FORMULATION AND EVALUATION OF GEL CONTAINING MANGIFERA INDICA LEAVES EXTRACT FOR ANTI-BACTERIAL ACTIVITY
HTML Full TextFORMULATION AND EVALUATION OF GEL CONTAINING MANGIFERA INDICA LEAVES EXTRACT FOR ANTI-BACTERIAL ACTIVITY
P. Sailaja Rao *, Bala Tripura Sundari and Sireesha Kalva
Sri Venkateshwara College of Pharmacy, Madhapur, Hyderabad - 500081, Telangana, India.
ABSTRACT: In the present study, an attempt was made to formulate ethosomal gel containing leaf extract of Mangifera indica, and was evaluated for antibacterial activity against gram-positive and gram-negative organisms. Different formulations of ethosomes using lecithin, cholesterol, and ethanol were prepared using Mangifera indica herbal leaf extracts. Carbopol 940 was used to prepare ethosomal gel. The entrapment efficiency of ethosomes was 65.1% - 96.54% and the average vesicle size was 920 nm. Three formulations of different doses (100, 200 and 300 mg) were selected based on entrapment efficiency and drug release. Prepared gels were then evaluated for physicochemical characteristics and drug content. The antibacterial activity was done using agar well diffusion method for the zone of inhibition. The pH of the gel formulations was found to be in the range of 5.4 - 6.2 and viscosities between 2250 - 2399 centipoises. The drug content of gels ranged between 74.67 - 82.31%. The zone of inhibition was measured as 9 mm for the dose of 200 mg formulation for gram-positive bacteria, Bacillus subtilis and 13 mm for gram-negative bacteria Pseudomonas aeruginosa. Norfloxacin and Ciprofloxacin were used as standard. It was found that the ethosomal gel of Mangifera indica herbal extract FN2 (Formulation 2, 200 mg) was found to possess greater zone of inhibition when compared to the other two formulations. From the study, it can be concluded that ethosomal gel served as an efficient drug delivery system for herbal extract with potential antibacterial activity.
| Keywords: |
Ethosomal gel, Mangifera indica, Anti-bacterial activity, Physico-chemical properties
INTRODUCTION: Herbal medicines play a significant role in in the health care system throughout the world. The focus is mainly on those herbal medicines that are easily available, cheaper, time tested and considered safer than most of the synthetic drugs. Plants which are used in traditional medicine contain a wide range of ingredients that can be used to treat chronic and infectious diseases. In 80% of the world population, the usage of antimicrobial agents derived from plants is considered as traditional health remedies.
Recognition and development of the medicinal and economic benefits of traditional medicinal plants are on the increase in both developing and industrialized countries 1. Numerous studies have been carried out to extract various natural products for screening anti-microbial activity, but attention has not been focused intensively on screening of formulations for their anti-bacterial activity.
Medicinal plants contain substances that can be used for therapeutic purposes or which are used as precursors for the synthesis of useful drugs. They also serve as therapeutic agents as well as important raw materials to produce traditional and modern drugs. World Health Organization recognized the value of traditional medicine and involved in creating strategies, guidelines, and standards for plant medicines 2.
As there is an increase in the failure of synthetic drugs, side effects and development of antibiotic resistance by pathogenic microorganisms lead to the development of the identification and screening of several medicinal plants for their potential anti-microbial activity. Mangifera indica is a plant which belongs to the family Anacardiaceae, grows in tropical and sub-tropical regions. Various parts of the plant were commonly used in folk medicine for a wide variety of remedies. Various parts of the plant were used to treat diarrhea, asthma, hypertension and insomnia 3. It is found worthwhile to investigate the antibacterial activity of M. indica leaves which was not reported yet. Formulation of the dosage form is necessary for easy administration of leaf extract of M. indica 4.
Drug delivery through the skin has been a promising concept for a long time because the skin is easy to access, has a large surface area with vast exposure to the circulatory and lymphatic networks and the route is non-invasive. Tropical preparations such as creams, ointments, and gels may be prepared where it can be spread to local inflammation sites. Transdermal drug delivery systems are gaining in popularity in this aspect. The main limiting factor of transdermal drug delivery, i.e., the epidermal barrier can be overcome by ethosomes when compared to transdermal dermal delivery 5.
