EXPLORATORY STUDIES ON DIABETIC WOUND HEALING POTENTIAL OF CIPADESSA BACCIFERA (ROTH.) MIQ
HTML Full TextEXPLORATORY STUDIES ON DIABETIC WOUND HEALING POTENTIAL OF CIPADESSA BACCIFERA (ROTH.) MIQ
A. B. Murkute and V. M. Shinde *
Poona College of Pharmacy, Bharati Vidyapeeth (Deemed to be University), Erandawane, Pune - 411038, Maharashtra, India.
ABSTRACT: Growing burden of degenerative metabolic diseases can be addressed with help of traditional and folkloric medicine. The purpose of this study is to evaluate an ethanolic defatted extract (EDE) of Cipadessa baccifera (CB) leaf for anti-diabetic wound healing activity in male albino Wistar rats. The defatted and non-defatted extracts were prepared using water and ethanol solvent system. They were evaluated for pharmacognostic study, wound healing activity using animal excision and incision models. Antioxidant activity was performed using DPPH assay. Ascorbic acid was used as reference standard for determination of antioxidant activity. The antimicrobial activity was also carried out by disc diffusion and well diffusion method using chloramphenicol as reference standard. The EDE of leaf showed higher total phenolic content. It was found to be 389.2 mg gal/l. These extract showed good DPPH radical scavenging activity, which was found to be 68.75% in comparison with ascorbic acid. This extract also showed a good zone of inhibition with disc diffusion and well diffusion method. On 16th day, the 200 mg/kg EDE treated group showed 95% wound closure while 100 mg/kg treated showed 75% wound closure. The EDE of CB leaf showed prominent antioxidant and anti-microbial activity. It also exhibited potential wound healing activity.
| Keywords: |
Diabetic wound healing, Cipadessa baccifera, Anti-bacterial, Anti-oxidant, Phenolic constituents, Excision and incision wound
INTRODUCTION: Traditionally Cipadessa baccifera (Family-Meliaceae) is used to treat various diseases like dysentery, malaria, pruritus (itchy skin), rheumatism, burns, diabetes, diarrhea, headaches, piles, snake poison, psoriasis 1, 2. It is an evergreen shrub growing up to a height of 1 - 4 m with coarse bark which is present in Western Ghats of India and Sri Lanka 1.
The leaf of CB contains terpenoids like sesquiterpenes such as caryophyllene, isoledene, cis-clamenene and β-sesquiphellandrene which exhibit anticancer, anti-inflammatory and immunosuppressive activity 3. The leaf is a source of bioactive compounds like limonoids such as gedunin, havanensin, mexicanolide and methyl angolensate types and cipadesins J-Q which acts as anti-feedant, antimicrobial, anti-malarial, anti-cancer 4.
Diabetes Mellitus (DM) is a metabolic disorder in which action of insulin hormone is impaired which affects glucose level in bloodstream 5. High levels of glucose in blood stream impair the function of white blood cells due to which body cannot fight against bacteria and show impaired wound healing. People with diabetes show poor blood circulation which makes it more difficult for the body to provide nutrients to wounded part 6. The wound has been defined as the disruption of anatomic or functional continuity of living tissue 7.
Wound infection is one of the most common diseases in developing countries due to the poor standard of living conditions 8. The wound healing process consists of four main events which are as follows stage 1: Haemostasis: is to prevent excessive blood loss. In this phase platelets cause vasoconstriction and blood coagulation which prevent excessive blood loss 9. Stage 2: Inflammation: In this stage of wound healing neutrophils and macrophages cells act as first line of defense against microorganisms and cell debris through phagocytosis mechanism 10. Stage 3: Proliferation: This stage involves reconstruction of skin cells with the help of macrophages, lymphocytes, angiocytes, neurocytes, fibroblasts, keratinocytes. Stage 4: Remodelling: In this stage tensile strength gets restored with the help of fibrocytes 11. The diabetic wound healing is complex involving blood sugar, lipid and protein also. Many plants from the traditional system of medicine have proved scientifically hypoglycemic activity in diabetes-induced animals acting on multiple targets 12. The present study is designed with an aim to develop a cost-effective approach for diabetic wound healing.
MATERIALS AND METHODS:
Plant Collection and Authentication: The leaves of CB were collected from Pune, Maharashtra, India. Authentication was done from the Botanical Survey of India, Pune (BSI/WRC/100-2/Tech./2017/12), and voucher specimen were preserved in departmental herbarium.
Extraction of Plant Material: The leaves sample of CB was collected and dried under sunlight. These sun-dried leaves of CB were subjected for size reduction using a mixer grinder to obtain a homogenous sample. Aqueous extract (AE) and ethanolic extract (EE) were prepared by using Soxhlet apparatus. Remaining two extracts were prepared after de-fatting with pet ether and named as ethanolic defatted extract (EDE) and aqueous defatted extract (ADE) by using the same method.
