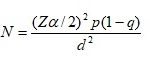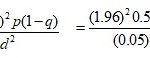DRUG USE EVALUATION OF ARTEMETHER-LUMEFANTRINE (COARTEM®) FOR FALCIPARUM MALARIA IN KARA MARA HOSPITAL, JI-JIGA TOWN, FAFAN ZONE, EASTERN ETHIOPIA
HTML Full TextDRUG USE EVALUATION OF ARTEMETHER-LUMEFANTRINE (COARTEM®) FOR FALCIPARUM MALARIA IN KARA MARA HOSPITAL, JI-JIGA TOWN, FAFAN ZONE, EASTERN ETHIOPIA
Abdirahman Omar * 1 and Alemu Tadesse 2
Department of Pharmaceutical 1, Chemistry and Pharmacognosy 2, School of Pharmacy, Haramaya University; Dire Dawa, Ethiopia.
ABSTRACT: Background: Malaria remains the most common public health problem in developing countries and is the number one cause of morbidity and mortality especially in children below five years of age. Since, November 2006, Coartem® an artemisinin combination therapy containing artemether-lumefantrine replaced sulphadoxine/ pyrimethamine as the first-line drug for treatment of uncomplicated malaria in developing countries including Ethiopia due to the development and spread of resistance of SP to Plasmodium falciparum. Objective: To evaluate the drug utilization pattern of coartem combination therapy in the treatment of P. falciparum Methods: A retrospective study was conducted from January 2015 - March 2015 on patient cards with coartem® prescription recorded from January 2014 to December 2015. Results: In this study, we observed that the dosing of coartem® mainly depends on age and weight. While the majority of patients 60.0% were given correct and 21.3% were received incorrect dose while the 18.7% patients were not stated doses. Conclusion: After analysis of dose, frequency, duration, indication, contra-indication, drug-drug interaction of coartem® given for patients in the hospital were indicated irrational of coartem® use. So, the health professionals especially pharmacy professionals should regularly and consistently check the rationality of the prescribed drugs.
| Keywords: |
Coartem, Combination therapy, Malariologist, Omission error, Falciparum malaria
INTRODUCTION: Malaria remains the most common public health problem in developing countries and is the number one cause of morbidity and mortality especially in children below five years of age 1.
Since, November 2006, Coartem® (Novartis Pharma, Basel Switzerland) an artemisinin combination therapy (ACT) containing artemether-lumefantrine (AL) replaced sulphadoxine/ pyrimethamine (SP) as the first-line drug for treatment of uncomplicated malaria in developing countries including Ethiopia due to the development and spread of resistance of SP to Plasmodium falciparum 1. Drugs play a crucial role in saving life in the health care system. The limited information available on drug use throughout the world indicated that drugs are not optimally used which leads to inappropriate use.
This inappropriate use has serious and economic consequences for individuals, community and the success of national health care system 2. Drugs have to be safe, efficient and have to be used rationally and require a particular concern due to their unwanted side effects and the threat of resistance. In general, the safe and effective use of drugs depends on prescription writing pattern. So, rational prescription pattern and providing correct information during dispensing is inviolable for proper utilization of drugs 3. In much of tropics, drug-resistant Plasmodium falciparum is increasing in distribution, frequency, and intensity. There is a growing belief among malariologists that, to prevent resistance, falciparum malaria, should be treated with drug combination 4. Hence, inexpensive, safe drugs and ultimately vaccines are needed to combat malaria 5.
Anti-malaria, combination therapy is the simultaneous use of two or more drugs with independent modes of action and thus unrelated biochemical targets 6. Combination therapy is not a new phenomenon. It has been deployed in the treatment of Tuberculosis, Leprosy, and AIDS. It delays the emergence of resistance and increases efficacy7. Artemether-lumefantrine (AL) coarterm® is one of the artemisinin combination therapies (ACT) drugs being used by most countries in sub-Saharan Africa.
