EFFECTS OF AQUEOUS METHANOLIC LEAF EXTRACTS OF INDIGENOUS PLANTS CLERODENDRUM COLEBROOKIANUM WALP AND CENTELLA ASIATICA LINN. ON CARBON TETRACHLORIDE INDUCED CARDIOTOXIC WISTER ALBINO RATS
HTML Full TextEFFECTS OF AQUEOUS METHANOLIC LEAF EXTRACTS OF INDIGENOUS PLANTS CLERODENDRUM COLEBROOKIANUM WALP AND CENTELLA ASIATICA LINN. ON CARBON TETRACHLORIDE INDUCED CARDIOTOXIC WISTER ALBINO RATS
Ratna Jyoti Das * and Bhaskar Mazumder
Department of Pharmaceutical Sciences, Dibrugarh University, Dibrugarh - 786004, Assam, India.
ABSTRACT: Two indigenous plants Clerodendrum colebrookianum walp and Centella asiatica Linn. were selected to determine its cardioprotective activity on male Wister albino rats. Empirical research was designed to evaluate the cardioprotective activity of the leaves of both with special reference to its putative protective role. Aqueous methanolic extracts of both the plants were prepared by continuous hot percolation method. Male albino rats of Wistar strain (250-300 g) were used for the study. Animals were divided into 10 groups of six animals each. Both pretreatment and post-treatment study was done for 16 days at two different doses of 300 mg/kg body weight and 700 mg/kg body weight, respectively. Carbon tetrachloride was used as a cardiotoxic agent. The in-vivo biological studies on serum and tissues of male Wister rats at the doses of 300 mg and 700 mg/kg body weight respectively was carried out taking silymarin as standard. The methanolic extract of both improved the cholesterol level along with significant improvement of SGPT (Serum glutamate pyruvate transaminase), SGOT (Serum glutamate oxaloacetate transaminase), ALP (Alkaline phosphatase) and total protein in respect to silymarin group. The test extract at higher dose was found to significantly reverse the elevated marker enzymes, i.e. SGOT, SGPT, ALP indicating its cardioprotective role. The higher dose extracts were also found to have a pronounced effect on oxidative stress parameters such as GSH and catalase on CCl4 induced rats. Followed by heart tissue separation and homogenization was done in KCl (10 mM) phosphate buffer (1.15%) with ethylene-diamine-tetraacetic acid (EDTA: pH 7.4) and centrifuged at 12000 rpm for 20 min. The supernatant was used for the measurement of malondialdehyde (MDA). Lipid peroxidation was estimated in terms of thiobarbituric acid reactive species (TBARS) using MDA as standard by the method of Buege and Aust 1978. The amount of MDA was calculated using a molar extinction coefficient of 1.56 × 105 M-1 cm-1 and expressed as nmoles of MDA formed per mg wet weight of tissue. For both pretreatment study and post-treatment study MDA concentration was maximum for the toxic control group and minimum for the control group. For the extracts of C. colebrookianum and C. asiatica at both the dose level 300 mg/kg body weight and 700mg/kg body weight is showing significant (p< 0.01) reduction in MDA production. For C. colebrookianum MDA production is less on a higher dose, and for C. asiatica at a low dose, only MDA production is less. It signifies both the plant extracts are having potent antioxidant activities, and it is evidenced from previous literature also.
| Keywords: |
Lipid peroxidation, Cardiotoxicity, Malondialdehyde, TBARS
INTRODUCTION: Medicinal herbs have a relevant and crucial role to play towards attaining the goal of proper human healthcare.
The effective phyto-components obtained from plants are generally regarded as safe (GRAS) and are environment-friendly. The components used are believed to have improved compatibility with human systems. Though effective, herbal medicines are not scientifically exploited; therefore, this domain needs proper study in the light of modern science 1. Carbon tetrachloride (CCl4), a clear, colorless, volatile, heavy and non-flammable liquid, is a well-known model compound for producing chemical tissue toxicity by the generation of free radicals in many tissues 2 such as liver, kidneys, heart, lung, testis, brain and blood 3, 4.