All extracts were dried in a rotary flash evaporator and stored in refrigerator and used for subsequent studies. All the chemicals and solvents used for extraction were of analytical grade.
Physico-Chemical Analysis and Phytochemical Tests: Physico-chemical analysis, i.e. percentage of ash values and extractive values and phytochemical tests were performed according to the methods prescribed in Khandelwal & Sethi V 13.
Antimicrobial Activity: Anti-microbial study was performed by using strains of Escherichia coli (MCC2246) and Staphylococcus aureus (MCC2408). Microorganisms were provided by National Centre for Microbial Resource (NCMR), Pune. Disc diffusion method was used as described by Vincent JG & Vincent HW (1944) 14, Tendencia EA (2004) 15. Also, a well diffusion study was performed according to Holder IA & Boyce ST (1994) 16, Maher Obeidat et al., (2012) 17.
Estimation of Total Phenolic Content: The total phenolic content was calculated as per the protocol described by Singleton VL & Rossi JA (1965) 18, Pobba Rajani et al., (2015) 19, Murugammal S & Ilavarasan R (2016) 20.
Estimation of Anti-oxidant Activity: Anti-oxidant activity was done by DPPH assay according to Blois MS (1958) 21, Gangwar M et al., (2014) 22, Valvi SR et al., (2011) 23, Kedare SB & Singh RP (2011) 24.
Diabetic Wound Healing Studies:
Animals: The wound healing activity was carried out using male albino Wistar rats weighing 150 - 200 g of either sex. They were housed under controlled conditions of room temperature (25 °C), humidity (50 ± 5), and 10 - 14 h light and dark cycles. The animals were housed individually in polypropylene cages containing sterile paddy husk bedding and given free access to food and water.
The experiments were designed and conducted in accordance with ethical norms approved by Committee for the Purpose of Control and Supervision on Experiments on Animals (CPSCEA) and Institutional Animal Ethical Committee (IAEC) with approval number CPCSEA/PCP/PCG 02/2018.
Grouping of Animals: The animals were randomly allocated into 4 groups (6 animals in each group). The details are as follows:
Group I: Control group (blank),
Group II: Treated with reference standard Povidon-Iodine ointment USP (manufactured by Nanz Med Science Pharma Pvt. Ltd.)
Group III: Animals treated with 100 mg/kg EDE,
Group IV: Animals treated with 200 mg/kg EDE as given in Table 1.
TABLE 1: GROUPING OF ANIMALS
| Groups | Drug | Route of administration | Dose |
| Group I Control | Water | - | - |
| Group II Standard | Povidone iodine | Topical | Standard dose |
| Group III | 100 mg/kg EDE | Oral | 1 ml |
| Group IV | 200 mg/kg EDE | Oral | 1 ml |
Wound Healing Models:
Alloxan Induced Diabetic Condition in Rats: The animals were injected intraperitoneally a single dose of Alloxan monohydrate (120 mg/kg, body weight) in normal saline. After 3 days, fasting blood glucose level was checked by Accu chek glucometer to confirm the diabetic status of the animal.
Excision Model: The excision model was carried out according to method described by Morton JJ & Malone MH (1972) 25, Riazul HT (2017) 26.
Incision Model: The incision model was performed according to the method described by Lee KH (1968) 27, Mugade M et al., (2017) 28.
Histopathology: For histopathological evaluation, the rats were sacrificed after the 21th day of excision and 10th day of incision. The healed skin tissue samples of rats were isolated and preserved in 10% buffer formalin for histological examination. After that histological evaluation was done in Chaitanya Laboratories, Baner, Pune 29.
RESULT AND DISCUSSION:
Physico-Chemical Analysis: Pharmacognostic studies such as extractive values, ash values, and preliminary phytochemical studies were determined to evaluate quality of raw material of plant CB. The ash values and extractive values of CB leaves were determined and given in Tables 2 and 3 respectively.
TABLE 2: DETERMINATION OF ASH VALUES
| S. no. | Parameters | % w/w |
| 1 | Total ash | 6.1 |
| 2 | Acid insoluble ash | 0.6 |
| 3 | Water-soluble ash | 6.5 |
| 4 | Water-insoluble ash | 1.5 |
In Table 2, the ash value is used to determine the identity and purity of crude drugs. Many times crude drugs are contaminated with an inorganic material such as silica. The change in ash value of crude drugs indicates adulteration.
TABLE 3: DETERMINATION OF EXTRACTIVE VALUES
| S. no. | Parameters | % w/w |
| 1 | Alcohol soluble extractive | 15.5 |
| 2 | Water soluble extractive | 20.23 |
As displayed in Table 3 alcohol soluble extractive value was found to be lesser than water-soluble extractive value. It may indicate that plant contains phytoconstituents which are more polar and soluble in water.