It is the first ACT available in a combination of Artemether and lumefantrine in 1:6 ratios and it is new effective and well-tolerated drug for malaria 8. It achieves its antimalarial effect through the large initial reduction in parasite biomass by artemether and subsequent removal of all the remaining viable parasites by the less active but more slowly eliminated lumefantrine. The combination also provides mutual protection of the two antimalarial drugs from the development of resistance 9.
Coartem® has complicated presentations (presented in a different package of 6, 12, 18, 24 tablets for treatment by patient weight bands, has a 14 month lead time for production and has a short half-life, that is 2 years, and health workers lack the experience of with its use 10. Ethiopia accepted AL as the first-line drug for the treatment of uncomplicated Plasmodium falciparum which is administered twice a day for three days, and it stated that it is not given to infants <5 kgs, pregnant women, severe malaria and prophylaxis 11.
Statement of the Problem: Malaria is an infectious disease that is caused by intracellular protozoa, the genus Plasmodium that is transmitted by the bite of an infected female Anopheles mosquito 12. Among Plasmodium species, the four, P. falciparum, P. vivax, P. malaria, and P. ovale, are causes of human malaria 13. It is estimated that one million children die from malaria annually in the world 14, 15. An estimated 90% of all global deaths due to malaria occur in sub-Saharan Africa 16. It has also slowed economic growth by 1.3% per year resulting in a gross domestic product which is 32% lower than it would have been had malaria been eradicated from Africa in 1960 17.
In sub-Saharan Africa, malaria has for a long time been treated using cheap and effective drugs such to which malaria parasite has gradually developed resistance. Because of this, a new artemisinin-based drug, coartem® is used. A major challenge is ensuring rational use and access at the household level. It is hoped that addressing those issues will increase the likely hood that AL Achieves its intended goals of reducing morbidity drug to malaria delaying the onset of drug resistance 18. Therefore appropriate drug utilization studies are important tools to evaluate whether drugs are properly utilized at all levels in the chain of drug use.
The Significance of the Study: The rationale for conducting this study is to provide the baseline information on Artemether-lumefantrine (coartem®), utilization pattern for uncomplicated malaria in study area by quantifying the present drug use practice in the hospital, significance of the drug and to know problem areas and recommend the concerned body for all essential data obtained after finalizing the data collection process.
Literature Review: Drugs to have correct action at a correct site in a desirable amount on time should be prescribed in the right dose, at a right frequency for long enough duration of time. However, some studies indicated that many physicians omit this information 19. A study done on evaluation of drug and medication-related prescribing errors in the primary health center in Bahrain, in may 2004 revealed that in 54.1% of the prescription with omission errors. The length of therapy was not specified in 27.7%, and the dosage form was not stated in 12.8%. In 43.5% of the prescription with errors of omission, there were 20.8% dosing frequency and 17.7% dose/strength related errors. 2.9% Errors of drug-drug interaction comprised of all prescribed over extended periods and (2.7%) low dosing frequency and (2.6%) excessive doing frequency 19.
According to the research done in Zambia among children weighing 50 kg or more sulphadoxine +pyrimethamine /SP/ was commonly prescribed (68%) whereas the recommended AL was prescribed for only 11% of children. Among children weighing more than 10 kg seen at facilities where AL was available, AL was prescribed for 22% of children and SP for 54% 19. A follow-up survey two years later found that the proportion of children weighing 5-9 kgs treated with AL rose from 11% to 27% and those weighing 10 kg or more changed from 22% to 42% 20. Research done in Kenya indicated that an average of 46.9% of patients received appropriate anti-malaria for the treatment of uncomplicated malaria. Percentage of encounters diagnosed as uncomplicated malaria and prescribed antimalarial consistent with the national treatment Guideline (NTG) for malaria in health center found in Inakueni, Kwale, Bondo, and Kisii towns were 68%, 66%, 60%, and 36% respectively 21.