It is biotransformed by hepatic microsomal cytochrome P450 to trichloromethyl-free radical (CCl3* or CCl3OO*) 5, 6, 7, 8 which in turn, initiate lipid peroxidation process 9, 10, 11. The most widely accepted mechanism of CCl4 induced cardiotoxicity is the formation of free radicals, which is a rate-limiting process in tissue peroxidative damage 12. This free radical and related reactive species may cause oxidative stress, which produces major interrelated rearrangements of cellular metabolism, increase in intracellular free calcium, damage to membrane ion transport and permeability, and destruction of the cells by lipid peroxidation 13. The accumulating of lipid peroxides introduces hydrophilic moieties hydrophobic phase and thus alter membrane permeability and cell function. This leads to loss of myocardial structural integrity and depressed cardiac function resulting in cardiotoxicity and congestive cardiac failure 12.
Cardiotoxicity is the consequence of dysfunction of heart electrophysiology or muscle damage. Acute myocardial infarction (MI) is the most important result of cardiotoxicity as the necrosis of a particular area of myocardium occurred by any disturbance in the blood supply to the heart. It may usually be as a result of blockage of a coronary artery. Reactive oxygen species may play a vital role in the pathogenesis of myocardium. Natural antioxidants said to have less toxicity and proved to be effective and safer, and they can scavenge the free radicals and avoid excess ROS formation in the body thereby helping in mitigating cardiac diseases and several other disorders. TBARS signifies the level of lipid peroxidation as a consequence of increased ROS generation.
As a result of cardiotoxicity, the sudden generation of ROS (Reactive oxygen species) can dramatically upset the balance with an increased demand on the antioxidant defense system. Malondialdehyde, a lipid peroxidation end product in tissue homogenate, was measured according to the method described by Wills 1969 with some modifications 14.
Aim of the study was to determine lipid peroxidation end product on carbon tetrachloride-induced cardiotoxic Wister albino rats using the leaf extracts of the plants Clerodendrum colebrookianum walp and Centella asiatica Linn. by determining the amount of MDA which is calculated using a molar extinction coefficient of 1.56 ×105 M-1 cm-1 and expressed as nmoles of MDA formed per mg wet weight of tissue.
MATERIALS AND METHODS:
Drugs and Chemicals: Sodium chloride was purchased from Qualigens. Aluminum chloride and Folin-Ciocalteau reagent were purchased from Merck. Gallic acids, quercetin, DPPH, Ascorbic acids, carbon tetrachloride, were procured from Himedia. Sodium nitroprusside was procured from Loba Chemie. Hydrogen peroxide and thiobarbituric acids were procured from Sigma Aldrich. All other reagents and solvents used were of analytical grade and used as procured without further purification.
Collection and Authentication of Plant Material: The leaves of Centella asiatica Linn. and Clerodendrum colebrookianum Walp was collected from Sivasagar district of Assam, India during June - July, and January -February.
The leaves were washed thoroughly followed by shade drying and were preserved as herbarium sheet. The authentication was carried out in Botanical Survey of India (BSI), Eastern Regional Centre, Shillong, authentication number being DU/PSC/HRB/RJD/1/2016 and DU/PSC/HRB/ RJD/2/2016 respectively.
Preparation of Plant Extract: The leaves were cleaned and cut into small pieces allowed for shed dry. The dried leaves were grinded and passed through mesh 40 and stored in an airtight container for further studies. The powdered shade dried leaves were packed in Soxhlet extractor for extraction with methanol.
The extract was concentrated by distilling off the solvent and evaporated to dryness in a rotary vacuum evaporator (BUCHI, Switzerland) followed by lyophilization (IIC, India) into a dried mass by lyophilizer. Further, the methanolic extract of the leaves of C. asiatica was subjected to fractionation using dichloromethane (DCM) to remove chlorophyll like substances.
Cardioprotective Effect of the Plant Extracts against Carbon Tetrachloride-Induced Cardiotoxicity in Male Wistar Rats: Male albino rats of Wistar strain (250-300 g) were used for the study. The animals housed in polypropylene cages in a temperature-controlled room (22 ± 2 °C) with relative humidity (44-55%) under 12/12 h light and dark cycles for one week before and during the experiments. Animals were provided with a standard rodent pellet diet and clean drinking water ad libitum. The study has got the clearance from the Institutional Animal Ethical Committee (IAEC) the Committee for Control and Supervision of Experiments on Animals (CPCSEA).
Experimental Design: Animals were divided into 10 groups of six animals each.