Ethosomes are non-invasive delivery carriers that enable drugs to reach the deep skin layers and systemic circulation. Plant drugs are considered safe of their natural origin 6. Even after exhibiting the promising therapeutic effects, most of the phytoconstituents fail to achieve bioavailability because of poor absorption. Large molecular sizes and low lipid solubility are the prominent factors causing poor absorption of phytoconstituents resulting in reduced bioavailability. Incorporation of these plant actives or extracts into vesicular carriers vastly improves their absorption and consequently bioavailability 7. Indirectly ethosomes has become an area of research in herbal formulations because of their enhanced skin permeation and improved entrapment efficiency. Therefore, the present study was aimed at formulating and investigating an effective anti-bacterial ethosomal gel formulation of methanolic extract of M. indica leaves.
MATERIALS AND METHODS:
Collection of Plant material and Preparation of Extract: Mangifera indica leaves were collected from the local market, Hyderabad, India and were further authenticated by Dr. Madhava Chetty, Botanist, Tirupati, Andhra Pradesh. All the other solvents and reagents were of analytical grade. Fresh leaves of the plant were washed with water immediately after collection. These were chopped into small pieces, air dried at room temperature for 10 days, grounded into fine powder and stored in airtight containers. Powder weighing 650 grams was macerated with 5 liters of pure methanol for 7 days at room temperature. Later it was filtered and the extract was concentrated under reduced pressure below 50 °C in a rotary vacuum evaporator. It was kept in a petri dish for air drying to remove the traces of methanol and finally a concentrated extract is formed 8, 9.
Preparation of Ethosomes: Lipid and cholesterol were measured accurately and dispersed in water by stirring it on a magnetic stirrer for 30 minutes heating at 40 °C. The organic phase containing 100 mg of extract was added to ethanol and to this propylene glycol was added, kept for stirring separately. Lipid solution was added drop by drop to the organic phase and kept for stirring on a magnetic stirrer for 1 hour. 12 batches of ethosomal formulations were prepared using different concentrations of lipid (100 - 400 mg) and ethanol (10 - 40%). The optimized formulation was chosen, and further ethosomal preparations of other doses (200 mg, 300 mg) were formulated. The formulations with high entrapment efficiency and drug release were selected to incorporate into gel formulations 10.
Preparation of Ethosomal Gel: The gels were prepared by dispersion method using carbopol 940 using dispersing gelling agent in distilled water. Then the mixture was allowed to swell overnight. The mixture was neutralized by dropwise addition of triethanolamine. Then, glycerol was added to the gel to balance its viscosity. To this gel solution, optimized ethosomal dispersion was added and mixed properly. Mixing was continued until a transparent gel appeared. Paraben was added as a preservative. The prepared gels were filled in glass vials and stored at 4 - 8 °C 11.
TABLE 1: OPTIMIZATION OF CONCENTRATION OF LECITHIN
| Formulation code | Drug concentration (mg) | Lecithin (mg) | Cholesterol (mg) | Ethanol
(ml) |
Propylene glycol
(ml) |
| F1 | 100 | 100 | 20 | 10 | 3 |
| F2 | 100 | 200 | 20 | 10 | 3 |
| F3 | 100 | 300 | 20 | 10 | 3 |
| F4 | 100 | 400 | 20 | 10 | 3 |
Ethosomal dispersions F1 - F4 were prepared by varying the lecithin concentration. The dispersions were evaluated and based on the rate of drug release the lecithin concentration was optimized.
TABLE 2: OPTIMIZATION OF CONCENTRATION OF CHOLESTEROL
| Formulation
code |
Drug concentration (mg) | Lecithin
(mg) |
Cholesterol
(mg) |
Ethanol
(ml) |
Propylene Glycol (ml) |
| F5 | 100 | 300 | 20 | 10 | 3 |
| F6 | 100 | 300 | 30 | 10 | 3 |
| F7 | 100 | 300 | 40 | 10 | 3 |
| F8 | 100 | 300 | 50 | 10 | 3 |
Ethosomal dispersions F5 - F8 were prepared by varying cholesterol concentration. Based on drug release the cholesterol concentration was optimized.
TABLE 3: OPTIMIZATION OF ETHANOL CONCENTRATION
| Formulation
code |
Drug concentration
(mg) |
Lecithin
(mg) |
Cholesterol
(mg) |
Ethanol
(ml) |
Propylene glycol
(ml) |
| F9 | 100 | 300 | 40 | 10 | 3 |
| F10 | 100 | 300 | 40 | 20 | 3 |
| F11 | 100 | 300 | 40 | 30 | 3 |
| F12 | 100 | 300 | 40 | 40 | 3 |
Ethosomal dispersions F9-F12 were prepared by varying the ethanol concentration and based on the drug release the ethanol concentration was optimized.