A research was done in the college of medicine, the University of Ibadan, ladan, Nigeria showed that a recently approved artemisinin-based tablet, coartem®, comprising artemether 120 mg and lumefantrine 20 mg, given in four doses provides effective antimalarial treatment for children in sub-Saharan countries. This open level study assessed the efficacy and safety of coartem® administered to 310 African children weighing 5-25 kg, with acute, uncomplicated falciparum malaria. Six doses of coartem® were administered over 3 days, with follow up at 7, 14 and 28 days. Treatment rapidly cleared parasitemia and fever. The overall 28-day cure rate was 86.5% and at 7 and 14 days exceeded 97.0%. On day 28 in infants (5-10 kgs) previously exposed to malarial infection/partially immune 88.6% and with those nonimmune 82.5% 22. A research done in Gahanna on children aged from 6-59 months old to see the feasibility and acceptability of the use of AL for management of uncomplicated falciparum malaria showed that all 235 children aged 6-35 months were correctly provided the one tablet per dose per treatment packaged compared with 119 of 125 children 36-59 months (95.2%). Only 5 of 17 children were referred appropriately. All 334 caregivers followed the correct drug administration schedule; hence a day for three days validation of drugs received indicated that all 204 children aged 6-35 months and 103/118 (87.3%) children aged 36-59 months received the correct drug dose. Adherence of agents and caregivers to the treatment was 308/334 (92.5%) 23.
MATERIALS AND METHODS:
Study Area and Period: The study was conducted at Karamara hospital in Jijiga. Jijiga is the capital city of the Somali region which is the second largest region next to Oromiya in Ethiopia. It is located at 610 km away from Addis Ababa; the capital city of Ethiopia, to the eastern part of Ethiopia. The town has 20 kebeles with a total population of 4, 439, 147 (2007 E.C) 24. The town is situated at an altitude range 1630m above sea level. The Kara Mara hospital has 26 medical doctors, 4 anesthesias, 2 cataract surgeon, 10 health officer, 35 clinical nurses, and other professional staffs. The study was conducted from November 2015 to June 2015.
Study Design: A retrospective study was conducted from January 2015 - March 2015 on patient cards with coartem® prescription recorded from January 2014 to December 2015. Patient cards were selected to assess coartem® prescription pattern by using systematic random sampling techniques.
Study Population: The study population was all patient cards with coartem® prescription between January 1/2014 to December 31/2015.
Source population: The source population was all patient cards (records) of coartem® prescription in Kara Mara Hospital.
Inclusion and Exclusion Criteria:
Inclusion Criteria: All patients who treated malaria, especially P. falciparum that are used for coartum which is recorded on the patient card at January 2014 to December 2015 was all included candidates for this study.
Exclusion Criteria: All patients who were treated for malaria that are used coartum a time rather than the specified duration, that is from January 2014 to December 2015 or those lost their medical cards, or poorly recorded medical patient cards were all be excluded from this study.
Sampling Techniques and Sample Size: The total sample size was determined using the statistical formula 25.
Where: Z= Reliability coefficient for the designed interval, N= The minimum sample size determined, P= Proportion of population possessing the characteristics of interest 50% (0.5), q= The proportion of population possessing the characteristics of interest, q= 1-p ≈1-05=0.5 (50%), d= the margin of sampling error tolerated.
In this study was including 150 patients that are recorded medical patient card from the time January 2014 to December 2015 that prescribed coartem.
Study Variables:
Dependent Variables: Dose, frequency, indication, contraindication.
Independent Variables: Duration, age, weight.
Data Collection: Data collection format that comprises patient information such as age, weight, malaria type, contraindications and drug data like; dose, frequency, and duration of administration were used to collect data. The data was collected by the investigator student after proper training about the data collection format under the supervision of the principal investigator. To evaluate the data collection instrument for its validity, reliability, and consistency, the pilot study was done on a small proportion of patient cards that are not included in the main research before the beginning of the data collection and some correction is going to be made accordingly. The data collected was checked for the completeness, accuracy, and consistency by the principal investigator every day.