Group, I served as normal control and received drinking water for 16 days orally and on the 16-day olive oil (1ml/kg i.p.). Group II served as a toxic control and received drinking water for 16 days orally and on the 16th CCl4 (1ml/kg i.p.) in 1:1 dilution with olive oil. Group III and IV served as pre-treatment groups (prophylactic). They received CCE (Clerodendrum colebrookianum Walp. Extract) at the dose of 300 and 700 mg/kg, orally for 16 days respectively, and on the 16th day received CCl4 (1 ml/kg i.p.) in 1:1 dilution with olive oil, 2 h after administration of the last dose of extract. Group V and VI served as pre-treatment groups (prophylactic). They received CAE (C. asiatica Linn. extract) at the dose of 300 and 700 mg/kg, orally for 16 days respectively, and on the 16th day received CCl4 (1 ml/kg i.p.) in 1:1 dilution with olive oil, 2 h after administration of the last dose of extract.
Group VII and VIII served as post-treatment groups (curative). They received drinking water orally for 16 days and on the 16th day they received CCl4 (1ml/kg i.p.) in a 1:1 dilution with olive oil, followed by plant extract at a dose of 300 mg/kg (CCE) and 700 mg/kg (CCE) orally at 2, 6, 12, 24 and 48 h after CCl4 intoxication. Group IX and X served as post-treatment groups (curative). They received drinking water orally for 16 days and on the 16th day they received CCl4 (1ml/kg i.p.) in a 1:1 dilution with olive oil, followed by plant extract at a dose of 300 mg/kg (CAE) and 700 mg/kg (CAE) orally at 2, 6, 12, 24 and 48 h after CCl4 intoxication 15-18 detailed in Table 1.
TABLE 1: EXPERIMENTAL DESIGN
| S. no. | Group | Treatment | No. of animals | Dose (Till 15th day) | Dose on the 16th day |
| 1 | I | Normal control | 6 | Distilled water | Olive oil (1ml/Kg BW ip) |
| 2 | II | Toxic control | 6 | Distilled water | CCl4 + Olive oil (1:1 ip) |
| 3 | III | Pre-treatment (CC) | 6 | 300mg/kg BW orally | CCl4 + Olive oil (1:1 ip)
2 hr after administration of dose |
| 4 | IV | Pre-treatment (CC) | 6 | 700 mg/kg BW orally | |
| 5 | V | Pre-treatment (CA) | 6 | 300mg/kg BW orally | |
| 6 | VI | Pre-treatment (CA) | 6 | 700 mg/kg BW orally | |
| 7 | VII | Post treatment (CC) | 6 | Distilled water | CCl4 + Olive oil (1:1 ip)
Followed by 300 mg/kg BW orally after 2, 6, 12, 24,48 h of treatment |
| 8 | VIII | Post-treatment (CA) | 6 | Distilled water | |
| 9 | IX | Post treatment (CC) | 6 | Distilled water | CCl4 + Olive oil (1:1 ip)
Followed by 700 mg/kg BW orally after 2, 6, 12, 24, 48 h of treatment |
| 10 | X | Post treatment (CA) | 6 | Distilled water |
Bio-Chemical Parameter Investigation: Blood samples from each group were collected through the retro-orbital route. The collected blood samples were allowed to stand for 20 min and then centrifuged for 15-20 min at 2000 rpm to separate the serum, and the later was used for biochemical estimations of parameters viz. Serum glutamate oxaloacetate transaminase (SGOT), serum glutamate pyruvate transaminase (SGPT), alkaline phosphatase (ALP), total cholesterol, total protein, bilirubin (Total and direct) 19.
Heart tissues were homogenized in KCl (10 mM) phosphate buffer (1.15%) with ethylene-diamine tetraacetic acid (EDTA: pH 7.4) and centrifuged at 12000 rpm for 20 min. The supernatant was used for the measurement of malondialdehyde (MDA)20.
Estimation of Glutathione (GSH): Heart tissues of the animals sacrificed by cervical dislocation were collected, washed in 0.9% saline, soaked in the filter paper, weighed and stored at -20 °C. Tissue fragments (200 mg) were thawed and homogenized on ice in 1 ml of 250 mm sucrose, 20 mg Tris-HCl, 1 mg dithiothreitol, pH 7.4, using glass-Teflon homogenizers. The homogenates were centrifuged at 75,000 rpm at 4 °C for 2 h. Supernatants were stored at -20 °C. Reduced glutathione was estimated by determination of dithiobis (2nitro)-benzoic acid (DTNB) reduced by SH-groups, as described by Ellman 1959 and expressed as mole/mg protein 21.