Evaluation of Prepared Ethosomes: Amongst all the formulations, F10 formulation was optimized based on % entrapment efficiency and drug release 12 - 14.
Morphology: The samples were visualized by scanning electron microscopy (SEM, Hitachi S-3700N), gives a three-dimensional image of the globules. One drop of ethosomal suspension was mounted on a stub covered with a clean glass. It was then air dried, and gold coated using sodium aurothiomalate to visualize under scanning electron microscope 10,000 magnifications.
Zeta Potential: Zeta potential was determined using Zetasizer (HORIBA SZ-100). Measurements were performed on the same samples prepared for size analysis. Zeta potential indicates the degree of repulsion between adjacent, similarly charged particles in the dispersion system.
Entrapment Efficiency (EE): Entrapment efficiency of Mangifera indica ethosomal vesicles was determined by centrifugation. The vesicles were separated in a high-speed cooling centrifuge at 20,000 rpm for 90 minutes. The sediment and supernatant liquids were separated, the amount of drug in the sediment was determined by lysing the vesicles using methanol. It was then diluted appropriately and estimated using UV visible spectrophotometer at 214 nm. From this, the entrapment efficiency was determined by the following equation –
Formulation of Gels: Gels were prepared by adding dispersing gelling agent to distilled water. Then the mixture was allowed to swell overnight. The mixture was neutralized by dropwise addition of triethanolamine. Then, glycerol was added to the gel to balance its viscosity.
EE% = (Total drug) - (free drug) × 100 / Total drug
TABLE 4: GELS PREPARED BY DISPERSION METHOD USING CARBOPOL 940 IN DIFFERENT RATIOS
| S. no. | Formulation | Carbopol 940 (% w/v) | Amount of extract |
| 1 | EG1 | 1 | 100 |
| 2 | EG2 | 1 | 200 |
| 3 | EG2 | 1 | 300 |
To this gel solution optimized ethosomal dispersion was added and mixed properly. Mixing was continued until a transparent gel appeared. Paraben was added as a preservative. The prepared gels were filled in glass vials and stored at 4 - 8 °C 15.
Evaluation of Prepared Gels: 16
Physicochemical Properties:
Appearance: The appearance was checked visually. They are light greenish.
pH Measurement: The pH was checked using pH meter (Systronics digital pH meter). The electrode was submerged into the formulation at room temperature, and the readings were noted.
Spreading Diameter: The spreadability of gel formulation was determined by measuring the spreading diameter of 1g of gel between two horizontal plates (20 cm × 20 cm) after 1 min. The standard weight applied on the upper plate was 125 gm.
Viscosity: Viscosity of prepared formulations was prepared by Brookfield Synchro Electric Viscometer (LVDV Pro II), spindle S64 (small sample adaptor) and the angular velocity was increased from 5, 10, 50, 100 rpm and values were noted.
Drug Content of the Formulated Gels: Drug content was estimated spectrophotometrically, 100 mg of the formulation was taken and dissolved in methanol and filtered. The volume was made up to 100 ml with methanol. The resultant solution was suitably diluted with methanol and absorbance was measured at 212 nm.
In-vitro Drug Release: The Franz diffusion cell consisted of two compartments (cells). The upper one is donor cell, consisting of two open ends and the lower one is receptor cell, with one open end capacity of 15 ml. One end of the donor compartment was covered with Himedia dialysis membrane (cut off molecular weight 12000 -14000), which was previously soaked in warm water and placed on the receptor compartment. The receptor cell contained a small magnetic bead and was rotated at a constant speed. The temperature in the donor and receptor cells was maintained at 37 °C, with the help of a thermostat. Phosphate buffer 7.4 was placed in the receptor cell.
A 5 ml sample of each formulation was transferred to the diffusion cell. 3 ml samples were withdrawn from the receptor cell at specified time intervals. Each time immediately after the removal of the sample, the medium was compensated with the fresh media. The samples were analyzed for drug content using a UV - Visible spectrophotometer at 212 nm 17.
Anti-bacterial Activity: The antibacterial activity of the extracts was evaluated using the agar well diffusion assay. Weighed Muller Hinton Agar as per requirement and dissolved in distilled water as per the guidelines are given by the manufacturer and autoclaved the media at 121 °C for 15 min.
After autoclaving, poured the media in sterile Petri plates and kept for solidification. Cork borer was sterilized by autoclaving or disinfected it by rinsing in alcohol followed by sterile water. On a Muller Hinton Agar plate aseptically 5 holes were punched using a cork borer. Using a marker, marked the underside of the Petri to label the wells.