Ethical Consideration: The head of Kara Mara hospital was informed about the study by formally written letter by School of Pharmacy Haramaya University, and thus ethical issues were respected throughout the study period.
RESULTS AND DISCUSSION:
Results: The study was conducted on 150 patient cards with coartem® prescription in Kara Mara hospital by using a cross-sectional retrospective study. From the study deferent variables were analyzed, and the results were presented as follows.
Socio - Demographic Characteristics: The majority of study subjects were females 94 (62.7%) while the majority of patients 121 (80.6%) were above ten years while only 29 (19.3%) patients were less than 10 years.
TABLE 1: NUMBER OF PATIENTS BY AGE GROUP WHO WERE TREATED BY COARTEM® IN KARA MARA HOSPITAL IN JIJIGA TOWN FAFAN ZONE, EASTERN ETHIOPIA
| Age group | Frequency (%) |
| 0-4yrs
5-9yrs 10-14yrs 15-19yrs 20-24yrs 25-29yrs 30-34yrs >35yrs |
20 (13.3%)
9 (6 %) 6 (4.0%) 4 (2.7%) 15 (10.0%) 46 (30.7%) 29 (19.3%) 21 (14.0%) |
The majority of patients above 35kg body weight were 116 (77.3%) who were treated with coartem® while only 4 (2.7%) was less than 5kg. Majority of patients 80 (53.3%) who received coartem® were others while only 4 (2.7%) patients were infants less than 5 kg, 38 (25.3%) were a nursing mother, and 28 (18.7%) were pregnant mothers.
TABLE 2: DISTRIBUTION OF PATIENTS BODY WEIGHT IN KARA MARA HOSPITAL, JIJIGA TOWN, FAFAN ZONE EASTERN ETHIOPIA, WHO WERE TREATED WITH COARTEM® FROM JAN 2014-DEC, 2015
| Body weight (kg) | Frequency (%) |
| <5kg
5-14 15-24 25-34 |
4 (2.7%)
25 (16.7%) 8 (2.09%) 1 (0.7%) |
| >35 | 116 (77.3%) |
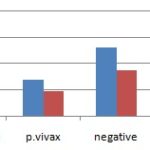
The Result from Laboratory and Assessment: Among patients treated with coartem®, 101 (67.3%) were done with the help of laboratory results while the only 49 (32.7) were not done laboratory result. The laboratory results of kara mara hospital from Jan. 2014-Dec. 2015 indicated that 98 (65.3%) patient’s blood serum were positive Plasmodium and 52 (34.7%) patients were negative Table.
FIG. 1: THE LABORATORY RESULTS OF PATIENTS TREATED WITH COARTEM® IN KARA MARA HOSPITAL FROM JAN 2014-DEC 2014
The majority of malaria cases for which coartem® prescribed were uncomplicated 110 (73.3%), and 40 (26.7%) were sever malaria cases.
The Result of Drug Information: After coartem® therapy, 51 (34.0%) of the clients came back to the hospital with similar history within a 4th and 14th day of therapy. But the remaining 99 (66.0%) were didn’t come back to the kara mara hospital.
TABLE 3: FREQUENCY AND DURATION OF COARTEM® TREATMENT IN KARA MARA HOSPITAL EASTERN ETHIOPIA FROM JAN 2014-DEC 2015
| Frequency of treatment | Frequency (%) |
| Correct
Incorrect |
85 (56.7%)
26 (17.3%) |
| Not stated | 39 (26.0%) |
| Duration of treatment | |
| Correct
Incorrect Not stated |
94 (62.7%)
19 (12.7%) 37 (24.7%) |
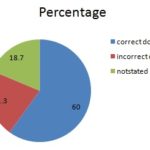
In this study, were seen that dosing of coartem® mainly depends on age and weight. Majority of patients 90 (60.0%) were given correct, and 32 (21.3%) were received incorrect dose while the 28 (18.7%) patients were not stated doses. The majority patient 85 (56.7) were given correct of coartem® frequency, and 26 (17.3%) were also given incorrect frequency while 39 (26.0%) patient were not stated frequency, and 94 (62.7%) patient were received correct duration, 19 (12.7%) were incorrect duration, and 37 (24.7) were not stated duration of treatment Table.
