calculated. Not more than two of the individual weights deviate from the average weight by more than the percentage shown in Table 4, and none deviates by more than twice that percentage.
Thickness: The thickness of three randomly selected tablets from each formulation was determined in mm using a vernier caliper (Pico India). The average values were calculated.
Hardness of the Tablet: The tablet hardness, the force required to break a tablet in a diametric compression force, was estimated using a hardness tester (Pfizer type) to determine the need for pressure adjustments on the tablet machine. Hardness can affect the disintegration properties. So if the tablet is too hard, it may not disintegrate in the required period. The hardness of the tablets was determined using Monsanto hardness tester and expressed in kg/sq.cm. Three tablets were randomly picked from each formulation, and the mean and standard deviation values were calculated.
Friability: Friability is a measure of the resistance of the tablets to abrasion. It is an important parameter which tests the breaking point and structural integrity of a tablet under conditions of storage, transportation, and handling before usage. The breaking point of a tablet is primarily based on its shape. The friability of the tablets was measured using Friability Test Apparatus (Imcorp, India) by tumbling them in a rotating drum. After tumbling, the integrity of the tablets and the weight loss are evaluated. Ten tablets are weighed initially, rotated at 25 rpm for 4 min (total 100 rotations) and re-weighed after removal of fines. Friability below 1.0 % w/w was considered acceptable 17.
% Weight loss = Initial weight – Final weight × 100 / Initial weight
Determination of the Surface pH Value: Surface pH of the tablets was determined to investigate the possibility of any in-vivo side effects. Since an alkaline pH may irritate the vaginal mucosa, it was determined to keep the surface pH as close to the system pH (4.0 – 5.0) as possible. A combined glass electrode was used for this purpose. The tablet was allowed to swell by keeping it in contact with 5 ml of distilled water (pH 6.5 ± 0.05) for 2 h at room temperature. The pH was measured by bringing the electrode in contact with the surface of the tablets and allowing it to equilibrate for 1 min.
Swelling Study: The swelling behavior of tablet can is described in terms of their water absorbing capacity with time. Tablets were initially weighed individually (W0), placed separately in 2% agar gel plates and incubated at (37 ± 1) °C. At regular 0.5 hour time intervals until 4 hours, each tablet was removed from the Petri-dish, and excess surface water was removed carefully using filter paper. The swollen tablet was then reweighed (Wt), and the percentage of swelling was calculated using the following formula:
% Swelling = {(Wt – W0)/ W0} × 100
Where Wt is the weight of the tablet at time t and W0 is the initial weight of the tablet. The swelling was calculated and plotted as a function of time. The slope of the linear plots was taken as the swelling rate 18.
In-vitro Muco-adhesion Study: Several types of the mucosa, including rat intestine, pig oral, bovine sublingual and goat vaginal mucosa have been used as model biological tissues for evaluation of bio-adhesion. A simple apparatus was devised to measure the minimum detachment force. In the evaluation of adhesion, it is important to use uniform surfaces that allow the formation of reproducible adhesive bonds. In the present study, goat vaginal mucosa was used as a model mucosal surface for bio-adhesion testing. For this purpose, the mucosa was removed immediately after slaughter and kept in phosphate buffer.
At the time of testing, a section of mucosa was secured keeping the mucosal side out on the upper glass vial using a rubber band and aluminum cap. The diameter of each exposed mucosal membrane was 1 cm. Then one vial with a section of mucosa and another vial were fixed on height adjustable pan. To a lower vial, a tablet was placed with the help of bi-layered adhesive tape, adhesive side facing downward. The height of the lower vial was adjusted so that a tablet could adhere to mucosa on the upper vial. A constant force was applied on the upper vial for 2 min, after which it was removed and the upper vial was then connected to the balance. Then the weight on right side pan was slowly added in increments of 0.5 gm till the two vials just separated from each other. The total weight required to detach the two vials was taken as a measure of the mucoadhesive strength 19, 20. From this mucoadhesive strength, the force of adhesion was calculated using the following formula:
Force of adhesion (Newton) = Muco-adhesive Strength (gm) × 9.81/1000
RESULTS: Evaluation of the physical parameters of powder of the herbal vaginal formulation indicated that while the moisture content was 8.2% w/w, the total ash content was 12.30% w/w, the insoluble acid ash was 0.77% w/w and water soluble ash was 10.66% w/w. The extractive value of aqueous extract was 1.64% w/w, and the extractive value of alcoholic extract was found to be 1.70% w/w. The phytochemical constituents like alkaloids, flavonoids, tannins, and carbohydrates were found to be present in the aqueous and alcohol extracts of the Research Formulation.