Phytochemical Tests: The preliminary phyto-chemical analysis of different extracts of CB was given in Table 4. The aqueous, aqueous defatted, ethanolic, EDE of the plant were subjected to qualitative preliminary phytochemical analysis. The preliminary phytochemical screening was carried out to know the type and nature of phytoconstituents present in different extracts of CB plant. The results were shown in Table 4. In the present study, EE and EDE leaf extracts of plat CB showed the presence of tannin, phenols, alkaloids, flavonoids, saponins, terpenoids, and AE and EDE extract alkaloids, flavonoids, saponin, phenols, tannin proteins, and amino acids.
These secondary metabolites contribute significantly towards the biological activities of medicinal plants, for example, anti-diabetic, hypoglycemic, anti-microbial, anti-oxidant, anti-inflammatory, anti-malarial activities, pruritus, rheumatism and burns, etc 30. The analysis revealed the presence many effective compounds like alkaloids, flavonoids, steroids, triterpenoids, carbohydrates, proteins, tannins and phenolic compounds, saponins, and glycosides in our plant species.
TABLE 4: THE PRELIMINARY PHYTOCHEMICAL ANALYSIS OF DIFFERENT EXTRACTS OF CB
| Cipadessa baccifera | |||||
| S. no. | Phytochemical | AE | ADE | EE | EDE |
| 1 | Alkaloids | + | + | + | + |
| 2 | Carbohydrates | + | + | - | - |
| 3 | Saponins | + | + | + | + |
| 4 | Protein | + | + | - | - |
| 5 | Steroid | - | - | - | - |
| Terpenoids | - | - | + | + | |
| 6 | Glycosides | - | - | - | - |
| 7 | Tannins and Phenols | + | + | + | + |
| 8 | Flavonoids | + | + | + | + |
| 9 | Amino acids | + | + | - | - |
AE= aqueous extract, ADE= aqueous defatted extract, EE= ethanolic extract, EDE= ethanolic defatted extract, - = absent, + = present)
Anti-microbial Study: The anti-microbial activity of five different extracts of CB leaves was evaluated using two different methods such as the disc diffusion method and well diffusion method. The measurements of zone of inhibition for each extract were calculated using scale in cm.
The results are displayed in Table 5 and 6. The above four different extracts were assessed for their anti-microbial activity against E. coli using disc diffusion method. As per above table, EE showed zone of inhibition 1.25 and 1.3 cm against E. coli at the concentrations of 140 mg/ml and 160 mg/ml respectively in comparison with standard antibiotic chloramphenicol. The EDE extract showed 1.35 cm and 1.55 cm at the concentration of 140 mg/ml and 160 mg/ml respectively against E. coli. EE at 140 mg/ml and 160 mg/ml concentrations showed 1 cm zone of inhibition for S. aureus. The EDE showed 1.1 and 1.2 cm zone of inhibition at the concentration of 140 mg/ml and 160 mg/ml respectively. AE and aqueous defatted extract (ADE) failed to show anti-microbial activity against E. coli and S. aureus in this method.
TABLE 5: MEASUREMENTS OF ZONE OF INHIBITION IN CM BY DISC DIFFUSION METHOD
| Extracts | Zone of inhibition (cm) * | |||||
| E. coli | S. aureus | |||||
| 140 mg/ml | 160 mg/ml | STD | 140 mg/ml | 160 mg/ml | STD | |
| AE | - | - | 1.75 ± 0.07 | - | - | 1.51 ± 10.14 |
| ADE | - | - | 1.1 ± 0.14 | - | - | 1.35 1 ± 0.07 |
| EE | 1.25 ± 0.07 | 1.3 ± 0.14 | 1.6 ± 0.42 | 1 ± 0 | 1 ± 0 | 1.51 ± 10.28 |
| EDE | 1.35 ± 0.07 | 1.55 ± 0.07 | 1.85 ± 0.07 | 1.1 ± 0.28 | 1.2 ± 0.28 | 1.75 ± 0.21 |
(*Mean ± standard deviation) n =2
The above four different extracts were assessed for their anti-microbial activity against E. coli and S. aureus using the well diffusion method. As per above table, At the concentration of 140 mg/ml and 160 mg/ml of EE showed zone of inhibition 0.95 cm and 1.05 cm respectively in comparison with standard antibiotic chloramphenicol against E. coli. The EDE showed zone of inhibition 1.05 cm and 1.15 cm at the concentration of 140 mg/ml and 160 mg/ml respectively against E. coli.
EE showed a zone of inhibition 1 cm and 0.70 cm at 140 mg/ml and 160 mg/ml respectively against S. aureus. Also EDE showed 1.1 cm and 0.95 cm zone of inhibition at the concentration of 140 mg/ml and 160 mg/ml respectively against S. aureus. AE and ADE failed to show antimicrobial activity against E. coli and S. aureus in this method. From the above result it can be concluded that the 140 mg/ml was the minimum inhibitory concentration in this method.