FIG. 2: THE DOSE OF COARTEM® TREATMENT IN KARA MARA HOSPITAL EASTERN ETHIOPIA FROM JAN 2014-DEC 2015
The majority of drugs prescribed with coartem® were antipyretics (Paracetamol) that was 90 (60.0%) and antibiotics 36 (24.0%).
TABLE 4: DRUGS PRESCRIBED WITH COARTEM® IN KARA MARA HOSPITAL, EASTERN ETHIOPIA FROM JAN 2014-DEC 2015
| Drugs | Frequency | Percentage |
| Paracetamol | 90 | 60.0 |
| Antibiotic | 36 | 24.0 |
| Vitamin B-complex | 22 | 14.7 |
| Others | 2 | 0.3 |
DISCUSSION: According to this study, the age of all patients treated with coartem® was stated in the patient cards. But for 90 (60.0%) of patients, the drug was given with considering their body weight while 32 (21.3%) patients whose body weight was not considered, 28 (18.7%) was not stated. The malaria diagnosis and treatment guideline for health workers in Ethiopia state that the age and weight of the patient must be considered simultaneously to give coartem® for the treatment of uncomplicated falciparum malaria 10.
The study indicated that the majority of patients 121 (80.6%) treated with coartem® were above 10 years old. While 29 (19.3%) were below 10 years. Where 116 (77.3%) were above 35kg and 34 (22.6%) were below 34kg. While the malaria diagnosis and treatment guideline states that the dose of coartem® patients above this weight are four tablets of strength 140 mg of each. There were also about 14 (9.3%) overdose and 18 (12.0%) under dose prescribing of coartem® incidences concerning the age of patients. The incidence was higher when compared to the research done in Bahrain on the evaluation of drug and medication-related prescribing errors in primary health center which was 2.6% and 2.7% respectively 11.
In the study were practiced of coartem® prescribing in terms of dose, frequency, and duration, there were 32 (21.3%) dose-related errors, and the dose of therapy was not stated 28 (18.7%) of patients. When compared to research was done in Bahrain the dose-related error which was 17.7% 11 were lower than errors observed in this study. On the other hand, the length of therapy and dosing frequency related errors were 37.3% and 43.3% respectively 11, which were higher than the finding of this study.
The probable reason for the difference is that coartem® has much-complicated dosage regimens than other drugs depending on different age groups and the research done in Bahrain includes some drugs whereas this study is limited to a single drug coartem®. The duration and frequency related errors were lower because all doses of coartem® in different age group have similar duration and frequency.
According to the malaria diagnosis and treatment guideline for Ethiopia coartem® is indicated for patients of uncomplicated falciparum malaria who are non-pregnant, non-nursing mothers and infants older than 3 months and weigh above 5 kg. But this study was shown that 4 (2.7%) infants, 28 (18.7%) pregnant, 38 (25.3%) nursing mothers as well as 80 (53.3%) patients were given coartem®.
A research done in Kenya indicated that an average of 53.1% of patients was not received an appropriate antimalarial for the treatment of uncomplicated malaria 9. Coartem® is active against Plasmodium falciparum, which is currently becoming resistant to various anti-malaria drugs. So, coartem® is reserved for Plasmodium falciparum as a first line drug. Therefore the diagnosis must be supported with laboratory findings to differentiate the causative species. This study indicated that 101 (67.3%) of patients were diagnosed with the help of laboratory results. The remaining 49 (32.7%) were given coartem® without any lab diagnosis. The malaria diagnosis and treatment guideline state that coartem® can be given if uncomplicated falciparum malaria is suspected and microscopy or rapid diagnostic tests are negative coartem® is not recommended.
