COMPARATIVE STUDY ON PHYSIO-CHEMICAL PARAMETERS OF ACACIA ARABICA & PROSOPIS JULIFLORA
HTML Full TextCOMPARATIVE STUDY ON PHYSIO-CHEMICAL PARAMETERS OF ACACIA ARABICA & PROSOPIS JULIFLORA
Sarika Nigam * 1, Vikram Singh 2, Archana Dongray 1 and Dilip Kumar Chanchal 3
College of Pharmacy 1, SRGI Ambabai, Jhansi - 284002, Uttar Pradesh, India.
Department of Pharmacognosy 2, SRI, Datia - 475661, Madhya Pradesh, India.
Department of Pharmacognosy 3, Insitute of Pharmacy, Bundelkhand University, Jhansi - 284128, Uttar Pradesh, India.
ABSTRACT: Aim of study: Compare the study on physio-chemical parameters of Acacia arabica and Prosopis juliflora. Material and Methods: The ethanolic extract of on Acacia arabica and Prosopis juliflora were using physio-chemical parameters and preliminary phytochemical investigation. Results and Discussion: In this study. I have done the comparative pharmacognostic study on Acacia arabia and Prosopis juliflora and conclude that the ethanolic extract of Acacia arabica plays a more significant role and has more significant value than the extract of Prosopis juliflora. Conclusion: The present study was aimed at the pharmacognostical study. Plants Acacia arabica and Prosopis juliflora were studies for pharmacognostical characteristic, namely, morphology, microscopy, which can be of utilized in identification and authentication of the plant.
| Keywords: |
Acacia Arabica, Prosopis juliflora, Phyto-Pharmacognostic
INTRODUCTION: All over the world, especially in developing countries, approximately 80% of the population continues to use traditional medicine in primary medical problems. In the past decade, therefore, research has been focused on scientific evaluation of traditional drugs of plant origin. There is an urgent need to systematically evaluate the plants used in traditional medicine. Such research could lead to new drug discovery or advance use of indigenous herbal medicines for treatment 1. Various Medicinal plants have been used for centuries as remedies for disease because they contain components of therapeutic values.
According to the WHO, 80% of the world population continues to rely mainly on traditional medicine for their health care 2. India has a rich heritage of traditional medicine constituting with its different components like Ayurveda, Siddha, and Unani, and traditional health care has been flourishing in this country for many centuries. Botanicals constitute of the major part of these traditional medicines. With the emerging world-wide interest, in adopting traditional practices, in the health care systems by exploiting their potential, the evaluation of the botanicals in these systems of medicine in India is utmost essential 3.
Herbalism (herbal medicine) as an alternative medical therapy is defined as the use of plants or substances derived from them, in treating disease, usually by medical herbalists without an orthodox medical qualification. Before the relatively recent application of scientific method into diagnosis and therapeutics, traditional medicines were mostly herbal 4. Ayurvedic system understanding the knowledge of plants used for Ayurvedic preparations in relation to their use as therapeutic agents, pharmacological properties, medicinal plants being imported; medicinal plant parts being exported, endangered medicinal plants and availability of medicinal plants in different bio-geographical zones of India can be utilized in drawing strategies for rational and more scientific use of medicinal plants in a way that can be extended for future scientific investigation in different aspects 5. There were thought to be roughly 1300 species of acacia worldwide, about 960 of them native to Australia, with the remainder spread around the tropical to warm-temperate regions of both hemispheres, including Europe, Africa, southern Asia, and the Americas.
Natural Products in Medicine: Natural products are products from various natural sources, plants, microbes, and animals. They can be an entire organism (e.g. a plant, an animal or a micro-organism), a part of an organism (e.g. leaves or flowers of a plant, an isolated animal organ), an extract of an organism or part of an organism and an exudate, or pure compound (e.g. alkaloids, coumarins, flavonoids, lignans, steroids and terpenoids) isolated from plants, animals or micro-organisms. The use of natural products, especially plants, for healing is an ancient and universal as medicine itself. Natural products have been an integral part of the ancient traditional medicine systems, e.g., Chinese, Ayurvedic, and Egyptian.