TABLE 6: MEASUREMENTS OF ZONE OF INHIBITION IN CM BY WELL DIFFUSION METHOD
| Extracts | Zone of inhibition (cm) * | |||||
| E. coli | S. aureus | |||||
| 140 mg/ml | 160 mg/ml | STD | 140 mg/ml | 160 mg/ml | STD | |
| AE | - | - | 2.3 ± 0.424 | - | - | 2.2 ± 0.14 |
| ADE | - | - | 2.2 ± 0.282 | - | - | 1.8 |
| EE | 0.95 ± 0.070 | 1.05 ± 0.070 | 1.9 ± 0.141 | 1 ± 0 | 0.7 ± 0.28 | 2.3 ± 0.28 |
| EDE | 1.05 ± 0.070 | 1.15 ± 0.070 | 2.1 ± 0.424 | 1.1 ± 0.14 | 0.95 ± 0.07 | 2.45 ± 0.21 |
(*Mean ± standard deviation) n =2
In this study, EDE extract of CB showed significant inhibitory activity against bacterial strains of E. coli and S. aureus in the disc diffusion and well diffusion method also. This anti-microbial activity is may be due to the presence of various phytoconstituents such as tannin, phenols, alkaloids, flavonoids, saponins, and terpenoids as reported in preliminary phytochemical screening. Secondary metabolites can be responsible for different therapeutic effects 31. Phytochemical constituents such as tannins, phenols, alkaloids, flavonoids, terpenoids, saponins and other several secondary metabolites of plants that provide a defense mechanism against various micro-organisms 32. Saponin showed antimicrobial property due to its ability to cause protein leakage and certain enzymes from cell 33. Flavonoids are hydroxylated phenolic substances which are synthesized by plants as defence mechanism against microbial infection 34. The anti-microbial activity may be beneficial to inhibit microbial infection generally observed in wounds.
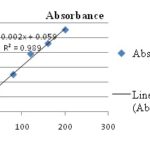
Estimation of Total Phenolic Content: The total phenolic content in the extract was assessed by using Folin–Ciocalteu reagent. Gallic acid was used as a reference standard. A linear calibration curve Fig. 1 was obtained by way of Y=0.002X+0.059 (R2 = 0.989) where X is the concentration of extract and Y is absorbance at 765 nm. The total phenolic content present in the EDE of CB was found to be higher as compared to other all extracts was 389.2 mg gal/l. The EE showed less total phenolic content than EDE was found to be 383.5 mg gal/l. The ADE of CB showed higher total phenolic content than AE 136.6 mg gal/l and 121.55 mg gal/l respectively.
Phenolic compounds from different plants have shown promising antibacterial activity 35. In the present study, the EDE showed higher phenolic content which may be responsible for antimicrobial and antioxidant activities. Leaves contain many varieties of anti-oxidants. Hence, the capacity of the antioxidant of each constituent was evaluating separately becomes very complicated 36.
There are various methods have been reported for the estimation of antioxidant capacity of various plant materials. These methods measure scavenging potential of the anti-oxidant compounds or extracts in terms of hydrogen donating ability 37. In this study, DPPH method was used to measure the antioxidant capacity of extracts of CB.
FIG. 1: CALIBRATION CURVE OF GALLIC ACID FOR THE PHENOLIC ESTIMATION
Antioxidant Activity:
DPPH Assay: 1, 1 Diphenyl 2- Picryl Hydrazyl is a stable free radical with red color which turns into yellow color when scavenged. The level of discoloration represents the scavenging potential of the extracts. The absorption of the DPPH solution was decreased after adding different extracts of CB was measured given in following Table 7.
TABLE 7: EVALUATION OF DPPH SCAVENGING ACTIVITY OF VARIOUS CB EXTRACTS
| % inhibition of CB extracts* | |||||
| Concentration (μg/ml) | STD | AE | ADE | EE | EDE |
| 50 | 79.4 ± 0.28 | 38.61 ± 0.23 | 40.35 ± 0.06 | 33.04 ± 0.07 | 33.67 ± 0.10 |
| 100 | 40.29 ± 0.08 | 43.3 ± 0.12 | 34.46 ± 0.04 | 38.76 ± 0.06 | |
| 150 | 44.61 ± 0.03 | 51.51 ± 0.06 | 36.79 ± 0.19 | 50.93 ± 0.20 | |
| 200 | 49.22 ± 0.12 | 57.28 ± 0.24 | 38.2 ± 0.28 | 59.06 ± 0.07 | |
| 250 | 55.95 ± 0.08 | 60.59 ± 00 | 41.67 ± 0.03 | 68.75 ± 0.02 | |
(*Mean ± standard deviation) n =2
From the above result, it was found that the EDE of CB leaves shown higher inhibition of DPPH radical than other extracts. The ethanolic extract of CB leaves shown less inhibition of DPPH radical. The EDE and ethanolic extract of CB was shown 68.75% and 41.67% at concentration of 250 μg/ml respectively. The ADE showed less DPPH inhibition than aqueous extract was found to be 60.59 % and 55.95 at higher concentration was 250 μg/ml respectively.
