EVALUATION OF THE ANTIOXIDANT AND ANTI-PROLIFERATIVE CHEMICAL CONSTITUENTS OF HYPTIS PECTINATA (LINN.) AERIAL INFUSION
HTML Full TextEVALUATION OF THE ANTIOXIDANT AND ANTI-PROLIFERATIVE CHEMICAL CONSTITUENTS OF HYPTIS PECTINATA (LINN.) AERIAL INFUSION
Samuel Ehiabhi Okhale*1, Samuel Sunday Ode 1, 2, Peters Oladosu 3 and Grace Eyinehi Ugbabe 1
Department of Medicinal Plant Research and Traditional Medicine 1, Department of Microbiology and Biotechnology 3, National Institute for Pharmaceutical Research and Development, Idu Industrial Area, P.M.B. 21 Garki, Abuja, Nigeria.
Department of Chemistry 2, Benue State University, Makurdi, Nigeria.
ABSTRACT: Objective: To evaluate the chemical composition, antioxidant and antiproliferative activities of Hyptis pectinata aerial part aqueous extract (HYP). Method: Chemical profiling using high-performance liquid chromatography with UV diode array detector (HPLC-UV-DAD), in-vitro antioxidant and anti-proliferative studies were done. Antioxidant and antiradical activities of HYP was investigated using 3-(4,5-dimethylimidazole-2-yl)-2,5-diphenyltetrazolium bromide (MTT) and DPPH radical scavenging assay. Result: The present study revealed several metabolites. HPLC analysis gave caffeic acid (13.92%), rutin (7.89%) and ferulic acid (5.44%) as some of the bioactive constituents. HYP showed antioxidant activity in both the DPPH and MTT assay. HYP exhibited antioxidant activity against DPPH and MTT from the first dilution at 10 mg/ml up to the ninth dilution at 0.08 mg/ml in a 2-fold dilution signifying the presence of antioxidant compounds at those concentrations. At 1 mg/ml to 32 mg/ml, HYP dose-dependently and significantly (P < 0.001) inhibited Sorghum bicolor seed radicle growth throughout 24-72 h compared to the negative control. Conclusion: Hyptis pectinata aerial part aqueous extract (HYP) possessed antioxidant and antiproliferative activities, hence providing preliminary evidence for its use to treat cancer.
| Keywords: |
Hyptis pectinata, Caffeic acid, Antioxidant, Anti-proliferative
INTRODUCTION: Medicinal plant has been discovered to play an important role in health care. Towards the end of the twentieth century, the World Health Organization (WHO) estimated that an impressive 80 % of the world’s population relies on natural medicines, with plant-derived medicines as the main component 1. Cancer generally refers to a group of diseases that cause cells in the body to change and grow out of control.
Incidence of cancer is on the increase worldwide, with an estimated 14.1 million new cancer cases in 2012. A recent report shows that cancer has claimed over nine million lives in 2015 2. Cancer is a complex multifactorial cell disease characterized by abnormal cellular proliferation 3.
To develop new anticancer drugs, different plants are being screened based on their ethnomedicinal uses. Many diseases such as cancer, diabetes, arteriosclerosis, inflammatory disease, autoimmunity, cardiovascular disease and Alzheimer’s have been associated with an increase of reactive oxygen species (ROS). Antioxidants are important substances that can protect the organism from the damage caused by oxidative stress.
As a result, there is a keen interest in the use of natural antioxidants from medicinal plants that may help an organism to keep the normal balance of ROS 4. The Lamiaceae family comprises of 250 genus and approximately 6970 species. The members of the family are spread all over the world, especially in the tropic and subtropical regions. The family grows abundantly in the Mediterranean areas, where it is possible to find the vast majority of the genus species. Many species of the Lamiaceae family are used in food due to the flavor and taste. Besides that, the members of the family are a good source of aromatic essential oils as well as ornamental plants 5.
Hyptis pectinata belongs to the family Lamiaceae. It is a perennial, erect aromatic herb to small shrub up to 4 m high with distinct, 4-angled stems. Leaves are opposite, petiolate, the blade ovate to nearly triangular with serrate margins, pubescent, 1.5 to 7 cm long, 1 to 2.5 cm wide. Flowers small, strongly zygomorphic, white or pink-tinged, borne in dense cyme-like clusters arising from spike-like inflorescences 30-60 cm long. The fruit is nutlet 6. Stems woody at the base, much branched, pubescent, 4-angled.