Estimation of Catalase: 10 µl samples were taken in a tube containing 3.0 ml of H2O2 in phosphate buffer. The time required for 0.05 optical density changes was observed at 240 nm against a blank containing the enzyme source in H2O2 free phosphate buffer diluted to 100 ml 22.
Antioxidant Activity: This was evaluated in the heart homogenate by estimating the concentration of MDA, a stable product of lipid peroxidation according to the method described by Ohkawa et al. 21 Myocardial activity of SOD, CAT, and GR were determined by the method of Kakkar et al., Aebi and Bentler respectively 24, 25, 26.
Measurement of Cardiac Lipid Peroxidation: Malondialdehyde, a lipid peroxidation end product in tissue homogenate, was measured according to the method described by Wills (1969) with some modifications. A 1 ml aliquot of tissue homogenate was mixed with 2 ml of thiobarbituric acid (TBA)-trichloroacetic acid (TCA) reagent (0.375 and 15%, respectively). The volume was made up to 3 ml with distilled water and boiled on a water bath at 95°C for 20 min.
The solution was then cooled under tap water. The reaction product (TBA–MDA complex) was extracted by adding 3 ml of n-butanol to the above solution. The absorbance of the pink colored extract in n-butanol was measured at 532 nm using a spectrophotometer (Spectramax Plus, Molecular Devices, USA). The amount of MDA was calculated using a molar extinction coefficient of 1.56 × 105 M-1 cm-1 and expressed as nmoles of MDA formed per mg wet weight of tissue. MDA concentration of the sample can be calculated using an extinction coefficient of 1.56 ×105 /M cm
MDA concentration (M) = Absorbance at 532 nm / 1.56 × 105
Statistical Analysis: The data were subjected to statistical analysis. All the values are expressed as mean ± SD, and data were analyzed by One-way ANOVA, using GraphPad INSTAT. The post-hock analysis was carried out by Dunnet’s multiple comparison tests to estimate the significance of the difference between individual groups (**P<0.01). Confidence interval has been considered as 99% and p<0.01 were considered significant. The IC50 value was calculated by plotting a graph with percent inhibition on y-axis and concentration on x-axis.
RESULTS:
Biochemical Parameter Investigation: The effects of different doses of the two extracts on the SGPT, SGOT, ALP, cholesterol, total and direct bilirubin, and total protein levels are summarized in Table 2 and 3. Both the extracts at doses of 300 and 700 mg/kg body weight showed a varied effect on biochemical parameters. Altered heart function and mitochondrial heart injury were evident from the data, indicating an increment in serum concentration of enzymes such as SGPT, SGOT, and ALP.
Heart injury contributes to the increased serum level of transaminase enzymes due to the easy availability of amino acids. After treating the test group in different dose level, a significant reversal of elevated marker enzymes was observed for both the extracts in dose level of 700 mg/kg body weight Table 2.
TABLE 2: EFFECTS OF EXTRACTS ON SGOT, SGPT, ALP AND CHOLESTEROL LEVEL ON EXPERIMENTAL ANIMALS
| Groups | SGOT (U/L)* | SGPT (U/L)* | ALP (U/L)* | Cholesterol (mmol/L)* |
| Vehicle control | 65.67 ± 2.80 | 49.58 ± 2.87 | 96.26 ± 3.79 | 42.54 ± 2.19 |
| Negative control | 259.65 ± 3.04# | 219.81 ± 3.71# | 309.95 ± 3.78# | 92.16 ± 2.30# |
| Standard drug | 62.59 ± 2.84# | 51.24 ± 1.97# | 108.24 ± 2.02# | 50.51 ± 1.90# |
| CA 300 mg/kg | 120.34 ± 1.96 | 101.61 ± 2.65 | 266.73 ± 3.32 | 86.95 ± 2.83 |
| CA 700 mg/kg | 106.14 ± 3.50 | 91.57 ± 1.97 | 183.96 ± 3.36 | 75.50 ± 1.31 |
| CC 300 mg/kg | 94.02 ± 2.90# | 73.61 ± 2.45# | 134.35 ± 2.08# | 61.01 ± 2.85# |
| CC 700 mg/kg) | 89.01± 1.20 | 69.12±1.23 | 121.12± 1.11 | 54.91±1.12 |
*Values are mean ± SEM (n = 6) #Values are statistically significant at p<0.05
The lipid profile gives an important indication of metabolic disturbance, including heart disease. The higher concentration of serum cholesterol, serum bilirubin, and a lower concentration of total protein may be attributed to different coronary heart diseases. The treatment with both the extracts at a high dose level of 700 mg/kg body weight showed marked declination in the levels of markers as compared to negative control along with moderate declination in markers with lower test dose as summarized in Table 3.