Valvi SR et al., (2011) 23, have shown the antioxidant potential of CB fruit collected from Kolhapur, Maharashtra, India. As per this study the free radical scavenging activity by DPPH method was found to be 53.19% and 40.21% respectively 23. In our study, EDE and ADE of leaves of CB plant showed relatively high values, which were 68.75% and 60.59%, respectively. As reported in various papers, there is direct correlation between the total phenolic content of compound and antioxidant capacity of plants. The present study also revealed that extract containing highest total phenolic content acts as potential antioxidant 29, 38.
In the present study, the secondary metabolites like tannins, phenols, and flavonoids are present. The phenolic acids and flavonoids due to their degree of hydroxylation are known for their potential radical scavenging activity. The CB leaves derived antioxidants like phenolic acids, tannins, flavonoids, and terpenoids could prevent the degenerative diseases onset because of redox properties. The redox properties of phyto-constituents are responsible for activities such as reducing agent, superoxide radical, hydrogen radical scavengers 9.
Alloxan Induced Diabetic Condition in Rats: The normal blood glucose level in the test rats was found 70-130 mg/dl. After induction of Alloxan monohydrate, it increases up to 180-310 mg/dl which shows diabetes produced in the test rats. Alloxan induced diabetic condition in rats was successful which shows there is an increase in blood glucose level of test rats. So, this condition proved that the test rats show diabetes which is further used for animal models.
Excision Model: From the above result, it was found that EDE showed high total phenolic content and free radical scavenging activity. It also exhibits good antimicrobial effect. Therefore, EDE was selected to studying animal models to determine its effect on wound healing. Excision wound studies showed reduction in wound size.
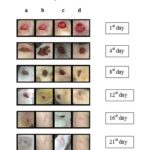
The healed area of wounds was calculated and showed in Table 8. The wound contraction was calculated and showed in % in Table 9. On 16th day, the 200 mg/kg EDE treated group showed 95% wound closure while 100 mg/kg treated showed 75% wound closure. The effect of EDE on the wound contraction is displayed photo-graphically in Fig. 2.
TABLE 8: HEALED AREA OF WOUND IN CM
| Groups | Day 1 | Day 4 | Day 8 | Day 12 | Day 16 | Day 21 |
| cm* | ||||||
| Control | 00 | 0.2 ± 0.08 | 0.5 ± 1.14 | 1 ± 0.14 | 1.4 ± 0.17 | 1.75 ± 0.10 |
| Standard | 00 | 0.3 ± 0.14 | 0.8 ± 0.14 | 1.7 ± 0.14 | 1.96 ± 0.05 | - |
| 100 mg/kg EDE | 00 | 0.1 ± 0.08 | 0.5 ± 0.08 | 1.1 ± 0.20 | 1.5 ± 0.14 | 1.83 ± 0.08 |
| 200 mg/kg EDE | 00 | 0.3 ± 0.12 | 0.7 ± 0.14 | 1.5 ± 0.14 | 1.9 ± 0.08 | - |
(*Mean ± standard deviation) n = 6
TABLE 9: RATE OF WOUND CLOSURE IN %
| Groups | Day 1 | Day 4 | Day 8 | Day 12 | Day 16 | Day 21 |
| %* | ||||||
| Control | 00 | 10 ± 4.47 | 25 ± 7.07 | 50 ± 7.07 | 70 ± 8.94 | 87.5 ± 5.24 |
| Standard | 00 | 15 ± 7.07 | 40 ± 7.07 | 85 ± 7.07 | 98.33 ± 2.58 | - |
| 100 mg/kg EDE | 00 | 5 ± 4.27 | 25 ± 4.27 | 55 ± 10.48 | 75 ± 7.07 | 91.66 ± 4.08 |
| 200 mg/kg EDE | 00 | 15 ± 6.32 | 35 ± 7.07 | 75 ± 7.07 | 95 ± 4.27 | - |
(*Mean ± standard deviation) n = 6
The increased dose of EDE extract of CB leaf showed good contraction in comparison with Povidone iodine used as reference standard. In this study, the CB leaf derived phenolic acids, tannins and flavonoids, and terpenoids. These showed the high anti-oxidant effect which enhances the healing of infected and non-infected wounds also. These effects produced by decreasing the damage caused by the oxygen radicals 39.