Even now, continuous traditions of natural product therapy exist throughout the third world, especially in the orient, where numerous minerals, animal substances, and plants are still in common use. This recent resurgence of interest in plant remedies has been spurred on by several factors: 6
- The effectiveness of plant medicines.
- The preference of consumers for natural therapies, a greater interest in alternative medicines, and a commonly held erroneous belief that herbal products are superior to manufactured products.
- Dissatisfaction with the results from synthetic drugs and the belief that herbal medicines might be effective in the treatment of certain diseases where conventional therapies and medicines have proven to be inadequate.
- The high cost and side effects of most modern drugs.
- Improvements in the quality, efficacy, and safety of herbal medicines with the development of science and technology.
- Patients’ belief that their physicians have not properly identified the problem; hence, they feel that herbal remedies are another option.
- A movement towards self-medication.
Medicinal plants are generally known as “Chemical Goldmines” as they contain natural chemicals, which are acceptable to human and animal systems. Of the 2,50,000 higher plant species on earth, more than 80,000 are medicinal.
The Red Data Book of India has 427 entries of endangered species of which 28 are considered extinct, 124 threatened, 81 vulnerable, 100 rare and 34 insufficiently known species 7.
The Origin, Scope, and Practice of Pharmacognosy: The history of herbal medicines is as old as human civilization. The documents, many of which are of great antiquity, revealed that plants were used medicinally in China, India, Egypt, and Greece long before the beginning of the Christian era. One of the most famous surviving remnants is Papyrus Ebers, a scroll some 60 feet long and a foot wide, dating back to the sixteenth century before Christ 8. Indians also, worked meticulously to examine and classify the herbs which they came across, into groups called Gunas. Charaka made fifty groups of ten herbs, each of which, according to him, would suffice an ordinary physician’s need.
Similarly, Sushrutha arranged 760 herbs in 7 distinct sets based on some of their common properties. A large portion of the Indian population even today depends on the Indian System of Medicine- Ayurveda, ‘An ancient science of life.’ The well-known treaties in Ayurveda are Charaka Samhita and Sushruta Samhita. The first pharmacist, Galen, was known to have had several pain-relieving materials, including opium in his apothecary 9.
Plant Profile:
Botanical name : Acacia arabica
Hindi name : Babul, Pankikar
Family : Fabaceae
Prosopis juliflora (Sw.) DC (Mimosaceae) commonly known as mesquite, is a shrub or small tree native to Mexico, South America, and the Caribbean. P. juliflora probably originates from Peru; it occurs naturally in dry areas of northern South America and Central America, Mexico and the southern USA. It has been introduced into many tropical areas, including Northeastern Brazil, Africa, Australia, Southeast Asia, and the Indian subcontinent. P. juliflora is xerophytic and is adapted to many soil types under a wide range of moisture conditions. The value of the tree lies in its exceptional tolerance of drought and marginal soils. It tolerates strongly saline soils and seasonal water-logging. P. juliflora has been planted successfully on soils with acid to the alkaline reaction. It is sometimes said to dry out the soil and compete with grasses, particularly in dry areas 10.
Chemical Constituents: Steroids, tannins, leucoanthocyanidin, and ellagic acid glycosides. A new monocyclic diketone, prosopidione, and two alkaloids, namely, juliprosinene and juliflorinine, have been isolated from the leaves 11.
Parts Used: Leaves, gum, bark, pods, and flowers.
FIG. 4: FLOWER OF PROSOPIS JULIFERA
Botanical name : Prosopis juliflora
Hindi : Kabuli kikar, angarajii
babul, vilayati babul
Family : Fabaceae
MATERIALS AND METHODS:
Materials, Instruments, and Chemicals: Plant materials, glass slide, grinding mixer, hot air oven, silica crucible, ashless filter paper (Whatman no. 44), Petri dish, stoppered conical flask, rotary flask shaker alcohol (95%), chloroform water, chloral hydrate solution, water.
Collection of Plant: The plant materials were collected from the Jhansi and Lucknow.
Authentication of Plant: The materials were authenticated at Indian Grassland and Fodder Research Institute, Jhansi, India. Sample specimens have been identified as Prosopis juliflora (SW.) DC. of the family Fabaceae.
Processing of Plant Material for Study: The materials for the final study were prepared by the following procedure:
Washing: Foreign material was identified and discarded through washing.