The total phenol content (µg Gallic acid equivalent/mg of extract) was calculated from the absorbance calibration standard curve (R2 = 0.997) generated with different concentrations of Gallic acid. The TPC was found to be 225.67 µg Gallic acid equivalent/mg of extract in the aqueous extract and 220.00 µg Gallic acid equivalent/mg of extract in the alcoholic extract of the research formulation which could be directly responsible for its antimicrobial, anti-leucorrhoea and astringent properties.
The present study examined the efficacy of various combinations of drug, excipients, polymers, etc. to develop a standardized vaginal tablet for curing bacterial, fungal and others vaginal infections and decreased the associated symptoms like excessive white discharge, itching, foul smell, etc.
One critical parameter during these experiments was to maintain a pH of around 4.5 to 5.0 in the vaginal mucosa during prolonged periods of intervention by the bio-adhesive tablets to inhibit the growth of pathologic organisms there.
The obtained results of the various parameters in respect of the 15 types of mucoadhesive and 5 types of effervescent mucoadhesive vaginal tablets have been presented in the Tables 4 and 5 and Fig. 1, 2, and 3.
TABLE 4: PHYSICAL PARAMETERS OF MUCO-ADHESIVE VAGINAL TABLET FORMULATIONS
| Formulation | Average Weight (mg) | Thickness (mm) | Hardness (kg/sq.cm) | Friability (% w/w) |
| F-I | 562.1 ± 18.84 | 3.44 ± 0.02 | 2.0 ± 5.13 | 0.92 |
| F-II | 577.0 ± 10.39 | 3.60 ± 0.05 | 2.4 ± 11.34 | 0.90 |
| F-III | 563.2 ± 13.95 | 3.42 ± 0.02 | 1.8 ± 7.51 | 1.04 |
| F-IV | 756.1 ± 15.63 | 4.40 ± 0.03 | 2.2 ± 6.21 | 0.92 |
| F-V | 726.8 ± 22.27 | 4.06 ±0.03 | 3.2 ± 4.46 | 0.85 |
| F-VI | 714.6 ± 19.36 | 4.22 ± 0.03 | 4.0 ± 8.74 | 0.87 |
| F-VII | 746.6 ± 26.39 | 4.38 ± 0.02 | 2.0 ± 7.28 | 0.97 |
| F-VIII | 735.8 ± 15.94 | 4.22 ± 0.01 | 3.4 ± 2.68 | 0.81 |
| F-IX | 706.0 ± 16.33 | 4.42 ± 0.02 | 3.8 ± 7.54 | 0.78 |
| F-X | 937.3 ± 18.95 | 6.00 ± 0.03 | 2.8 ± 11.31 | 0.89 |
| F-XI | 928.0 ± 42.20 | 5.82 ± 0.03 | 8.0 ± 4.52 | 0.42 |
| F-XII | 927.8 ± 25.30 | 5.80 ± 0.04 | 6.0 ± 2.53 | 0.66 |
| F-XIII | 932.2 ± 35.60 | 5.92 ± 0.05 | 5.6 ± 4.04 | 0.68 |
| F-XIV | 939.0 ± 28.21 | 5.66 ± 0.05 | 8.8 ± 6.69 | 0.38 |
| F-XV | 932.0 ± 25. 82 | 5.78 ± 0.03 | 7.2 ± 8.14 | 0.35 |
| ET-1 | 1000.0 ± 32.16 | 6.20 ± 0.03 | 6.4 ± 7.52 | 0.31 |
| ET-2 | 1026.4 ± 36.15 | 6.40 ± 0.03 | 1.8 ± 5.39 | 1.08 |
| ET-3 | 1005.7 ± 27.68 | 6.36 ± 0.06 | 3.0 ± 14.97 | 0.98 |
| ET-4 | 983.3 ± 39.64 | 6.18 ± 0.04 | 6.2 ± 8.37 | 0.69 |
| ET-5 | 979.6 ± 21.71 | 6.12 ± 0.04 | 2.6 ± 9.83 | 0.89 |
Number of tablets taken for each parameter (n = 10).