FIG. 2: PHOTOGRAPHS OF EFFECT OF CB ON THE WOUND CONTRACTION SHOWING HEALING OF WOUND AFTER-TREATMENT AT THE END OF STUDY (A- CONTROL, B- POVIDONE IODINE, C- 100 mg/kg EDE, D- 200 mg/kg EDE)
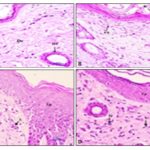
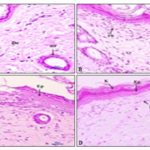
Histopathological Evaluation of Excision Model: The histopathological evaluation is very crucial while evaluating diabetic wounds. The photographs of histological changes in skin were given in Fig. 3. The skin of rats of control group did not reveal any lesion of pathological significance. Normal skin wound, in non-diabetic animal showed normal histology and skin layers such as epidermis and dermis. On the other hand, diabetic animals treated with EDE extract showed hyperplastic epidermis and lymphocytic infiltration suggesting possible inflammation. Descriptions of histopathological changes are given in Table 10. Excised skin treated with EDE extract CB showed multifocal moderate epithelial hyperplasia and lymphocytic infiltration at dermis suggesting healing of wound.
FIG. 3: HISTOLOGICAL CHANGES IN THE RAT SKIN (A- CONTROL, B- STANDARD, C- 100 mg/kg EDE, D- 200 mg/kg EDE)
TABLE 10: DESCRIPTION OF HISTO-PATHOLOGICAL ANALYSIS OF RAT SKIN
| Figure ID | Description |
| a | Normal: Skin: Showing normal histology, Epidermis (Ep), Dermis (De), Hair Follicle (HF) {H & E, 400X} |
| b | Standard: Skin: Showing newly formed epithelium (Ep), keratin deposition (K) and Fibroblasts (Fb) in dermis {H & E, 400X} |
| c | Excision- 100 mg/kg EDE: Skin: Showing hyperplastic epidermis (Ep) and lymphocytic infiltration (L) at dermis {H & E, 100X} |
| d | Excision- 200 mg/kg EDE: Skin: Showing hyperplastic epidermis (Ep) and lymphocytic infiltration (L) at dermis (D) {H & E, 100X} |
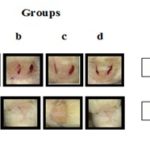
Incision Model: The tensile strength was measured and reported in Table 11. Tensile strength was found to increase in EDE treated extract of CB group as compared with control. In 200 mg /kg treated group 60.3% tensile strength was found which corresponds to histopathological study. An increase in tensile strength may be attributed to keratin deposition in the wound. The effect of EDE on the wound contraction is displayed photo-graphically in Fig. 4.
TABLE 11: EFFECT OF EDE EXTRACT OF CB ON TENSILE STRENGTH OF THE DIABETIC RATS SKIN
| Group | *% Tensile strength |
| Control | 47.5 ± 1.04 |
| Standard | 61.2 ±1.17 |
| 100 mg/kg EDE | 51.6 ± 1.9 |
| 200 mg/kg EDE | 60.3 ± 1.4 |
(*Mean ± standard deviation) n = 6
FIG. 4: PHOTOGRAPHS OF EFFECT OF CB ON THE WOUND CONTRACTION SHOWING HEALING OF WOUND AFTER-TREATMENT AT THE END OF STUDY (A- CONTROL, B- POVIDONE IODINE, C- 100 mg/kg EDE, D- 200 mg/kg EDE)
Histopathological Evaluation of Incision Model: The photographs of histological changes in skin were given in Fig. 5. The description of the histopathological analysis of rat skin was displayed in Table 12. The skin of rat of control group did not reveal any lesion of pathological significance.
Incised skin treated with 100 mg/kg EDE showed focal moderate erosion of surface epithelium, newly formed epithelium (epithelization) and fibroblasts proliferation in the dermis. In addition to this, at 200 mg/kg EDE showed keratin deposition. New formation of epithelium and keratin deposition marks healthy wound closure at 200 mg/kg dose.
FIG. 5: HISTOLOGICAL CHANGES IN THE RAT SKIN (A- CONTROL, B- STANDARD, C- 100 mg/kg EDE, D- 200 mg/kg EDE)
TABLE 12: DESCRIPTION OF HISTOP-ATHOLOGICAL ANALYSIS OF RAT SKIN
| Figure ID | Description |
| I | Normal: Skin: Showing normal histology, Epidermis (Ep), Dermis (De), Hair Follicle (HF) {H & E, 400X} |
| II | Standard: Skin: Showing newly formed epithelium (Ep), keratin deposition (K)
and Fibroblasts (Fb) in dermis {H & E, 400X} |
| III | Incision- 100 mg/kg EDE: Skin: Showing erosion of surface epithelium (Er), newly formed epithelium (Ep) and Fibroblasts (Fb) in dermis {H & E, 400X} |
| IV | Incision- 200 mg/kg EDE: Skin: Showing newly formed epithelium (Ep), keratin deposition (K) and Fibroblasts (Fb) in dermis {H & E, 400X} |
From histopathological data of incised and excised wounds at the skin of rats showed healing potential manifested by reduced erosions, epithelisation, fibroblast proliferation, epithelial hyperplasia and inflammation at wound area.