TABLE 3: EFFECTS OF EXTRACTS ON DIRECT BILIRUBIN, TOTAL BILIRUBIN AND TOTAL PROTEIN LEVEL ON EXPERIMENTAL ANIMALS
| Groups | Direct bilirubin (mg/ dl)* | Total bilirubin (mg/ dl)* | Total protein (mg/ dl)* |
| Vehicle control | 0.47 ± 0.08 | 1.29 ± 0.34 | 8.04 ± 0.58 |
| Negative control | 1.16 ± 0.2# | 4.12 ± 0.12# | 4.17 ± 0.34# |
| Standard drug | 61.58 ± 1.84# | 50.29 ± 1.98# | 107.23 ± 2.01# |
| CA 300 mg/kg | 0.57 ± 0.02# | 1.65 ± 0.18# | 7.64 ± 0.38# |
| CC 700 mg/kg | 0.78 ± 0.03 | 3.14 ± 0.23 | 6.62 ± 0.45 |
| CC 300 mg/kg | 0.64± 0.02 | 2.33 ± 0.30 | 5.72 ± 0.51 |
| CC 700 mg/kg) | 0.60 ± 0.04# | 1.81 ± 0.11# | 5.01 ± 0.42# |
*Values are mean ± SEM (n = 6) #Values are statistically significant at p<0.05
Enzyme Activity Determination: The effects of different doses of both the extracts on the oxidative stress were determined in the CCl4 induced Wister rats using Silymarin as standard drugs. The results of oxidative stress marker status are summarized in Table 4. A decreased level of GSH (Glutathione) was observed in the negative control group indicating a reduction in GSH synthesis or degradation of GSH by oxidative stress. The treatment with extracts both C.A and C.C at the dose level of 700 mg/kg body weight showed a prominent increase of reductase power with reductase activity of 12.34 ± 0.43 as compared to the negative control group 5.69 ± 0.37 and standard group with 13.61 ± 0.44. Antioxidants may have a role in the prevention of cardiovascular diseases. Catalase is a haem-containing an enzyme which detoxifies H2O2 into water and oxygen. The level of catalase was improved significantly by both the extracts in a dose-dependent manner in the test group, indicating its potentiality to detoxify the reactive oxidation species.
TABLE 4: EFFECTS OF EXTRACTS ON GLUTATHIONE AND CATALASE ACTIVITY LEVEL ON EXPERIMENTAL ANIMALS
| Groups | Glutathione (U/mg protein) | Catalase (U/mg protein) |
| Vehicle control | 15.64 ± 0.72 | 21.85 ± 0.81 |
| Negative control | 5.69 ± 0.37 | 9.42 ± 0.63 |
| Standard drug | 13.61 ± 0.44 | 18.23 ± 0.71 |
| CA 300 mg/kg | 7.10 ± 0.51 | 10.94 ± 0.75 |
| CC 700 mg/kg | 9.55 ± 0.61 | 12.52 ± 0.83 |
| CC 300 mg/kg | 11.35 ± 0.55 | 15.66 ± 0.46 |
| CC 700 mg/kg | 12.34± 0.43 | 17.21± 0.39 |
*Values are mean ± SEM (n = 6). Values are statistically significant at p<0.05
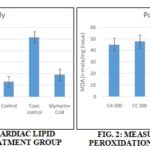
Measurement of Cardiac Lipid Peroxidation: Malondialdehyde, a lipid peroxidation end product in tissue homogenate, was measured according to the method described by Wills (1969) with some modifications. Results obtained were detailed in Table 5 and Fig. 1 and 2.
TABLE 5: MEASUREMENT OF CARDIAC LIPID PEROXIDATION
| Particulars | CA 300 | CC 300 | CA 700 | CC700 | Control | Toxic control | Std+ CCl4 |
| Pre-treatment (nano moles of MDA formed/ mg tissue) | 32.02± 0.017** | 38.45± 0.018** | 25.65± 0.013** | 32.02± 0.017 ** | 12.82± 0.010** | 51.270± 0.008** | 19.225± 0.018** |
| Post treatment (nano moles of MDA formed/ mg tissue) | 44.84± 0.018** | 47.66± 0.018** | 45.37± 10.32** | 44.84 ± 0.018** | 12.83± 0.018** | 51.25± 0.018** | 19.25± 0.018* |
Values are expressed as Mean ± SD.; (n = 6); One Way ANOVA followed by Turkey – Kramer Multiple Comparison tests; **p<0.01 vs. control group.