TABLE 5: OTHER PHYSICAL PARAMETERS OF MUCOADHESIVE VAGINAL TABLET FORMULATIONS
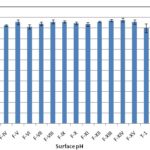
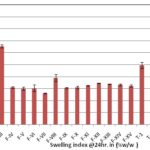
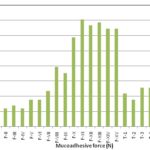
| Formulation | Surface pH | Swelling Index at 24hour (%w/w) | Muco-adhesive force (Newton) |
| F-I | 3.54 ± 0.11 | 23.92 ± 0.31 | 0.196 ± 0.04 |
| F-II | 3.50 ± 0.09 | 26.60 ± 1.95 | 0.118 ± 0.03 |
| F-III | 3.40 ±0.11 | 131.00 ± 2.63 | 0.137 ± 0.03 |
| F-IV | 4.62 ± 0.04 | 62.01 ± 1.68 | 0.118 ± 0.03 |
| F-V | 4.78 ± 0.11 | 60.21 ± 2.84 | 0.177 ± 0.02 |
| F-VI | 4.56 ± 0.10 | 60.99 ± 5.37 | 0.177 ± 0.05 |
| F-VII | 4.70 ± 0.08 | 52.49 ± 0.99 | 0.235 ± 0.03 |
| F-VIII | 4.80 ± 0.09 | 78.00 ± 5.78 | 0.392 ± 0.03 |
| F-IX | 4.80 ± 0.06 | 61.03 ± 1.32 | 0.353 ± 0.05 |
| F-X | 4.72 ± 0.06 | 62.19 ± 2.24 | 0.589 ± 0.03 |
| F-XI | 4.68 ±0.09 | 65.26 ± 0.62 | 0.706 ± 0.02 |
| F-XII | 4.80 ± 0.04 | 69.39 ± 0.44 | 0.667 ± 0.02 |
| F-XIII | 4.86 ± 0.05 | 67.89 ± 0.51 | 0.687 ± 0.03 |
| F-XIV | 4.88 ± 0.09 | 66.50 ± 1.69 | 0.647 ± 0.05 |
| F-XV | 4.79 ± 0.11 | 64.71 ± 2.14 | 0.647 ± 0.07 |
| ET-1 | 4.50 ± 0.21 | 99.20 ± 5. 16 | 0.216 ± 0.04 |
| ET-2 | 4.42 ± 0.18 | 90.56 ± 1.82 | 0.177 ± 0.04 |
| ET-3 | 4.48 ± 0.24 | 78.39 ± 3.61 | 0.255 ± 0.03 |
| ET-4 | 4.38 ± 0.22 | 98.94 ± 3.24 | 0.255 ± 0.03 |
| ET-5 | 4.40 ± 0.21 | 190.10 ± 7.81 | 0.157 ± 0.02 |
Number of tablets taken for each parameter (n = 3).
DISCUSSION: The vaginal microbiota is dominated by Gram-positive Lactobacillus bacteria, which maintain the acidic pH in the vagina (pH 4.5) by converting glycogen from exfoliated epithelial cells into lactic acid and protect it from pathogen invasion by the production of organic acid, bacteriocins, and hydrogen peroxide. The ecology of the vagina is influenced by factors such as the glycogen content of epithelial cells, glucose, pH, hormonal levels, trauma during sexual intercourse, birth-control method, age, antimicrobial treatment, and delivery. The acidic environment of the vagina is a natural barrier to infection and irritation since it represses the growth of bad bacteria which prefer a less acidic environment. Therefore, as long as the good bacteria count is high and the vaginal pH is acidic, bad bacteria have a very slim chance of overgrowing, making the chance of infection low. However, the pH changes with age, stages of the menstrual cycle, infections and sexual arousal while menstrual, cervical and uterine secretions and semen act as alkalizing agents and increase the pH 21.