CONCLUSION: The Cipadessa baccifera showed prominent anti-oxidant and antimicrobial activity thereby justifying its use in traditional medicine. It also exhibited potential wound healing activity suggesting need of detail mechanism-based study to underline exact mechanism of action.
ACKNOWLEDGEMENT: Authors are thankful to Principal Dr. K. R. Mahadik for providing the necessary infrastructure and facilities required for carrying out research work and Dr. A. G. Namdeo for suggestions in research work.
CONFLICT OF INTEREST: NIL
REFERENCES:
- Bandi AKR and Leeb DU: Secondary metabolites of plants from the genus cipadessa: chemistry and biological activity. Chemistry & Biodiversity 2012; 9: 1403-21.
- Malarvannan S, Giridharan R, Sekar S, Prabavathy VR and Nair S: Ovicidal activity of crude extracts of few traditional plants against Helicoverpa armigera (Hubner) (Noctuidae: Lepidoptera). J of Bio 2009; 2(1): 64-71.
- Kavitha KR, Bopaiah AK and Kolar AB: Chemical composition of the essential oil from the leaves of Cipadessa baccifera (roth.) miq. International Journal of Pharmaceutical Sciences and Research 2016; 7(1): 392-96.
- Jing Ning, Ying-Tong Di, Xin Fang, Hong-Pin He, Yuan-Yuan Wang, Yan Li, Shun-Lin Li, and Xiao-Jiang Hao: Limonoids from the Leaves of Cipadessa baccifera. J Nat Prod 2010; 73: 1327-31.
- Olokoba AB, Obateru OA and Olokoba LB: Type 2 diabetes mellitus: a review of current trends. Oman Medical Journal 2012; 27(4): 269-73.
- Jeeva S and Anlin SY: A review of antidiabetic potential of ethnomedicinal plants. Medicinal & Aromatic Plants 2014; 3(4): 1-8.
- Robson MC, Steed DL and Franz MG: Wound healing: biologic features and approaches to maximize healing trajectories. Curr Probl Surg 2001; 38: 72-140.
- Hunt TK, Hopf H and Hussain Z: Physiology of wound healing. Adv Skin Wound Care 2000; 13(2 S): 6-11.
- Geethalakshmi R, Sakravarthi C, Kritika T, Arul Kirubakaran M, and Sarada DVL: Evaluation of anti-oxidant and wound healing potentials of Sphaeranthus amaranthoides f. Bio Med Res Int 2013; 1-7.
- Orsted HL, David K, Louise Forest-Lalande RN and Francoise M: Basic principles if wound healing. Wound Care Canada 2011; 9(2): 4-12.
- McKay IA and Leigh IM: Epidermal cytokines and their roles in cutaneous wound healing. British Journal of Dermatology 1991; 124(6): 513-18.
- Dhalwal K, Shinde VM, Singh B and Mahadik KR: Hypoglycemic and hypolipidemic effect of Sida rhombifolia ret USA in diabetic induced animals. International Journal of Phytomedicine 2010; 2: 160-65.
- Khandelwal KR and Sethi V: Practical pharmacognosy techniques and experiments. Nirali Publication, Pune, Edition, 2013, 1-23.
- Vincent JG and Vincent HW: Filter paper disc modification of the Oxford cuo penicillin determination. Proceeding of the Society for Experimental Biology and Medicine 1944; 55(3): 162-64.
- Tendencia EA: Disk diffusion method. In Laboratory manual of standardized methods for antimicrobial sensitivity tests for bacteria isolated from aquatic animals and environment, Tigbauan, Iloilo, Philippines: aquaculture department. Southeast Asian Fisheries Development Center 2004; 13-9.
- Holder IA and Boyce ST: Agar well diffusion assay testing of bacterial susceptibility to various anti-microbials in concentrations non-toxic for human cells in culture. Burns 1994; 20(5): 426-29.
- Obeidat M, Shtnawi M, Al-alawi M, Eans Al-Zu’Bi, Al-Dmoor H, Al-Qudah M, El-Qudah J and Otri I: Anti-microbial activity of crude extracts of some plant leaves.Research Journal of Microbiology 2012; 7(1): 59-7.
- Singleton VL and Rossi JA: Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Vitic 1965; 1: 144-58.
- Rajani P, Kotaiah R, Jayaveera KN and Chandra Sekhar KB: Evaluation of antioxidant and anticancer activities of Cipadessa baccifera. Asian Journal of Pharmaceutical and Clinical Research 2015; 8(5): 312-15.