In Taiwan, it occurs at lowlands in the eastern part. The seeds are oval, dark brown to blac. It is seen along roadsides or waste places and associated with other obnoxious weeds such as Conyza Canadensis (L.) Cronq.var. canadensis, Biden spilosa L. var. radiate Schultz-Bip., Ipomoea triloba L., Chloris barbata Sw. and Chamae sycehirta (L.) Mill. This herb can be distinguished from its similar species, Hyptis suaveolens (Linn.) Poit, by its comb-shaped cymes, pink or whitish violet flowers and smaller calyx with white trichomes between bristly calyx-lobes 7.
Hyptis pectinata L. Poit, known as ‘sambacaitá’ in Brazil is used to treat inflammatory and painful disorders. The leaf has been reported to possess antioxidant properties 8. Hyptis pectinata (L.) Poit is a widely distributed species in Nigeria, with local names as “Kimbar awaaki” (Hausa), ”Jogbo or Alatoriyo” (Yoruba) and ”Idumuje” (Igbo). The leaves are used as a tea, for the treatment of inflammatory conditions, malaria, infections, and cancer. The leaves and bark are used as an infusion for the treatment of throat and skin inflammations, bacterial infections, pain, fungal infections and cancer. The leaves of the plant are edible which can be served as food 9, 10, 11. Hyptis species are aromatic plants with the presence of biologically active substances such as antimicrobial, antifungal, anti-HIV, analgesic, anti-inflammatory activities, anti-oxidant activity, molluscicidal, hemostatic, anti-ulcer, cytotoxicity and insecticide properties 12-23.
Anti-oxidants are important due to their ability to scavenge free radicals such as reactive oxygen species (ROS), which are known to cause damage to cells at the molecular level. Oxidative stress is implicated in various diseases related to degenerative disorders, arthritis, diabetes, cancer, and immune system decline. Antioxidant compounds play a crucial role in the treatment of these diseases by acting to reduce oxidative stress and thus decreasing the extent of oxidative damage. There is a global demand for antioxidants from natural sources such as plants 24.
This study aims to evaluate the phytochemical, antioxidant and anti-proliferative properties of the aerial part aqueous extract of Hyptis pectinata (L.) growing in Northern Nigeria.
MATERIALS AND METHODS:
Chemicals and Reagents: Unless otherwise stated, all chemicals and reagents were of analytical grade and purchased from Sigma Aldrich (Germany). All the solvents for the chromatographic purpose were HPLC grade, purchased from Sigma Germany.
Collection of Plant Material: The plant material was collected by Mr. Muazzam Ibrahim from Suleja, Niger State and identified and authenticated by Mr. Lateef of NIPRD Abuja.
Plant Preparation and Extraction: The plant sample was powdered using mortar and pestle. Hot water extract of Hyptis pectinata was prepared as described 25, with some modifications. Briefly, 100 g of powdered dried plant material was stirred into 2 Litres of boiling water. This is more or less the equivalent of a teaspoon full in a cup of boiling water. It was decided on this method of preparation as it is mostly used as an infusion by traditional healers. This mixture was left to cool and stand overnight at room temperature, after which it was filtered and concentrated to dryness. This method gave a yield of 3.25 g (3.25 % w/v). The dried extracts were used immediately for analysis.
Experimental Plant: The experimental plant, Sorghum bicolor (Guinea corn) seeds were purchased from Dumez small market, Niger State and identified and authenticated by Mr. Muazzam Ibrahim of NIPRD Abuja. The seed viability test was performed by placing the seeds inside a beaker containing water. The seeds that floated were discarded while the submerged seeds were cleansed with methylated spirit, dried and used for the study.
Phytochemical Analysis: The aqueous extract of Hyptis pectinata aerial part was analyzed for the presence of alkaloids, anthraquinones, tannins, phlobatannins, saponins, flavonoids, terpenes, steroids and cardiac glycosides using previously described methods 26.