Aseptically 20 µl of the indicator organism was spread onto the MH agar plate. Allowed the plate to stand for 5 min. Placed 10 µl, 25 µl, 50 µl, 75 µl and 100 µl of the extract in the appropriate wells. The plates were incubated at 37 °C for 24 hrs. The zones of inhibition were measured in millimeters, using a ruler on the underside of the plate. Same Procedure was repeated with standard with concentrations of 10 µg, 25 µg, 50 µg, 75 µg and 100 µg 18.
Gram Positive Strain: Bacillus subtilis.
Gram Negative Strain: Pseudomonas aeruginosa.
Standard Drug for Gram-Positive: Norfloxacin.
Standard Drug for Gram Negative: Ciprofloxacin.
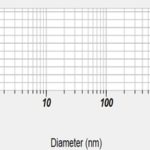
RESULTS AND DISCUSSION: The microscopic evaluation showed the surface morphology of ethosomes. It was observed that most of the vesicles were spherical and its smooth surface was further confirmed by SEM. The vesicular size of the ethosomes significantly increased with increase in phospholipid concentration and decreased with increased concentration of ethanol.
FIG. 1: THE AVERAGE SIZE OF ETHOSOMES AS 926 nm
FIG. 2: THE PARTICLE SIZE OF ETHOSOMES
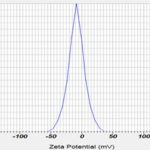
The zeta potential of the ethosomes was determined using zeta sizer. From Fig. 3 the value of the
optimized ethosomal formulation was found to be -8.8mv which indicated that ethosomes were stable.
FIG. 3: THE ZETA POTENTIAL OF ETHOSOMES
The entrapment efficiency of ethosomes was found to be in the range of 65.31 - 89.38%. The entrapment efficiency was found to be higher for the formulation F10. The entrapment efficiency was influenced by amounts of ethanol, lecithin, and cholesterol which were used for preparation. Of all the factors examined the concentration of ethanol was found to influence the entrapment efficiency to a significantly increased level due to the formation of a thinner membrane.
TABLE 5: ENTRAPMENT EFFICIENCY AND % DRUG RELEASE OF DIFFERENT FORMULATIONS
| S. no. | Formulation Code | Entrapment Efficiency | % Drug Release |
| 1 | F1 | 65.31 ± 0.22 | 63.98 ± 0.37 |
| 2 | F2 | 68.42 ± 0.5 | 72.2 ± 0.54 |
| 3 | F3 | 70.88 ± 0.31 | 74.75 ± 0.2 |
| 4 | F4 | 71.5 ± 0.66 | 72.85 ± 0.72 |
| 5 | F5 | 68.65 ± 0.26 | 73.53 ± 0.24 |
| 6 | F6 | 72.73 ± 0.9 | 77.06 ± 0.14 |
| 7 | F7 | 70.82 ± 0.67 | 80.58 ± 0.21 |
| 8 | F8 | 75.2 ± 0.36 | 76.8 ± 0.12 |
| 9 | F9 | 82.4 ± 0.44 | 82.62 ±0.73 |
| 10 | F10 | 89.58 ± 0.26 | 87.79 ± 0.50 |
| 11 | F11 | 84.33 ± 0.45 | 86.5 ± 0.42 |
| 12 | F12 | 86.21 ± 0.33 | 87.88 ± 0.5 |
In the in-vitro drug release, the cumulative percentage of drug releases from various ethosomal formulations were done. The formulation F10 showed higher drug release of 87.79% in 8 hrs. Therefore, F10 has been selected for formulating the ethosomal gel and based on this, different doses of 100, 200 and 300 mg drug extract were also formulated. In the evaluation of ethosomal topical gel, all the formulations were found to be opaque, light greenish in color, odorless, semi-solid and had a smooth appearance.
TABLE 6: EVALUATION OF PHYSICOCHEMICAL PROPERTIES OF GEL FORMULATIONS
| Formulation | Colour | Appearance | Spread ability (g.cm/sec) | pH | Viscosity (cps) | Drug content % |
| EG1 | Greenish | Homogenous | 35.07 ± 0.86 | 5.6 | 2399 | 74.67 |
| EG2 | Greenish | Homogenous | 33.72 ± 0.52 | 5.8 | 2574 | 78.92 |
| EG3 | Greenish | Homogenous | 34.62 ± 0.67 | 5.5 | 2250 | 82.31 |
The pH for all the formulations exhibited in the range of 5.4 - 6.2. The formulations were analyzed Spectrophotometrically at 212 nm. All the formulations were found to possess uniform drug content.