- Murugammal S and Ilavarasan R: Phytochemical standardization of the leaves of a medicinal plant Cipadessa baccifera Roth Miq. Journal of Pharmacy Research 2016; 10(9): 609-13.
- Blois MS: Anti-oxidant determinations by the use of a stable free radical. Nature 1958; 181(4617): 1199-00.
- Gangwar M, Gautam MK, Sharma AK, Tripathi YB, Goel RK and Nath G: Anti-oxidant capacity and radical scavenging effect of polyphenol rich Mallotus philippensis fruit extract on human erythrocytes: an in-vitro The Scientific World Journal 2014; 2014: 1-12.
- Valvi SR, Rathod VS and Yesane DP: Screening of three wild edible fruits for their antioxidant potential. Current Botany 2011; 2(1): 48-2.
- Kedare SB and Singh RP: Genesis and development of DPPH method of anti-oxidant assay. J Food Sci Technol 2011; 48(4): 412-22.
- Morton JJ and Malone MH: Evaluation of vulnerary activity by open wound procedure in rats. Arch Int Pharmacodyn Ther 1972; 196(1): 117-26.
- Riazul HT, Marium B, Sohanur R, Rubaba K, Taslima B and Ahmed SU: Wound healing effect of Euphorbia hirta (Euphorbiaceae) in alloxan induced diabetic rats. BMC Complement Altern Med 2017; 17(1): 423-37.
- Lee KH: Studies on the mechanism of action of salicylates. 3. Effect of vitamin A on the wound healing retardation action of aspirin. J Pharmacol Sci 1968; 57(7): 1238-40.
- Mugade M, Patole M and Pokharkar V: Bioengineered mannan sulphate capped silver nano particles for accelerated and targeted wound healing: physicochemical and biological investigations. Biomedicine & Pharma-cotherapy 2017; 91: 95-10.
- Djeridane A, Yousfi M, Nadjemi B, Boutassouna D, Stocker P and Vidal N: Anti-oxidant activity of some Algerian medicinal plants extracts containing phenolic compounds. Food Chem 2006; 97(4): 654-60.
- Jeevan Ram A, Bhakshu LM and Venkata Raju RR: In-vitro anti-microbial activity of certain medicinal plants from Eastern Ghats, India, used for skin diseases. J Ethnopharmacol 2004; 90(2): 353-57.
- Murugan T, Albino Wins J and Murugan M: Anti-microbial activity and phytochemical constituents of leaf extracts of Cassia auriculata. Indian J Pharm Sci 2013; 75(1): 122-25.
- Bonjar GH, Nik AK and Aghighi S: Anti-bacterial and anti-fungal survey in plants used in indigenous herbal-medicine of south east regions of Iran. J Biol Sci 2004; 4(3): 405-12.
- Zablotowicz RM, Hoagland RE and Wagner SC: Effect of saponins on the growth and activity of rhizosphere bacteria. Adv Exp Med Biol 1996; 405: 83-5.
- Cowan MM: Plant products as antimicrobial agents. Clin Microbiol Rev 1999; 12(4): 564-82.
- Oyedemi BOM, Shinde VM, Shinde K, Kakalou D, Stapleton PD and Gibbons S: Novel R-plasmid conjugal transfer inhibitory and antibacterial activities of phenolic compounds from Mallotus philippensis (Lam.) Mull. Arg. Journal of Global Antimicrobial Resistanc 2016; 5: 15-21.
- Dhalwal K, Shinde VM, Namdeo AG and Mahadik KR: Anti-oxidant profile and HPTLC-densitometric analysis of umbelliferone and psoralen in Aegle marmelos. Pharmaceutical Biology 2008; 46(4): 266-72.
- Shinde V, Dhalwal K and Mahadik KR: Review on antioxidant potential ofsome important medicinal plants. Pharmacologyonline 2007; 2: 1-11.
- Cai YZ, Luo Q and Sun M Corke H: Anti-oxidant activity and phenolic compounds of 112 chinese medicinal plants associated with anti-cancer. Life Sci 2004; 74(17): 2157-84.
- Martin A: The use of anti-oxidants in healing. Dermatologic Surgery 1996; 22(2): 156-60.
How to cite this article:
Murkute AB and Shinde VM: Title: exploratory studies on diabetic wound healing potential of Cipadessa baccifera (roth.) miq. Int J Pharmacognosy 2019; 6(8): 277-86. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.6(8).277-86.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
4
277-286
795
504
English
IJP
A. B. Murkute and V. M. Shinde *
Poona College of Pharmacy, Bharati Vidyapeeth (Deemed to be University), Erandawane, Pune, Maharashtra, India.
vaibhavshinde847@gmail.com
12 August 2019
25 August 2019
27 August 2019
10.13040/IJPSR.0975-8232.IJP.6(8).277-86
31 August 2019