Drying: Plant material was dried in the shed to prevent decomposition of the chemical constituents.
Grinding: Material ground till homogeneous powder was formed.
Physiochemical Standardization: (According to Ayurvedic Pharmacopoeia of India):
Determination of Moisture Content (Loss on Drying): An excess of water in medicinal plant materials will encourage microbial growth, the presence of fungi or insects, and deterioration following hydrolysis. Limits for water content should, therefore, be set for every given plant material. This is especially important for materials that absorb moisture easily or deteriorate quickly in the presence of water.
Methodology: About 2 g of the prepared air-dried material was accurately weighed in a previously dried and tared Petri dish. The sample was distributed evenly and was placed in the drying chamber (Oven). Drying was carried out by heating to 100-105 °C; the Petri dish was removed from the oven and was kept in the desiccator and allowed to cool and then weighed. The experiment was repeated until two consecutive weighings did not differ by more than 5 mg unless otherwise stated in the test procedure. The loss in weight on drying was then calculated. The same procedure was repeated for the Acacia arabica and Prosopis juliflora Table 6.
Determination of Total Ash Value: The residue remaining after incineration is the ash content of the drug, which simply represents inorganic salts, naturally occurring in drug or adhering to it or deliberately added to it as a form of adulteration. Many a time, the crude drugs are admixed with various mineral substances like sand, soil, calcium oxalate, chalk powder, or other drugs with different inorganic contents. For determination of total ash, the powdered drug is incinerated to burn out all organic matter. Ash value is a criterion to judge the identity or purity of crude drugs. Total ash usually consists of carbonates, phosphates, silicates, and silica Table 7.
Methodology: 2 g of the powdered drug was accurately weighed in a tared silica crucible. The powdered drug was spread as a fine layer at the bottom of the crucible. The crucible was incinerated at a temperature not exceeding 450 °C until free from carbon. The crucible was cooled and weighed. The procedure was repeated until a constant weight was observed. The percentage of the total ash was calculated in triplicate concerning the air-dried drug. The same procedure was repeated for the A. arabica and Prosopis juliflora.
% Ash value was calculated by the formula:
2 g powdered drug contain = X g of ash
100 g powdered drug contain = 100 X/2 = 50X
X = difference in the weight
Determination of Total Acid Insoluble Ash Value: (Ayurvedic Pharmacopoeia of India 1989): Acid-insoluble ash, which is a part total ash insoluble in diluted hydrochloric acid is also recommended for natural drugs. Adhering dirt and sand may be determined by acid-insoluble ash contain.
Methodology: The ash obtained as described in the determination of total ash was boiled with 10% 25 ml of hydrochloric acid for 5 min. The insoluble ash was collected on an ashless filter paper by filtration, and it was washed with hot water. The insoluble ash was transferred into a tared silica crucible, ignited, cooled and weighed. The procedure was repeated until a constant weight was observed. The percentage of acid insoluble ash was calculated concerning the air-dried drug. The same procedure was repeated for Acacia arabica and Prosopis juliflora.
The formula used for calculation:
2 g powdered drug contain = X gram acid insoluble ash
100 g of powdered drug contain = 100X/2 = 50X
X = difference in the weight
Determination of Extractive Values: Extractive value is a measure of the content of the drug extracted by solvents. Extractive value can be water soluble and alcohol soluble Table 8.
Water Soluble Extractive:
Methodology: 2 g of previously weighed air-dried powdered material was taken in a glass-stoppered flask and macerated with 100 ml of chloroform water (1:99). It was shaken frequently for 6 h and then allowed to stand for 18 h. It was filtered rapidly, taking precautions against loss of the solvent. 10 ml of filtrate was evaporated to dryness in a tared flat-bottomed Petri dish in triplicates, dried at 105°C, cooled in a desiccator and weighed. The percentage of water-soluble extractive was calculated concerning air-dried drug. The same procedure was repeated for Acacia arabica and Prosopis juliflora.