The viscosity of all the gel formulations ranged from 2250 - 2574 cps. The viscosity of the formulations decreased on increasing the shear rate, i.e. pseudoplastic behavior was noted. In the in vitro drug release, the cumulative percentage drug release after 8 hrs was highest for all the three doses of extracts using 1% carbopol. The drug content of the gels ranged between 74.67 - 82.31%.
The herbal ethosomal gel was subjected to acute skin irritation studies and anti-inflammatory activity.
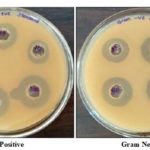
FIG. 4: ZONE OF INHIBITION IN mm
TABLE 7: ZONE OF INHIBITION FOR GRAM POSITIVE AND GRAM-NEGATIVE BACTERIA
| Strain | (10 µg) Zone of Inhibition (mm) | (25 µg) Zone of Inhibition (mm) | (50 µg) Zone of Inhibition (mm) | (75 µg) Zone of Inhibition (mm) | (100 µg) Zone of Inhibition (mm) |
| Gram Positive | 4 | 9 | 10 | 11 | 13 |
| Gram Negative | - | 11 | 13 | 15 | 16 |
TABLE 8: ZONE OF INHIBITION FOR GRAM-POSITIVE BACTERIA
| S. no. | EG (Formulation) | (10 µg) Zone of Inhibition (mm) | (25 µg) Zone of Inhibition (mm) | (25 µg) Zone of
Inhibition (mm) |
(75 µg) Zone of Inhibition (mm) | (100 µg) Zone of Inhibition (mm) |
| 1 | 100 mg | 0 | 2 | 4 | 6 | 8 |
| 2 | 200 mg | 1 | 2 | 5 | 6 | 9 |
| 3 | 300 mg | 1 | 2 | 4 | 4 | 7 |
| 4 | Control | 0 | 0 | 0 | 0 | 0 |
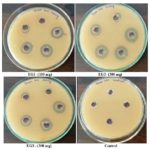
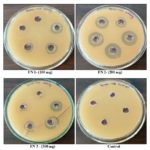
FIG. 5: ZONE OF INHIBITION OF INDIVIDUAL FORMULATIONS FOR GRAM-POSITIVE BACTERIA
TABLE 9: ZONE OF INHIBITION FOR GRAM-NEGATIVE BACTERIA
| S. no | (FN) Formulation | (10 µg) Zone of
Inhibition (mm) |
(25 µg) Zone of
Inhibition (mm) |
(50 µg) Zone of Inhibition (mm) | (75 µg) Zone of Inhibition (mm) | (100 µg) Zone of
Inhibition (mm) |
| 1 | 100 mg | 0 | 6 | 6 | 2 | 0 |
| 2 | 200 mg | 6 | 9 | 11 | 12 | 13 |
| 3 | 300 mg | 0 | 1 | 4 | 6 | 9 |
| 4 | Control | 0 | 0 | 0 | 0 | 0 |
FIG. 6: ZONE OF INHIBITION OF INDIVIDUAL FORMULATIONS FOR GRAM-NEGATIVE BACTERIA
Anti-bacterial Activity: The results of the antibacterial activity by the agar well diffusion method of the three formulations EG1, EG2, EG3 were presented in Table 8 and 9 for gram-positive and gram-negative bacteria. It was found that all the formulations inhibited the growth of almost all the selected bacteria in the range of 1 - 13 mm at different concentrations Fig. 5 and 6. Among them formulation EG 2 showed great activity against Pseudomonas aeruginosa followed by Bacillus subtilis. To overcome the basic needs in developing countries, the use of medicinal plants play a significant role in combating infectious diseases.
CONCLUSION: The present study demonstrated that the M. indica herbal ethosomal gel formulation possesses significant topical anti-bacterial property, supporting the use as an effective medicine to overcome the infections due to pathogenic micro-organisms.
ACKNOWLEDGEMENT: Authors are thankful to Principal Dr. Bhagavan Raju, Sri Venkateshwara College of Pharmacy, for support, encouragement and providing facilities to carry out the work. Also, thankful to Osmania University, Hyderabad who extended their support for the project.
CONFLICT OF INTEREST: The authors have declared no conflicts of interest.