DISCUSSION: There is a dynamic relationship between reactive oxygen species (ROS) and antioxidants in the human body. Healthy cells can scavenge free radicals effectively using antioxidants. In pathological conditions like cardio toxicity, sudden generation of ROS can dramatically change the balance with an increasing demand of antioxidants defense mechanism. Once free radicals are generated, they alter the structural and functional integrity of the cells by a variety of mechanisms including lipid peroxidation 27. Endogenous antioxidants are depleted due to the accumulation of ROS.
In the present study, cardiotoxicity was associated with increased oxidative stress as evidenced by an increase in myocardial TBARS (a marker for cellular injury). Lipid peroxidation was estimated in terms of Thiobarbituric acid reactive species (TBARS) using MDA as standard by Buege and Aust 1978 method 28. Malondialdehyde (MDA), a lipid peroxidation end product in tissue homogenate, was measured according to the method described by Wills 1969 with some modifications. Results obtained were detailed in Table 2. For both pre-treatment study and post-treatment study MDA concentration was maximum for the toxic control group and minimum for the control group. For the extracts of C. colebrookianum and C. asiatica at both the dose level 300 mg/kg body weight and 700 mg/kg body weight is showing a significant reduction in MDA production. For C. colebrookianum MDA production is less on a higher dose, and for C. asiatica at low dose, only MDA production is less. It signifies both the plant extracts are having potent antioxidant activities, and it is evidenced from previous literature also 29.
CONCLUSION: The observations made in the present study revealed that both the plant C. colebrookianum and C. asiatica possesses significant antioxidant activity which might help prevent or slow the progress of various oxidative stress related to cardiovascular diseases. From the above study, it can be stated that the cardioprotective activity of the methanolic leaf extracts of Clerodendrum colebrookianum Walp and Centella asiatica Linn., particularly the higher doses were significantly able to reverse and restore the serum imbalances induced by CCl4 in the experimental animals. The study thereby justifies the traditional usage of the plant in treating hypertension hence, fully supporting the in vivo cardioprotective status. Further study is warranted about elucidation and characterization of active biomolecules from the extract to augment the traditional practices.
ACKNOWLEDGEMENT:
- The authors are thankful to BSI, Shillong, Meghalaya, India, for providing identification and authentication facility of the plant materials.
- The authors also extend their gratitude towards the Division of Pharmaceutical Technology, Defence Research Laboratory, Tezpur, Assam, India, to share their expertise.
- The authors are grateful to Dibrugarh University for providing laboratory facility and a Central Animal House Facility.
- The authors are grateful to the University Grant Commission for funding the research work.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Andersen OM and Markham KR: Flavonoids: Chemistry, Biochemistry and Applications. Boca Raton: CRC Press 2006.
- Adaramoye OA: Comparative effects of vitamin E and kolaviron (a biflavonoid from Garcinia kola) on carbon tetrachloride-induced renal oxidative damage in mice. Pakistan J of Biological Sciences 2009; 12: 1146-51.
- Ahmad FF, Cowan DL and Sun AY: Detection of free radical formation in various tissues after acute carbon tetrachloride administration in gerbil. Life Sciences 1987; 41: 2469-2475.
- Ozturk F, Ucar M, Ozturk IC, Vardi N and Batcioglu K: Carbon tetrachloride-induced nephrotoxicity and protective effect of betaine in Sprague-Dawley rats. Urology 2003; 62: 353-56.
- Rechnagel RO, Glende EA and Plaa GL: Carbon tetrachloride hepatotoxicity: an example of lethal cleavage. CRC Critical Reviews in Toxicology 1973; 2: 263-97.
- Brattin WJ, Glende Jr EA and Recknagel RO: Pathological mechanisms in carbon tetrachloride hepatotoxicity. Journal of Free Radicals in Biology & Medicine 1985; 1: 27-38.