High Performance Liquid Chromatography (HPLC) Analysis: The chromatographic system includes Shimadzu HPLC system consisting of Ultra- Fast LC-20AB prominence, equipped with SIL- 20AC autosampler; DGU-20A3 degasser; SPD-M20A UV diode array detector (UV-DAD); column oven CTO-20AC, system controller CBM- 20 Alite and Windows LC solution software (Shimadzu Corporation, Kyoto Japan); column, VP-ODS 5µm and dimensions (150 x 4.6 mm). The chromatographic conditions included mobile phase solvent A: 0.2% v/v formic acid and solvent B: acetonitrile; mode: isocratic; flow rate 0.6 ml/min; injection volume 20µl of 20 mg/ml solution of HYP in water; detection was at UV 254 nm wavelength. Reference standards, rutin, quercetin, caffeic acid, ferulic acid and apigenin (Fluka, Germany) 50 µg/ml in methanol were analyzed separately under the same condition as the extract (HYP). The HPLC operating conditions were programmed to give the following: solvent B: 20% and column oven temperature 40°C. The total run time was 40 minutes 2.
Antioxidant Assay: The method of Muraina et al., 2009 27 was used with some modifications. To evaluate the DPPH radical scavenging activity of the extract, ten microlitres of 10 mg/ml of HYP in methanol was spotted on TLC plate and eluted in a polar solvent comprising the mixture of ethyl acetate and methanol in the ratio of 3:2.
The developed plate was dried and immediately sprayed with 0.05 % DPPH reagent in methanol and left at room temperature for 30 min. The DPPH test is based on the ability of the extracts to donate radical hydrogen to scavenge the stable DPPH radical. When this radical reacts with the anti-oxidant compound, it is reduced with the loss of the deep violet color to light yellow. Anti-oxidant compounds of HYP bleached the purple color of DPPH reagent, thereby appeared as yellow spots Fig. 2. The yellow spots were evaluated as positive antioxidant activity 28.
A two-fold dilution of 20 mg/ml of an extract with 50µl of distilled water in a 96-well micro-dilution plate was performed. After that, 50µl of 0.2 mg/ml of 3-(4,5-dimethylimidazole-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) was added to every well, and the plate was incubated at 37 ºC after which the result was read. Distilled water was used as negative control and ascorbic acid was used as standard drug. Formation of bluish coloration or precipitate was indicative of antioxidant activity. The lowest concentration of the sample at which the presence of antioxidant activity is detected was recorded as the minimum inhibitory concentration (MIC). The experiment was done in triplicate.
Determination of Growth Inhibitory Effects: The modified methods of Ayinde et al., 29 and Chinedu et al., 30 were used for this study. HYP (3.2 g) was dissolved in 100 ml of distilled water to obtained 32 mg/ml stock solution. Various concentrations (1 mg/ml, 2 mg/ml, 4 mg/ml, 8 mg/ml, 16 mg/ml and 32 mg/ml) of HYP were prepared. Methotrexate used as a standard drug at a concentration of 0.05 mg/ml. Petri dishes were layered with cotton wool and filter paper (Whatman no. 1). The filter paper was divided into 24 sections in a dart fashion.
Twenty four (24) seeds of S. bicolor were placed in a separate sample bottle for pre-treatment. The control seeds were treated with 15 ml of distilled water. The test seeds were treated with the different concentrations of HYP as the seeds in each specific bottle received 15 ml of a particular concentration. Duration of pre-treatment in the sample bottle was 24 h. The seeds were then arranged in the specified Petri dish, incubated in a dark cupboard at ambient temperature and observed for growth after 24 h. The mean lengths (mm) of radicle emerging from the seeds were measured after 24, 48 and 72 h. The percentage growth inhibition was calculated as [(mean radicle length control - mean radicle length treated)/mean radicle length control] × 100. Percentage growth was calculated as 100 ‒ % inhibition. The experiment was performed in duplicate.
Statistical Analysis: The data obtained were expressed as mean ± standard error mean and analyzed using Graph pad prism (version 7). Two-way analysis of variance (ANOVA) was used (P<0.001).
RESULTS AND DISCUSSION: Hyptis pectinata is widely used in tropical countries due to its flavor and medicinal properties. The results Table 1 demonstrate that H. pectinata possessed significant cytotoxic and growth inhibitory activities at the concentrations investigated compared to water as a negative control (p<0.0001).
Phytochemical Analysis: Qualitative analysis of the aqueous extracts of Hyptis pectinata indicated the presence of phytoconstituents like saponins, tannins, flavonoids, terpenes, sterols, and essential oils. Previous studies reported that flavonoids, saponins, terpenes, sterols, and essential oil were the major phytochemical compounds present in Hyptis pectinata 31.