In study 98 (65.3%) of patients were Plasmodium positive which 70 (46.7%) falciparum positive, 28 (18.6%) of patients were also positive with Plasmodium vivax and treated with coartem®. At baseline 52 (34.7%) of patients were negative that were treated. while only 70 (46.7%) of patients were treated from their malaria as per the guideline. When this result is compared with the research done on patients received appropriate antimalarial for the treatment of uncomplicated malaria in Kenya, it is lower than obtained in Inakueni, Kluale, and Bondo 98%, 66%, and 60% respectively and greater than observed in the other town Kissi which was 36% 9. The finding of this study was shown that 51 (34.0%) of patients were returned to the hospital with the similar history between the 4th and 14th day of treatment.
The malaria diagnosis and treatment guideline state that if a patient comes back to the hospital with similar history after coartem® treatment within this period, the treatment is quinine indicating that there would be suspected of resistance to coartem®. But this incidence should be assured by further research.
In rational drug utilization, the very necessary and crucial thing is considering the drug interaction. According to a study among drugs prescribed together with coartem® 36 (24.0%) were antibiotics among which 10(6.6%) were ciprofloxacillin. The Ethiopian national drug formulary states that coartem® has interaction with ciprofloxalillin. The interaction is in prolonging the QTc interval of the cardiac system, which is pharmacodynamic interaction.
CONCLUSION: In general this study that was done in the kara mara hospital, there was irrational use of coartem® in many perspectives. After analysis of dose, frequency, duration, indication, contraindication and body weight, and drug-drug interaction, it was indicated that there were irrationalities of the drug use of coartem®. All of which are important variables that affect the effectiveness of the treatment, development of adverse drug reaction (ADR) and development of resistance. In the hospital, coartem® was also prescribed for the treatment of Plasmodium vivax and severe malaria which is contraindicated and also to patients whose blood film is negative after microscope examination. And all those irregularities (problems) should be resolved rationally.
RECOMMENDATIONS: Based on the findings of this study large and comprehensive efforts should be done in the hospital to ensure rational use of coartem® by identifying existing prescribing and dispensing problems.
Special training to prescribes and dispensers must be given to make them more familiar to this newly emerging drug on its use, major adverse drug reactions, and indications. This reduces irrational to use that would lead to resistance development.
The hospital should ensure that the availability of reference materials, guidelines like malaria diagnosis and treatment guideline and standard treatment guideline and formularies which assist the best use of coartem® is guaranteed and easily accessible to all investigators willing to do any means of data analysis.
ACKNOWLEDGEMENT: Nil
CONFLICT OF INTEREST: Nil
REFERENCES:
- Procurement of artemether/lumefantrine(Coartem) through WHO 2006
- Yenet W: Baseline survey on drug prescribing indicators for outpatients in JUSH, SW-E. Ethio J Health Science 2005; 15(2): 147-156.
- Introduction STG for zonal hospital first education. Addis Ababa, Ethiopia: Chamber printing house, X-xvii. 2004;
- Favci and Lango: Harrison’s principle of internal medicine. USA-New York: Medical publishing division, Edition 16th, 2005.
- Sahanah F, Foltin J, Edmonson K and Brown Y: The pharmacology basis of therapeutics. USA-New York: Medical publishing division, Edition 11th, 2006.
- WHO Guidelines for the treatment of malaria. Geneva; 2006: 250-260.http://www.who.int/macaria/docs/treatment guideline.
- UNICEF/ UNDP/ World bank/WHO special program for research and training in tropical diseases: social science research on tropical diseases. Version1; Geneva: WHO/TDR; 2004 Feb 18.