- Rikans LE, Hornbrook KR and Cai Y: Carbon tetrachloride hepatotoxicity as a function of age in female Fischer 344 rats. Mechanisms of Ageing and Development 1994; 76: 89-99.
- Shenoy KA, Somayaji SN and Bairy KL: Hepatoprotective effects of Ginkgo biloba against carbon tetrachloride induced hepatic injury in rats. Indian Journal of Pharmacology 2001; 33: 260-66.
- Yuan LP, Chen FH, Ling L, Bo H, Chen ZW, Li F, Zhong MM and Xia LJ: J Pharmacol 2008; 60: 1393-02.
- Upur H, Amat N, Blazekovi B and Talip A: Food Chem Toxicol 2009; 47: 2022-30.
- Adewole S, Salako A, Doherty O and Naicker T: Effect of melatonin on carbon tetrachloride-induced kidney injury in Wistar rats. African Journal of Biomedical Research 2010; 10.
- Plaa GL and Witschi H: Chemicals, drugs, and lipid peroxidation. Annual Review of Pharmacology and Toxicology 1976; 16: 125-42.
- Giordano FJ: Oxygen, oxidative stress, hypoxia, and heart failure. J Clin Invest 2005; 115: 500-08.
- Deb L and Dutta A: Evaluation of mechanism for antihypertensive action of Clerodendrum colebrookianum used by folklore healers in North East India. J Ethnopharmacol 2012; 143(1): 207-12.
- Jadon A, Bhadauria M and Shukla S: Protective effect of Terminalia bellerica And gallic acid against carbon tetrachloride-induced damage in albino rats. Journal of Ethnopharmacology 2007; 109: 214-18.
- Khan MR, Rizvi W, Khan GN, Khan RA and Shaheen S: Carbon tetrachloride-induced nephrotoxicity in rats: Protective role of Digera muricata. Journal of Ethnopharmacology 2009; 122: 91-99.
- Vaca CE, Wilhelm, JM and Harms-Rihsdahl M: Interaction of lipid peroxidation product with DNA. Gen Toxicol 1988; 195: 137-49.
- Mohamed AM: Prophylactic role of L-carnitine and ubiquinone in combating the cardio-toxicity induced by carbon tetrachloride in rat. International Journal of Academic Research 2010; 2.
- Verley H, Gowenlock AH and Bell M: Practical clinical biochemistry. London: William Heinemann Medical Books; Edition 5th, 1980.
- Upreti GC, Davis C and Oliver J: Preparation of representative homogenates of biological tissues: Effect of salt on protein extraction. Analytical Biochemistry 1991; 198(2): 298-01.
- Mulder TPJ, Manni JJ, Roelofs HMJ, Peters WHM and Wiersma A: Glutathione-s-transferases and glutathione in human head and neck cancer. Carcinogenesis 1995; 16: 619-24.
- Bergmeyer HV and Bernt E: Methods of enzymatic analysis, New York, NY: Academic Press, Vol. 3, 1974.
- Aebi H: Catalase: In: Bergmeyer, Hv eds. Methods in enzymatic analysis. New York: Academic Press Inc; 1994. PMCid:PMC44120
- Bentler E: Effect of flavin compounds on glutathione reductase activity: In-vivo and in-vitro J Clin Invest 1975; 48: 1957-66. PMid:5822598 PMCid:PMC 322432. Available from: https://doi.org/10.117/ JCI106162
- Akpabio UD, Udo UE and Akpankpan AE: Evaluation of phytochemical, proximate and mineral element composition of the stem of Costus afer. Asian J Plant Sci Res 2012; 2: 607-12.
- Sun L: Physical association of the APIS complex and general transcription factors. Biochem Biophys Res Commun 2002; 296(4): 991-99.
- Buege JA and Aust SD: Microsomal lipid peroxidation. In Methods in Enzymology. Academic Press, Vol. 52, 1978: 302-10.
- Devi R, Banerjee SS and Maulik SK: In-vitro and in-vivo antioxidant activity of different extracts of the leaves of Clerodendrum colebrookianum Walp in the rat. Journal of Pharmacy and Pharmacology 2003; 55: 1681-86.
How to cite this article:
Das RJ and Mazumder B: Effects of aqueous methanolic leaf extracts of indigenous plants Clerodendrum colebrookianum Walp and Centella asiatica Linn. on carbon tetrachloride induced cardiotoxic wister albino rats. Int J Pharmacognosy 2019; 6(6): 202-08. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.6(6).202-08.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.