FIG. 1: HPLC SPECTRUM OF AERIAL PART AQUEOUS EXTRACT OF HYPTIS PECTINATA (HYP). Sixteen peaks were detected from the HPLC spectrum. Caffeic acid (13.92%), rutin (7.98%) and ferulic acid (5.44%) appeared at retention times of 4.965, 6.822 and 7.830 minutes respectively.
High Performance Liquid Chromatography (HPLC) Analysis: From the HPLC spectrum Fig. 1 a total of sixteen peaks were detected with retention times of 2.775, 3.089, 3.359, 3.599, 4.139, 4.965, 5.561, 6.822, 7.830, 8.367, 8.996, 10.938, 12.538, 13.376, 17.459 and 25.990 minutes. Caffeic acid (13.92%), rutin (7.98%) and ferulic acid (5.44%) appeared at retention times of 4.965, 6.822, 7.830 and 25.990 minutes respectively. Percentage composition of the constituent was calculated based on the area under the curve (AUC). Detection was at UV 254 nm.
Antioxidant Activity: The aqueous extract of Hyptis pectinata aerial part (HYP) gave antioxidant activity with both DPPH and MTT. The antioxidant activity of HYP was performed employing 3-(4,5-dimethyl thiazole-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) and 96-well microdilution technique. Concentration of the extract ranged from 10 mg/ml to 0.01 mg/ml and the concentration of the reference ascorbic acid ranged from 0.1 mg/ml to 0.0001 mg/ml. The bluish coloration formed due to the antioxidant activity of HYP was observed from the first dilution at 10 mg/ml up to the ninth dilution at 0.08 mg/ml in 2-fold dilutions signifying the presence of antioxidant compounds at those concentrations. The ascorbic acid was active from 0.1 mg/ml to 0.013 mg/ml. Therefore, the anti-oxidant properties of H. pectinata could play a useful role in food preservation and also in the prevention of oxidative damage associated with many diseases. The caffeic acid, rutin, and ferulic acid content may have contributed to the observed antioxidant activity of H. pectinata since caffeic acid, rutin and ferulic acid have been reported to possess antioxidant property 32-35.
FIG. 2: THIN LAYER CHROMATOGRAM OF AERIAL PART AQUEOUS EXTRACT OF HYPTIS PECTINATA (HYP) ON SPRAYING WITH 0.05% DPPH. 3 is gallic acid, 4 is betulinic acid, 5 is HYP dissolved in methanol, 6 is caffeic acid dissolved in ethanol, 7 is quercetin, 8 is ferulic acid dissolved in methanol, 9 is rutin. DPPH gave the purple colored background of the TLC; the light-yellow spots bleached the DPPH as evidence of their antioxidant action.
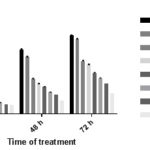
Growth Inhibitory Effects of Hyptis pectinata on Sorghum bicolor Seeds: As a preliminary anticancer screening, the radicle lengths of fast-growing seeds such as Sorghum bicolor has been utilized for the testing of suspected anticancer agents. Generally, cancer cells have a characteristic of fast proliferation, and this is also the case with meristematic cells of Sorghum bicolor seeds when exposed to favorable conditions 2.
That was the justification for the use of the method for this study. As shown in Fig. 2, there was a dose-related reduction in the length of the radicle of Sorghum bicolor seeds treated with the various concentration of H. pectinata extract compared to the distilled water used as control at 24 - 72 h. Rapid and progressive growth was observed in the water control seed radicle lengths.