- Van vugt M, Brockman A and Gemperli B: A randomized comparison of artemether-benflumetol and artesunate-mefloquine in the treatment of multidrug resistant falciparum malaria: anti microbe agent Chemother 1998; 42(3): 135-139. Accessed from: http://www.prosone.org/
- Whit NJ: Preventing antimalarial drug resistance through combinations. Drug Resistance Updates 1997; 1(5): 3-6. Available from: Linkinghub.elsovier.com/
- Tetteh G, Njoroge E and Wambua P: Assessment of the use of ant malarial medicines in public and private sectors of Kenya: Research findings for evidence-based strategy development. 2006; accessed from http://www. popline. org/
- Malaria diagnosis and treatment guideline for health workers in Ethiopia, Edition 2nd, 2004: 449.
- Chinbuah M, Gyapong O and Pagnoni F: Feasibility and acceptability of the use of Artemether-lumefantrine in the home management of uncomplicated malaria in children 6-59 months old in Gahanna. Accessed from: http.interseciencewhiley.Com/Journal/11859879/abstracts.
- Snow R, Gaig MH. Newton CRJC and Steketee RW: The public health burden of Plasmodium falciparum malaria in Africa: deriving the number working paper No.11. Bethesda, Maryland: Fogarty International Center, National Issues of Health 2003.
- WHO, UNICEF: The African malaria report: Geneva. 2003;
- Pugemalila JB, lwanga CR and Kilama WL: Six malaria day in 2006. How far have we come after the Abuja Declaration? Mala J 2006; 5: 102.
- Roll back malaria: Malaria in Africa. Accessed from RBM website may 2007; http;//www.rbm.who.int.comc-upload10/0001015 /370/Rbminfosheet.
- Dill J, Custem, Wensin KA, Wouters B, Voorthor Zeen J and Voorn W: Comparison of amodiaquine and sulphadoxine-pyrimethamine as first-line treatment of falciparum malaria in Kenya: Trans R. soc Trop med Hyg 1999; 93(2): 185-188.
- Khalid AS, Khajaa AL, Alansarib TM and Awatif H: Evaluation of drug utilization and prescribing errors in infants: A primary care prescription based study 2007; 81 (2): 350-357. Available at: hhh://www.Journals, Elsevier health com/periodicals/abstract
- Zurovac D, Ndhlovu M, Rowe AK, Hamer DH, Thea DM and Snow RM: Treatment of pediatric malaria during a period of drug transition to artemether-lumefantrine in Zambia, A cross-sectional study. BMS 2005; 331(10): 734-737.
- Zurovac D, Ndhlovu M and Sipilanyambe N: Pediatric malaria case management with artemether-lumefantrine in Zambia, repeat cross-sectional study: Mal J 2007; 6(1): 31.
- The Amazon network for the surveillance of anti-malaria drug resistance and the Amazon malaria initiative, No. 3. April – June 2005 Available from: http://www.paho.org/ English /
- Falade C, Maxanga M, Prmji Z, Ortmatn CE, Mayek S and Palancios PI: Efficacy and safety of Artemether-Lumefantrine (coartem) tablets (six-dose regimen) in African infants and children with acute, uncomplicated falciparum malaria: Trans R Soc Trop Mednyg 2005; 99(6): 459-467.
- Batty G, Grant R, Agarwal R, Lowe D. Potter J, Pearson M and Jackson S: Using prescribing indicators to measure the quantity of prescribing to elderly medical in patients age and aging 2003; 32 (3): 292-298.
- Amhara region finance and economics Beuro. Population size by sex and age group, Chagni town administration, 2009: 41-42.
- Degu G and Tessema F: Estimation. Biostatistics for health students lecture note. Gonder University 2005; 163-185.How to cite this article:Omar A and Tadesse A: Drug use evaluation of artemether-lumefantrine (coartem®) for falciparum malaria in Kara Mara Hospital, Ji-Jiga Town, Fafan Zone, Eastern Ethiopia. Int J Pharmacognosy 2016; 3(7): 306-13. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.3(7).306-13.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.