The formula used for calculation:
10 ml of extract solution contain = X g extract
100 ml of extract solution contain = X100/10 =10X g extract
2 g powdered drug contain = 10X gram extract
100 g powdered drug contain= 10X × 100/2 g extract = 500X%
X = difference in pre-weight and final weigh
Alcohol Soluble Extractive:
Methodology: 2 g of previously weighed air-dried powdered material was taken in a glass-stoppered flask and macerated with 100 ml of ethanol. It was shaken frequently for 6 h and then allowed to stand for 18 h. It was filtered rapidly, taking precautions against loss of the solvent. 10 ml of filtrate was evaporated to dryness in a tared flat-bottomed petri dish in triplicates, dried at 105 °C, cooled in a desiccator and weighed. The percentage of ethanol-soluble extractive was calculated concerning air-dried drug. The same procedure was repeated for Acacia arabica and Prosopis juliflora.
Formula used for calculation:
10 ml of extract solution contain = X g extract
100 ml of extract solution contain = X 100/10 =10X g extract
2 g powdered drug contain = 10X g extract
100 g powdered drug contain = 10X × 100/2 g extract = 500X%
X = difference in pre-weight and final weigh
Determination of Total Sugars:
Reagent Used:
- 80% ethanol, 80% phenol, concentrated sulphuric acid.
- D-Glucose standard solution: (0.1 mg/ml) dissolve in 100 ml of 80% ethanol.
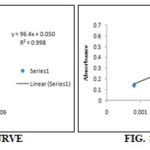
Methodology: 0.5 g powdered material was homogenated in 80% ethanol with the help of centrifuge at 2000 rpm for 15 min. The supernatant obtained is made up to known volume (generally up to 10 ml or depending on the expected concentration of sugar). Taken 0.2 ml aliquot, added 0.1 ml of 80% phenol and 5 ml conc. Sulphuric acid, then made the volume up to 10 ml with 80% ethanol, cooled in an ice bath. Total sugar was calculated by using D-Glucose (mg/ml) as the standard whose reading was y = 33.8x + 0.179, r2 = 0.985 at 490 nm using UV-1 Double beam spectrophotometer, where y was the absorbance and x the D-Glucose equivalent (mg/ml). The same procedure was repeated for Acacia arabica and Prosopis juliflora Table 1. 12
TABLE 1: PREPARATION OF CALIBRATION CURVE FOR SUGAR CONTENT. (STD. USED D-GLUCOSE)
| S.
no. |
Amount from stock (ml) | 80% phenol solution (ml) | Conc. sulphuric acid (ml) | Dist. water (ml) upto | Conc.
(mg/ml) |
Abs. at
490 nm |
| 1 | 0.1 | 0.1 | 5 | 10 | 0.001 | 0.218 |
| 2 | 0.2 | 0.1 | 5 | 10 | 0.002 | 0.238 |
| 3 | 0.3 | 0.1 | 5 | 10 | 0.003 | 0.281 |
| 4 | 0.4 | 0.1 | 5 | 10 | 0.004 | 0.322 |
| 5 | 0.5 | 0.1 | 5 | 10 | 0.005 | 0.345 |
| 6 | Blank | 0.1 | 5 | 10 |
Determination of Total Starch: Estimation of total starch in plant material was carried out according to 12 spectrophotometric methods.
Reagents Used:
- 80% ethanol, 80% perchloric acid, 80% phenol, concentrated sulphuric acid
- D-Glucose standard solution: (0.1 mg/ml) in 100 ml of distilled water.
Methodology: 0.5 g powdered material was homogenated in 80% ethanol with the help of centrifuge at 2000 rpm for 15 min. To the residue thus obtained, added 4 ml of distilled water, heated on the water bath for 15 min and macerated with the help of glass rod. To each of the samples, added 3 ml of 52% perchloric acid and centrifuged at 2000 rpm for 15 min. The supernatant thus obtained was made up to a known volume (generally up to 10 ml or depending on the expected concentration of starch). Taken 0.1 ml aliquot, added 0.1 ml of 80% phenol and 5 ml conc. Sulphuric acid made the volume up to 10 ml. Cooled and calculated total starch by using D-Glucose (mg/ml) as standard whose reading was y = 12.6x + 0.170, r2= 0.908, at 490 nm using UV-1 Double beam spectrophotometer, where y was the absorbance and x the D-Glucose equivalent (mg/ml). The same procedure was repeated for Acacia arabica and Prosopis juliflora Table 2.