FIG. 3: INHIBITORY EFFECTS OF HYPTIS PECTINATA ARIAL AQUEOUS EXTRACT (HYP) ON THE GROWTH OF SORGHUM BICOLOR SEED RADICLE
TABLE 1: MEAN RADICAL LENGTH, PERCENTAGE INHIBITION AND PERCENTAGE GROWTH FOR SORGHUM BICOLOR SEEDS TREATED HYPTIS PECTINATA ARIAL AQUEOUS EXTRACT HYP
| Treatment | Mean radicle length (mm) | Percentage inhibition* | Percentage growth† | ||||||
| 24 h | 48 h | 72 h | 24 h | 48 h | 72 h | 24 h | 48 h | 72 h | |
| Control (H2O) | 19 ± 19 | 32.05 ± 0.45 | 39.2 ± 0.23 | 0 | 0 | 0 | 100 | 100 | 100 |
| Methotrexate | 4.45 ± 0.21 | 6.75 ± 0.15 | 10.08 ± 0.09 | 76.58 | 78.94 | 74.29 | 23.42 | 21.06 | 25.71 |
| HYP (1 mg/ml) | 17.3 ± 0.28 | 28.48 ± 0.15 | 37.43 ± 0.19 | 8.95 | 11.14 | 4.52 | 91.05 | 88.86 | 95.48 |
| HYP (2 mg/ml) | 13 ± 0.17 | 17.45 ± 0.27 | 26.33 ± 0.39 | 31.58 | 45.55 | 32.83 | 68.42 | 54.45 | 67.17 |
| HYP (4 mg/ml) | 10.3 ± 0.17 | 15.05 ± 0.11 | 24.55 ± 0.18 | 45.79 | 53.04 | 37.37 | 54.21 | 46.96 | 62.63 |
| HYP (8 mg/ml) | 8.68 ± 0.09 | 13.48 ± 0.16 | 20.45 ± 0.21 | 54.32 | 57.94 | 47.83 | 45.68 | 42.06 | 52.17 |
| HYP (16 mg/ml) | 5.5 ± 0.19 | 10.78 ± 0.13 | 17.85 ± 0.13 | 71.05 | 66.37 | 54.46 | 28.95 | 33.63 | 45.54 |
| HYP (32 mg/ml) | 4.53 ± 0.17 | 9.6 ± 0.15 | 10.08 ± 0.20 | 76.16 | 70.05 | 74.29 | 23.84 | 29.95 | 25.71 |
*Percentage inhibition was calculated as [(mean radicle length control-mean radicle length treated)/mean radicle length control] x 100. †Percentage growth was calculated as 100 - % inhibition. Methotrexate (0.05 mg/ml) was used as positive control; n=20.
****HYP significantly (P<0.0001) inhibited S. bicolor seed growth at 24 h, 48h and 72h for all concentrations studied compared with the negative control (distilled water). Mean radicle length (mm) is presented as the mean ± standard error of the mean.
At 72 h, the mean radicle lengths of the control seeds was 39.2 ± 0.23 mm while the mean radicle length of the seeds treated with 1 mg/ml, 2 mg/ml, 4 mg/ml, 8 mg/ml, 16 mg/ml and 32 mg/ml were 37.43 ± 0.19, 26.33 ± 0.39, 24.55 ± 0.18, 20.45 ± 0.21, 17.85 ± 0.13, 10.08 ± 0.20, corresponding to percentage inhibitions of 4.52%, 32.83 %, 37.37%, 47.83%, 54.46%, 74.29% respectively and the positive control methotrexate gave 74.29% growth inhibition at 0.05 mg/ml Table 1.
This is indicative of the cytotoxicity of Hyptis pectinata aerial part hot water infusion which is in agreement with previous studies 36. Therefore, the growth inhibitory effect of HYP was concentration-dependent.
CONCLUSION: The hot water extract of Hyptis pectinata (HYP) exhibited antioxidant activity and growth inhibitory effects. Hence, by extension, it can inhibit cancerous cells. The bioactive chemical constituents of HYP detected by high-performance liquid chromatography were caffeic acid, ferulic acid, and rutin. This study provided preliminary evidence that supports the ethnomedicinal use of Hyptis pectinata aerial part as tea in the treatment of malaria and cancer.
ACKNOWLEDGEMENT: The authors wish to thank Department of Medicinal Plant Research and Traditional Medicine, National Institute for Pharmaceutical Research and Development, Idu Industrial Area, Abuja, Nigeria, for providing technical support for this study.
CONFLICT OF INTEREST: We declare that we have no conflict of interest.
REFERENCES:
- Serafini MR, Vergne DMC and Rabelo TK: Determination of chemical and physical properties of Hyptis pectinata essential oil and their redox active profile. Journal of Bio-technology and Pharmaceutical Research 2012; 3(1): 1-9.
- Okhale SE, Tijani AY and Ezugwu BO: Assessment of anti-proliferative potential of Hexalobus crispiflorus (Annonaceae). British Biotechnology Journal 2016; 15(3): 1-7.
- Abu-rish EY, Kasabri VN, Hudaib MM and Mashalla SH: Evaluation of anti-proliferative activity of some traditional anticancer herbal remedies from Jordan. Tropical Journals of Pharmaceutical Research 2016; 15(3): 469-474.