FIG. 1: SURFACE pH OF MUCOADHESIVE AND EFFERVESCENT VAGINAL TABLETS
FIG. 2: SWELLING INDEX OF MUCOADHESIVE AND EFFERVESCENT VAGINAL TABLETS
FIG. 3: BIOADHESIVE FORCE OF MUCOADHESIVE AND EFFERVESCENT VAGINAL TABLETS
The drug-free tablets were prepared using the mixture of polymer and microcrystalline cellulose (MCC) with or without effervescent. Carbopol 934P (CP) is a mucoadhesive polymer which has been investigated as a useful adjuvant for bio-adhesive drug delivery system. However, the drug release rate from the solid formulation of CP is slow. At the same time, swelling is also important for the assessment of adhesion. Shortly after swelling, adhesion does occur, but with a weak bond formed. To develop maximum adhesion strength, an optimum water concentration was needed for polymer particles. The porous channels increased the area of contacting between polymer particles and water so that the polymers could be hydrated more easily.
It has been observed that the first three mucoadhesive vaginal formulations, i.e., F-I, F- II and F-III, were prepared by mixing of a single polymer (Carbopol) in various ratios as a disintegrating agent, and Sodium Starch Glycolate for developing mucoadhesive action with the active drug ingredient. The best outcomes were observed in case of F-III having average weight 563.2 ± 13.95 mg, thickness 3.42 ± 0.02 mm, hardness 1.8 ± 7.51 kg/sq.cm, friability 1.04% w/w, surface pH 3.40 ± 0.11, swelling Index at 24 h 131.00 ± 2.63% w/w and mucoadhesive force of 0.137 ± 0.03 Newton. After three trials of each of these 3 formulations, they were not considered feasible due to quick disintegration, less pH value, more swelling index, and less mucoadhesive tension.
The next set of formulations, F-IV to F-VI, were prepared as standard mucoadhesive sustained release herbal vaginal drug delivery system in the form of tablets by mixing of the same concentration of active ingredient of research drug (500 mg) with various ratios of the polymers such as Hydroxy-Propyl-Methyl Cellulose (HPMC K4 M) to get sustained release property along with a binder (maize starch), sodium starch Glycolate (SSG), diluent (Lactose monohydrate) and glidant (Talc) to increase total weight of tablets up to 700 mg. Only in the case of F-VI, Gum acacia was added to increase its binding properties.
In this group of formulations, the best parameters were exhibited by formulation F-VI having 714.6 ± 19.36 mg average weight, 4.22 ± 0.03 mm thickness, 4.0 ± 8.74 kg/sq.cm hardness, 0.87% w/w friability, 4.56 ± 0.10 surface pH, 60.99 ± 5.37% w/w swelling Index at 24 h and 0.177 ± 0.05 Newton mucoadhesive force. However, the formulation of F-VI has not been considered feasible due to its low hardness and mucoadhesive tension.
Another set of formulations, F-VII, F-VIII and F-IX, were developed by adding various concentrations of the polymers specially Carbopol 934P, Hydroxy-Propyl-Methyl Cellulose (HPMC K4 M), diluent Di-Calcium Phosphate (DCP), Lactose monohydrate and talc to the research drug. The best physical parameters in this set were obtained in case of formulation F-IX having average wt 706.0 ± 16.33 mg, thickness 4.42 ± 0.02 mm, hardness 3.8 ± 7.54 kg/sq.cm, friability 0.78% w/w, surface pH 4.80 ± 0.06, swelling index at 24 h 61.03 ± 1.32% w/w and mucoadhesive force 0.353 ± 0.05 Newton. However, the formulation F-IX was not found very feasible due to high friability and low mucoadhesive strength.
The other set of drug formulations, F-X to F-XV were developed keeping the weight of the vaginal tablets around 900 mg by mixing 500 mg of the active ingredient drug with different proportions of polymers like Carbopol 934P, HPMC K4M, Di-Calcium Phosphate, Lactose monohydrate, talc, magnesium stearate, etc. After many trials and assessments, the best physical parameters are obtained in case of formulation F-XII which exhibits average weight 927.8 ± 25.30 mg, thickness 5.80 ± 0.04 mm, hardness 6.0 ± 2.53 kg/sq.cm, friability 0.66% w/w, surface pH 4.80 ± 0.04, swelling index at 24 h 69.39 ± 0.44% w/w and muco-adhesive force of 0.667 ± 0.02 Newton.