TABLE 2: PREPARATION OF CALIBRATION CURVE FOR STARCH CONTENT. (STD. USED AS SOLUBLE STARCH)
| S.
no. |
Amount from stock (ml) | 80% phenol solution (ml) | Conc. sulphuric acid (ml) | Dist. water (ml) upto | Conc.
(mg/ml) |
Abs. at
490 nm |
| 1 | 0.1 | 0.1 | 5 | 10 | 0.001 | 0.185 |
| 2 | 0.2 | 0.1 | 5 | 10 | 0.002 | 0.197 |
| 3 | 0.3 | 0.1 | 5 | 10 | 0.003 | 0.209 |
| 4 | 0.4 | 0.1 | 5 | 10 | 0.004 | 0.211 |
| 5 | 0.5 | 0.1 | 5 | 10 | 0.005 | 0.241 |
| 6 | Blank | 0.1 | 5 | 10 |
Determination of Total Tannins: Estimation of tannin percentage in the plant material was carried out according to the method described in AOAC 13.
Reagents Used:
- Saturated sodium carbonate solution: It was prepared by adding 35 g anhydrous sodium carbonate to every 100 ml distilled water, dissolved it at 70-80 ºC and get cool overnight, filtered through glass wool.
- Tannic acid standard solution: (0.1 mg/ml) dissolve 10 mg tannic acid in 100 ml of distilled water.
- Folin and Ciocalteu’s phenol reagent.
Preparation of Standard Curve: Standard curve was prepared using tannic acid as standard (10 mg tannic acid in 100 ml of distilled water).
Methodology: Extracted 2 g powdered plant material with 100 ml distilled water by boiling on a water bath for 6-8 h, filtered and made up the volume to 100 ml in the volumetric flask. Took 1 ml aliquot of it, added 5 ml Folin & Ciocalteu’s reagent, 10 ml saturated sodium carbonate and made the volume up to 100 ml in a volumetric flask. The instrument was calibrated through blank and took the corresponding absorbance of different samples, total tannin content calculated by using y = 0.091x + 0.065, r2 = 0.998, at 760 nm, using UV-1 Double beam spectrophotometer, where y was the absorbance and x the tannic acid equivalent (mg/ml). The same procedure was repeated for Acacia arabica and Prosopis juliflora Table 3.
TABLE 3: PREPARATION OF CALIBRATION CURVE FOR TANNIN CONTENT (STD. USED AS TANNIC ACID)
| S. no. | Amount from stock (ml) | Folin- Ciocalteu’s phenol reagent(ml) | Saturated sodium carbonate solution (ml) | Distilled water
(ml) Upto |
Conc.
(mg/ml) |
Abs. at
760 nm |
| 1 | 1 | 5 | 10 | 100 | 0.001 | 0.152 |
| 2 | 2 | 5 | 10 | 100 | 0.002 | 0.238 |
| 3 | 3 | 5 | 10 | 100 | 0.003 | 0.332 |
| 4 | 4 | 5 | 10 | 100 | 0.004 | 0.444 |
| 5 | 5 | 5 | 10 | 100 | 0.005 | 0.531 |
| 6 | Blank | 5 | 10 | 100 |
Determination of Total Phenolics: Total phenols estimation can be carried out with Folin-Ciocalteu reagent (FCR) 14.
Principle: Phenols react with an oxidizing agent phosphomolybdate in Folin-Ciocalteu reagent under alkaline conditions and result in the formation of a blue colored complex, the molybdenum blue which is measured at 650 nm colorimetrically.
Reagents:
- Folin-Ciocalteus reagent (FCR)
- 20% Na2CO3
- Standard (10 mg gallic acid in 100 ml of methanol).
Methodology: Prepare a stock solution (1mg/ml) of extract in methanol. From the stock, solution take a suitable quantity of the extract into 25 ml volumetric flask and add 10 ml of water and 1.5 ml of Folin Ciocalteus reagent, keep the mixture for 5 min and then add 4 ml of 20% Na2CO3 and makeup to 25 ml with distilled water.