- Nascimento AKL, Melo-Silveira RF and Dantas-Santos N: Antioxidant and anti-proliferative activities of leaf extracts from Plukenetia volubilis Linneo (Euphorbiaceae). Evidence-Based Complementary and Alternative Medicine 2013; 2013: 1-10.
- Barbosa LCA, Martins FT, Teixeira RR, Polo M and Montanari RM: Chemical variability and biological activities of volatile oils from Hyptis suaveolens (L.) Poit. Agriculturae Conspectus Scientificus 2013; 78(1): 1-10.
- WHO: Medicinal plants in the south Pacific. WHO Regional Publications, Western Pacific Series No. 19, WHO Regional Office for the Western Pacific, Manila,
- Chen SH and Wu MJ: Notes on three newly naturalized plants in Taiwan. Taiwania 2005; 50(1): 29-39.
- Paixão MS, Melo MS, Oliveira MG, Santana MT, Lima AC, Damascena NP, Dias AS, Araujo BS, Estevam CS, Botelho MA and Quintans LJ Jr: Hyptis pectinata: redox protection and orofacial antinociception. Phytother Res 2013; 27(9): 1328-33.
- Falcao RA, do Nascimento PLA and de Souza SA: Anti-leishmanial phenylpropanoids from the leaves of Hyptis pectinata (L.) Poit. Evidence-based complementary and alternative Medicine 2013; 2013: 1-7.
- Stanfo KC, Petek M, Grdisa M, Pintar J, Bedekovic D, Custic MH and Satovic Z: Medicinal Plants of the family Lamiacaea as functional foods. Czech Journal Food Science 2016; 34(5): 377-390.
- Patricia SO, Costa MJC and Alves JAB: Chemical composition and antimicrobial activity of the essential oil of pectinata. Quimica Nova 2008; 31(7): 1648-1652.
- Raymundo LJRP, Guilhon CC, Alviano DS, Matheus ME, Antoniolli AR, Socrates CH, Cavalcanti, Alves PB, Alviano CS and Patricia: Characterization of the anti-inflammatory and anti-nociceptive activities of the Hyptis pectinata (L) Poit. Journal of Ethnopharmacology 2011; 134(3): 725-732.
- Fidyt K, Fiedorowiczm A, Strzadala L and Szummy A: β-caryophyllene and β-caryophyllene oxide Natural compounds of anticancer and analgesic properties. Cancer Med 2016; 5(10): 3007-3017.
- Hossan MDS, Nahian A, Jahan R and Rahmatullah M: Rosmarinic acid: A review of its anti-cancer action. World Journal of Pharmacy and Pharmaceutical Sciences 2014; 3(9): 57-70.
- de Olivereira TM, de carvalho RBF, de Costa IHF, de Oliveira GAL, de souza AA and de Lima SG: Evaluation of p-cymene, a natural antioxidant 2014; 53(3): 423-428.
- Nascimento PFC, Alviano WS and Santo NP: Hyptis pectinata essential oil: chemical composition and anti-streptococcus mutans activity. Wiley Online Library 2008; 24(6): 485-489.
- Melo GB, silva RL and Melo VA: Enhancement of liver regeneration by the association of Hyptis pectinata with laser therapy. Phytomedicine 2005; 50 (5): 949-954.
- Bispo MD, Mourão RHV and Franzotti EM: Anti-nociceptive and anti-edematogenic effects of the aqueous extract of Hyptis pectinata leaves in experimental animals. Journal Ethnopharmacol 2001; 76(1): 81-86.
- Malan K, Dusart G, Marion C, loukou Y, Simeon de Bouchberg M, Atisso M and Pelissier Y: Anti-bacterial activity of the essential oil of Hyptis pectinata. plantes Medicinales et phytotherapie 1986; 20(4): 323-329.
- Adriano A, Alves FJ, Kaline WA, de sa Gilberto F and Severino J: Investigating the potential of metal-organic framework material as an adsorbent for matrix solid-phase dispersion extraction of pesticides during an analysis of dehydrated Hyptis pectinata medicinal plant by GC/MS. Journal of AOAC International 2012; 95(5): 1338-1342.
- sarmento-Neto JF, do Nascimento LG, Felipe CFB and de Souse DP: Analgesic potential of essential oils. Molecules 2016; 21(20): 2-29.