The effervescent mucoadhesive vaginal tablets, ET-I to ET-V, were prepared by adding specific effervescent polymers like sodium bicarbonate and citric acid, along with Talc and magnesium stearate with the active research drug. The swelling rate was noticed to be increasing with an increasing amount of effervescent being applied to the formulation; however, the hardness and muco-adhesive tension were found to have decreased. The phenomenon of increasing swelling could be explained by the good disintegration effect of effervescent, which made tablets increase in volume and weight and led to the construction of porous channels on the surface and inside of tablets 22. The porous channels increased the area of contact between the polymer particles and water so that the polymers could be hydrated more easily. Among effervescent mucoadhesive vaginal tablets, the best results were noticed in ET-V formulation, which is average wt 979.6 ± 21.71 mg, thickness 6.12 ± 0.04 mm, hardness 2.6 ± 9.83 kg/sq.cm, friability 0.89% w/w, surface pH 4.40 ± 0.21, swelling index at 24 h 190.10 ± 7.81% w/w and mucoadhesive force 0.157 ± 0.02 Newton.
To achieve the desired therapeutic effect, a good vaginal delivery system for curing vaginitis needs to reside at the site of infection for a prolonged period. Hence, there is a need to develop an effective drug delivery system that should prolong the contact of the drug with the vaginal mucosal surface. Thus vaginal tablets appear to be useful dosage forms as they are easy to apply, portable and provide effective local absorption. During this study, the formulation F-XII provides the best physical parameters such as hardness, pH value, swelling index and bio-adhesive tension which are essential for maintaining the vaginal flora. The results indicate that this formulation F-XII will provide sustained slow releasing of anti-vaginitis and anti-leucorrheal drug delivery system in the form of an effective mucoadhesive vaginal tablet.
CONCLUSION: The study suggests that the various relevant physical parameters of a vaginal herbal formulation which directly impact its efficacy and therapeutic action in respect of vaginitis can be suitably controlled and modified by varying the composition of its constituents such as active drugs, polymers, mucoadhesive agents, diluents, glidants, etc. The herbal formulation comprising of the aqueous extract of stem barks of Symplocos racemosa Roxb. and Ficus glomerata Roxb. exhibits the best therapeutic efficacy in the form of non-effervescent mucoadhesive tablet F –XII as indicated by its hardness, friability, pH, swelling index and mucoadhesive force.
ACKNOWLEDGEMENT: This research work was supported by grants from the Indian Council of Medical Research, Government of India (Sanction Letter No. 59/1/2011/BMS/TRM dated 12.12.2013) and the authors are grateful for this support. The authors would like to acknowledge the contribution of Mr. Santanu Mohapatra, Scientist, Pharma Development division, RPG Life Sciences Limited, Navi Mumbai, India, for his help in mucoadhesive vaginal tablet formulation & development.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Vermani K and Garg S: The scope and potential of vaginal drug delivery. Pharmaceutical Science and Technology Today 2000; 3(10): 359-364.
- Jimenez-Castellanos MR, Zia H and Rhodes CT: Mucoadhesive drug delivery systems. Drug Dev Ind Pharm 1993; 19: 143-94.
- Duchene D, Touchard F and Peppas NA: Pharmaceutical and medical aspects of bioadhesive systems for drug administration. Drug Dev Ind Pharm 1988; 14: 283-18.
- Peppas NA and Buri PA: Surface, interfacial and molecular aspects of polymer bioadhesion on soft tissues. J Control Release 1985; 2: 257-75.
- Shastri SN: Charak Samhita (Chikitsa Sthanam), Chapter 3. Chaukhamba Bharati Academy, Varanasi, India 1988; 47.
- Sharma PV: Introduction to Dravyaguna (Indian Pharmacology), Chaukhamba Orientalia, Varanasi, India, 1995; 2: 616, 666.