Keep the mixture for 30 min and record absorbance at 765 nm. Total phenolic content was calculated as Gallic acid (mg/ml) using the following equation based on the calibration curve: y = 131.8x + 0.044, r2 = 0.997, where y was the absorbance and x was the Gallic acid equivalent (mg/ml). The same procedure was repeated for Acacia arabica and Prosopis juliflora Table 4.
TABLE 4: PREPARATION OF CALIBRATION CURVE FOR PHENOLICS CONTENT
| S. no. | Amount from stock (ml) | Dist. water (ml) | Folin- Ciocalteu’s phenol reagent(ml) | 20% sodium carbonate solution (ml) | Dist. water
(ml) upto |
Conc.
(mg/ml) |
Abs. at 765 nm |
| 1 | 0.2 | 10 | 1.5 | 4 | 25 | 0.0008 | 0.139 |
| 2 | 0.4 | 10 | 1.5 | 4 | 25 | 0.0016 | 0.264 |
| 3 | 0.6 | 10 | 1.5 | 4 | 25 | 0.0024 | 0.369 |
| 4 | 0.8 | 10 | 1.5 | 4 | 25 | 0.0032 | 0.467 |
| 5 | 1 | 10 | 1.5 | 4 | 25 | 0.0040 | 0.565 |
| 6 | Blank | 10 | 1.5 | 4 | 25 |
Determination of Total Flavonoids: Total flavonoids were estimated using the method, used to estimate total flavonoid contents of the extract solution based on the formation of a complex flavonoid-aluminum 15.
Extract Preparation: 2 g of dried powdered material of all the parts were cold percolated with a known volume of methanol.
Reagents Used:
- 2 % AlCl3 (ethanolic solution)
- Standard (10 mg rutin in 100 ml. of ethanol)
FIG. 9: CALIBRATION CURVE
Methodology: To 0.5 ml of sample (methanolic extract), 0.5 ml of 2% methanolic AlCl3 solution was added. Standard solutions were prepared in the same way by using 0.2, 0.4, 0.6, 0.8, and 1 ml. of stock solution (quercetin solution).
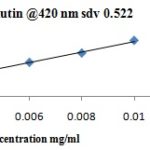
Yellow color indicated the presence of flavonoids. Blank was prepared by using 0.5 ml. 2 % methanolic AlCl3. All the solutions were made up to 5 ml with methanol and after 1 h absorbance of standard and sample solutions were recorded at 420 nm against a blank. Total flavonoid content was calculated as rutin (mg/ml) using the following equation based on the calibration curve: y = 165.1X + 0.069, r2 = 0.998, where y was the absorbance and x was the rutin equivalent (mg/ml). The same procedure was repeated for Acacia arabica and Prosopis juliflora Table 5.
TABLE 5: PREPARATION OF CALIBRATION CURVE FOR FLAVONOID CONTENT
| S.
no. |
Amount from
stoke (ml) |
2% Aluminium chloride solution (ml) | Dist. Water
(ml) upto |
Conc.
(mg/ml) |
Abs. at
420 nm |
| 1 | 0.2 | 0.5 | 10 | 0.002 | 0.401 |
| 2 | 0.4 | 0.5 | 10 | 0.004 | 0.739 |
| 3 | 0.6 | 0.5 | 10 | 0.006 | 1.065 |
| 4 | 0.8 | 0.5 | 10 | 0.008 | 1.354 |
| 5 | 1 | 0.5 | 10 | 0.010 | 1.745 |
| 6 | Blank | 0.5 | 10 |
RESULTS:
Physicochemical Analysis:
TABLE 6: MOISTURE CONTENT
| Observation | Acacia arabica (Leaves) | Acacia arabica (Bark) | Acacia arabica (Twig) | Prosopis juliflora (Leaf) | Prosopis juliflora (Bark) | Prosopis juliflora (Twig) |
| Range % | 3.90-4.2% | 4.90-5.40% | 5.2-5.7% | 4.6-4.1% | 4.2-4.5% | 5.5-5.3% |
| Average % | 3.95% | 5.20% | 5.45% | 4.75% | 4.35% | 5.4% |
TABLE 7: ASH VALUE
| Species and
Parts |
Total ash
(%) |
Average
(%) |
Acid-insoluble ash (%) | Average (%) | Water soluble ash (%) | Average
(%) |
| A. arabica (leaf) | 9.25-9.55 | 9.4 | 1.13-1.25 | 1.21 | 0.855-0.895 | 0.870 |