REFERENCES:
- Dhanalakshmi D and Manimegalai K: Anti-bacterial activity of leaf and seed extracts of Delonix regia and Achyranthus aspera against selected bacterial strains. Int J Pharm Med 2013; 2(2): 31-35.
- Bibitha B, Jisha VK, Salitha CV, Mohan S and Valsa AK: Anti-bacterial activity of different plant extracts, short communication. Indian J Microbiol 2002; 42: 361-363.
- Mascud Parvez GM: Pharmacological activities of Mango (Mangifera indica): A Review. Journal of Pharmacognosy and Phytochemistry 2016; 5(3): 01-07.
- Ateb DA and Erdourul T: Anti-microbial activities of various medicinal and commercial plant extracts. Turk J Biol 2003; 27: 157-62.
- Tarun P, Soniya, Roopesh S, Vishal S, Gaurav S, Satyanand T, Chirag P and Anil G: Ethosomes: A recent vesicle of transdermal drug delivery system. Int J Res Dev Pharm L Sci 2013; 2(2): 285-292.
- Nimisha N: Development and Evaluation of herbal Cosmoceutical for skin care. Int J Pharm Bio Sci 2013; 4(2): 86-2.
- Jordan SA, Cunningham DG and Marles RJ: Assessment of herbal medicinal products: Challenges and opportunities to increase the knowledge base for safety assessment. Toxicology and applied Pharmacology 2010; 243: 198-16.
- Ajila CM and Prasad Rao UJ: Protection against hydrogen peroxide-induced oxidative damage in rat erythrocytes by Mangifera indica peel extract. Food chemical toxicology 2008; 46(1): 303-09.
- Rodriguez J, Di-pierro D and Gioia M: Effects of natural extracts from Mangifera indica L and its active compound, mangiferin, on energy state and lipid peroxidation of red blood cells. Biochemical Biophysics 2006; 1760(9): 1333-42.
- Tarun P, Soniya, Roopesh S, Vishal S, Gaurav S, Satyanand T, Chirag P and Anil G: Ethosomes: A recent vesicle of transdermal drug delivery system. Int J Res Dev Pharm L Sci 2013; 2(2): 285-92.
- Sujitha B, Krishnamurthy B and Muthukumaran M: Formulation and evaluation of Piroxicam loaded ethosomal gelfor transdermal delivery. International Journal of advanced Pharmaceutical genuine research 2014; 2(1): 34-45.
- David SRN, Hui MS, Pin CF, Ci FY and Rajabalaya R: Formulation and in-vitro evaluation of ethosomes asvesicular carrier for enhanced topical delivery of isotretinoin. International Journal of drug delivery 2013; 5(1): 28.
- Rodriguez J, Di-pierro D and Gioia M: Effects of natural extracts from Mangifera indica L and its active compound, mangiferin, on energy state and lipid peroxidation of red blood cells. Biochemical Biophysics 2006; 1760(9): 1333-42.
- Indira S, Priyanka R and Prathima S: Formulation and evaluation of ethosomal topical gels of Etoricoxib. International Journal for Pharmaceutical Research Scholars 2015; 4(1): 93-03.
- Bhana R, Verma A and Jain S: Development and characterization of ethosomes bearing losartan potassium for transdermal drug delivery. International Journal of Pharmacy and Pharmaceutical Sciences 2013; 5(1): 35-40.
- Missal G, Dixit G and Gulkari V: Formulation and evaluation of herbal gel. Indian Journal of natural products and Research 2012; 3(4): 501-05.
- Esayed MM, Abdallah OY, Naggar VF and Khalafallah NM: Lipid vesicles for skin delivery reviewing three decades of research. International Journal of Pharmaceutics 2007; 332: 1-16.
- Nita T, Arai T and Takamatsu H: Anti-microbial activity of extracts prepared from tropical and subtropical plants on methicillin-Resistant Staphylococcus aureus. J Health Sci 2002; 48: 273-76.
How to cite this article:
Rao PS, Sundari BT and Kalva S: Formulation and evaluation of gel containing Mangifera indica leaves extract for anti-bacterial activity. Int J Pharmacognosy 2018; 5(1): 61-68. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.5(1).61-68.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
7
61-68
664
789
English
IJP
P. S. Rao *, B. T. Sundari and S. Kalva
Sri Venkateshwara College of Pharmacy, Madhapur, Hyderabad, Telangana, India.
sailajarao476@gmail.com
21 July 2017
23 August 2017
17 September 2017
10.13040/IJPSR.0975-8232.IJP.5(1).61-68
01 January 2018