- Barbosa CV, Aquino PGV, Ribeiro-Júnior: Cytotoxic and antitumor activities of Hyptis pectinata (Sambacaitá) extract. Pharmacology Online 2012; 3: 70-74.
- Pereda-Miranda R, Hernandez L, Villavicencico MJ, Novelo M Iberra P, Chai H and Pezzuto JM: Structure and stereochemistry of pectinolides A-C, novel antimicrobial and cytotoxic 5,6-dihydro-alpha-pyrones from Hyptis pectinata. Jou of Natural Products 1993; 56(4): 583-93.
- Sepahvand R, Delfan B, Ghanbarzadeh S, Rashidipour M, Veiskarami GH and Ghasemian-Yadegari J: Chemical composition, antioxidant activity and antibacterial effect of essential oil of the aerial parts of Salvia sclareoides. Asian Pac J Trop Med 2014; 7(S-1): S491-S496.
- Fernandes AC, Cromarty A, Albrecht C, Constance E and Rensburg JV: The antioxidant potential of Sutherlandia frutescens. Ethnopharmacol. 2004; 95: 1-5.
- Sofowora A: Medicinal plants and traditional medicine in Africa, Spectrum Books Limited, Ibadan, Nigeria, edition 3rd, 2008; 199-202.
- Muraina IA, Suleiman MM and Eloff JN: Can MTT be used to quantify the antioxidant activity of plant extracts? Phytomedicine 2009; 16(2009): 665-668.
- Tepe DD, Atalay S, Munevver S and Moschos P: Anti-microbial and antioxidant activities of the essential oil and various extracts of Salvia tomentosa Miller (Lamiaceae). Food Chemistry 2005; 90: 333-340.
- Ayinde BA and Agbakwuru U: Cytotoxic and growth inhibitory effects of the methanol extract Struchium sparganophora Ktze (Asteraceae) leaves. Pharmacognosy Magazine 2010; 6(24): 293-297.
- Chinedu E, Arome D, Ameh SF and Ameh GE: Evaluation of the anti-proliferative and cytostatic effect of Citrus sinensis (orange) fruit juice. Int J App Basic Med Res 2014; 4(3): 20-22.
- Fakhroo A and Sreerama L: Qualitative analysis of phytochemical compounds In Ocimum basilicum grown i.n Qatar. International Journal of Applied Pharmaceutical and Biological Research 2016; 1(4): 1-7.
- João LAS, Vanderson SB and Adriano BCF: Evaluation of chemical constituents and antioxidant activity of coconut water (Cocus nucifera ) and caffeic acid in cell culture. Annals of the Brazilian Academy of Sciences 2013; 85(4): 1235-46.
- Skowron MJ and Grześkowiak AZ: Analysis of anti-oxidant activity, chlorogenic acid, and rutin content of sinensis infusions using response surface methodology optimization. Food Anal Methods 2014; 7: 2033-41.
- Kikuzaki H, Hisamoto M, Hirose K, Akiyama K and Taniguchi H: Anti-oxidant properties of ferulic acid and its related compounds. J Agric Food Chem 2002; 50(7): 2161-8.
- Wang Y, Zhang Y, Sun B, Tong Q and Ren L: Rutin protects against pirarubicin-induced cardiotoxicity through TGF-β1-p38 MAPK Signaling Pathway. Evidence-Based Complementary and Alternative Medicine 2017: 1-10.
- Suzery and Cahyono B: Evaluation of the cytotoxicity effect of Hyptis pectinata Poit (Lamiaceae) extracts using BSLT and MTT methods. J Sains dan Matematika 2014; 22(3): 84-88.
How to cite this article:
Okhale SE, Ode SS, Oladosu P and Ugbabe GE: Evaluation of the antioxidant and anti-proliferative chemical constituents of Hyptis pectinata (Linn.) aerial infusion. Int J Pharmacognosy 2018; 5(1): 8-14. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.5(1).8-14.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
2
8-14
647
925
English
IJP
S. E. Okhale *, S. S. Ode, P. Oladosu and G. E. Ugbabe
Department of Medicinal Plant Research and Traditional Medicine, National Institute for Pharmaceutical Research and Development, Abuja, Nigeria.
samuelokhale@gmail.com
13 May 2017
22 August 2017
16 September 2017
10.13040/IJPSR.0975-8232.IJP.5(1).8-14
01 January 2018