| A. arabica (bark) | 8.00-8.10 | 8.05 | 1.19-1.045 | 1.033 | 0.842-0.888 | 0.865 |
| A. arabica (twig) | 7.5-7.9 | 7.75 | 0.999-1.35 | 1.0746 | 0.865-0.840 | 0.852 |
| P. juliflora (leaf) | 8.00-8.20 | 8.1 | 1.26-1.55 | 1.41 | 0.752-0.782 | 0.767 |
| P. juliflora (bark) | 8.20-8.30 | 8.25 | 1.02-1.15 | 1.085 | 0.768-0.807 | 0.787 |
| P. juliflora (twig) | 8.35-8.40 | 8.4 | 0.930-1.10 | 1.037 | 0.812-0.853 | 0.832 |
TABLE 8: TOTAL PHENOLIC CONTENT
| Species and parts | Phenolic content (%) | Average (%) | |
| A. arabica (leaf) | 0.0405-0.0406 | 0.04055 | |
| A. arabica (bark) | 0.0400-0.0402 | 0.0401 | |
| A. arabica (twig) | 0.0328-0.0348 | 0.0338 | |
| P. juliflora (leaf) | 0.0406-0.0408 | 0.047 | |
| P. juliflora (bark) | 0.825-0.00851 | 0.00838 | |
| P. juliflora (twig) | 0.0138-0.140 |
0.01392 |
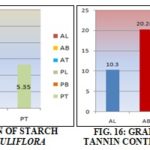
TABLE 9: TOTAL TANNIN CONTENT
| Species and parts | Tannin content (%) | Average (%) |
| A. arabica (leaf) | 10.2 5-10.35 | 10.30 |
| A. arabica (bark) | 20.25-20.32 | 20.28 |
| A. arabica (twig) | 22.42-22.53 | 22.47 |
| P. juliflora (leaf) | 5.38-5.40 | 5.39 |
| P. juliflora (bark) | 10.65-10.88 | 10.774 |
| P. juliflora (twig) | 15.49-15.62 | 15.51 |
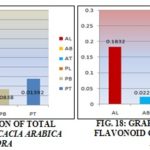
TABLE 10: TOTAL FLAVONOID CONTENT
| Species and parts | Flavonoid content (%) | Average (%) |
| A. arabica (leaf) | 0.1822-0.1842 | 0.1832 |
| A. arabica (bark) | 0.0220-0.0235 | 0.02275 |
| A. arabica (twig) | 0.030-0.0340 | 0.0270 |
| P. juliflora (leaf) | 0.2412-0.2436 | 0.2424 |
| P. juliflora (bark) | 0.05336-0.05454 | 0.05390 |
| P. juliflora (twig) | 0.00672-0.00680 | 0.00675 |
TABLE 11: TOTAL STARCH CONTENT
| Species and parts | Starch content (%) | Average (%) |
| A. arabica (leaf) | 3.68-3.96 | 3.75 |
| A. arabica (bark) | 4.14-4.46 | 4.30 |
| A. arabica (twig) | 9.9-10.65 | 10.34 |
| P. juliflora (leaf) | 4.14-4.34 | 4.24 |
| P. juliflora (bark) | 4.8-5.2 | 5.0 |
| P. juliflora (twig) | 5.25-5.45 | 5.35 |
TABLE 12: TOTAL SUGAR CONTENT
| Species and parts | Suger content (%) | Average (%) |
| A. arabica (leaf) | 6.17-6.95 | 6.56 |
| A. arabica (bark) | 2.24-2.62 | 5.65 |
| A. arabica (twig) | 2.10-2.18 | 2.14 |
| P. juliflora (leaf) | 11.45-11.71 | 11.58 |
| P. juliflora (bark) | 11.17-11.99 | 11.68 |
| P. juliflora (twig) | 8.2-9.0 | 8.6 |
Phytochemical Analysis:
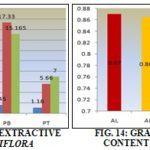
TABLE 13: EXTRACTIVE VALUE
| Species and
parts |
Hexane soluble (%) | Average (%) | Alcohol soluble (%) | Average
(%) |
Water soluble (%) | Average (%) |
| A. arabica (leaf) | 5.9-6.1 | 6 | 10.66-11.00 | 10.83 | 11.13-11.44 | 11.33 |
| A. arabica (bark) | 4.14-4.3 | 4.2 | 15.51-15.83 | 15.55 | 8.98-9.35 | 9.165 |
| A. arabica (twig) | 1.06-1.22 | 11.4 | 2.64-3.10 | 2.87 | 6.02-6.36 | 6.19 |
| P. juliflora (leaf) | 6.38-6.66 | 6.57 | 11.85-12.15 | 12.0 | 18.26-18.56 | 18.416 |
| P. juliflora (bark) | 1.53-1.86 | 1.645 | 17.83-18.16 | 17.33 | 15.10-15.23 | 15.165 |
| P. juliflora (twig) | 1.16-1.16 | 1.16 | 5.5-5.8 | 5.66 | 6.8-7.2 | 7.0 |
DISCUSSION:
Physicochemical Parameters: Determination of various physicochemical constants were carried out according to the methods provided in Ayurvedic Pharmacopoeia of India (API) as well as in WHO guidelines.