- Sharma PC, Yelne MB and Dennis TJ: Database on medicinal plants used in Ayurveda, Central Council for Research in Ayurveda and Siddha, Department of Indian system of medicine, Govt. of India, New Delhi 2001; 3: 76-87, 166.
- Joshi U and Mohini U: Evaluation of Antioxidant Activity of Aqueous Extract Bark of Ficus glomerata. Research Journal of Pharmacy and Technology 2008; 1(4): 537-538.
- Ahmad VU, Abbasi MA, Hussain H, Akhtar MN, Farooq U, Fatima N and Choudhary MI: Phenolic glycosides from Symplocos racemosa: natural inhibitors of phosphor-diesterase I. Phytochemistry 2003; 63(2): 217-220.
- Devmurari VP: Antibacterial Evaluation and Phytochemical Screening of Symplocos racemosa Roxb, International Journal of Pharm. Tech Research 2010; 2(2): 1359-63.
- Furniss BS, Hannaford AJ, Smith PWG and Tatchell AR: Vogel’s Textbook of Practical Organic Chemistry, 5th Addison Wesley Longman Inc. 1989; 1205.
- Harborne JB: Phytochemical Methods: A guide to Modern Technique of Plant Analysis, 2nd Chapmann and Hall: London 1984.
- Baba SA and Malik SA: Determination of total phenolic and flavonoid content, antimicrobial and antioxidant activity of a root extract of Arisaema jacquemontii. Journal of Taibah University for Science 2015; 9: 449-454.
- Shukla S, Mehta A, Bajpai KV and Shukla S: In vitro antioxidant activity and total phenolic content of ethanolic leaf extract of Stevia rebaudiana Food Chem. Toxicol 2009; 47: 2338–2343.
- Rahamatullah S, Thakur RRS, Martin JG, Woolfson AD and Donnelly RF: Mucoadhesive drug delivery systems. Journal of Pharmacy and Bio Allied Sciences 2011; 3(1): 89-100.
- Patel A, Patel K and Patel J: Development and evaluation of mucoadhesive vaginal tablet of sertaconazole for Vaginal candidiasis. Int J of Pharm Tech Research 2011; 3(4): 2175-2182.
- Bhanja S, Ellaiah P, Martha SK, Sahu PK, Tiwari SP, Panigrahi BB and Das D: Formulation and in-vitro evaluation of mucoadhesive buccal tablets of Timolol maleate. Int J Pharm Biomed 2010; 1(4): 129-134.
- Karasulu HY, Hilmioglu S, Metin DY and Guneri T: Efficacy of a new sertaconazole bioadhesive vaginal tablet on Candida albicans. IL Farmaco 2004; 59: 163-167.
- Gursoy A, Sohtorik I, Uyanik N and Peppas NA: Bioadhesive controlled release systems for vaginal delivery, STP Pharma 1989; 5: 886-892.
- Bagul U, Gujar K, Dhat S, Aphale S and Bhavsar M: In-vitro study of mucoadhesive strength of polymers for mucoadhesive drug delivery systems. Int J of Current Ph’ceutical Research 2009; 1(1): 42-46.
- Vermani K and Garg S: The scope and potential of vaginal drug delivery. Pharmaceutical Science and Technology Today 2000; 3(10): 359-364.
- Chime SA, Onunkwo GC and Onyishi II: Kinetics and mechanisms of drug release from swellable and non-swellable matrices: a review. Research J of Ph’ceutical, Biological and Chemical Sciences 2013; 4(2): 97-103.
How to cite this article:
Gupta M, Kumar S, Bhowmik BB, Karmakar N and Sasmal S: Formulation, development and standardization of an ayurvedic muco-adhesive extended release vaginal tablet. Int J Pharmacognosy 2018; 5(1): 15-25. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.5(1).15-25.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
3
15-25
641
968
English
IJP
M. Gupta *, S. Kumar, B. B. Bhowmik, N. Karmakar and S. Sasmal
Department of Dravyaguna, Institute of Post Graduate Ayurvedic Education and Research, Kolkata, West Bengal, India.
mradu_gupta@hotmail.com
11 May 2017
21 August 2017
17 September 2017
10.13040/IJPSR.0975-8232.IJP.5(1).15-25
01 January 2018