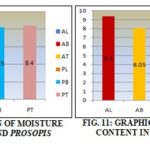
Total Moisture Content: Result shows that the leaf of Acacia arabica had maximum moisture content followed by stem bark, and twig, while Prosopis juliflora twig had maximum moisture content followed by bark and twig. According to the results tabulated in the table.
Total Ash Value, Water Soluble Ash and Acid Insoluble Ash: Result obtained from the study shows that the leaf of Acacia arabica has more percentage of Ash content followed by stem bark and twig. But in Prosopis juliflora twig contain maximum percentage followed by stem bark and twig. Leaf of Acacia arabica has a maximum percentage of ash content, as per the results tabulated in the table.
Total Extractive Value: The results of total extractive values shows that twig of Acacia arabica contains large percentage of hexene soluble extract followed by leaf and bark while leaf of Prosopis juliflora has maximum followed by bark and twig, the maximum percentage in Prosopis juliflora leaf and minimum twig.
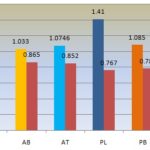
FIG. 12: GRAPHICAL PRESENTATION OF ACID INSOLUBLE AND WATER SOLUBLE ASH CONTENT IN ACACIA ARABICA AND PROSOPIS JULIFLORA. Leaf of Acacia arabica and Prosopis juliflora have maximum water soluble Ash content followed by bark and twig. Twig of Prosopis juliloera has maximum acid insoluble ash content followed by stem bark and twig. While leaf of Acacia arabica has maximum acid insoluble ash content followed by stem bark and twig.
But in ethanol both stem bark have maximum content followed by leaf and twig, Prosopis juliflora stem bark has maximum ethanol extract content and minimum in twig. But in water-soluble extract, the leaf contains more percentage of extract followed by stem bark, and twig and according to the results tabulated in the table.
Total Sugar Content: Results obtained from the present study shows the level of sugar in the 80% ethanolic extracts of all the parts. According to the data shown in table leaf of Acacia arabica has maximum sugar content followed by stem bark and twig.
While twig of Prosopis juliflora has maximum percentage followed by stem bark and twig. Acacia arabica leaf has a larger amount of sugar content, and Prosopis juliflora leaf has minimum content.
Total Starch Content: Results obtained from the present study shows the level of starch in the 80% ethanolic extracts of all the parts. A. arabica and P. juliflora both twig contain a maximum percentage of starch, and both leaves contains a minimum percentage. According to the data shown in table, twig contains a larger amount of starch and leaf contain a minimum amount of starch.
Total Tannin Content: Results obtained from the present study shows the level of tannins in the water extract of the various parts. Twig of both Acacia arabica and Prosopis juliflora contains a higher amount of tannin percentage followed by twig and stem bark lower amount of tannin content in leaf. Acacia arabica twig contains maximum while Prosopis juliflora leaf contains a minimum percentage of tannin content, according to the results tabulated in the table.
Total Phenolics Content: Results obtained from the present study shows the level of phenolic compound in the methanolic extract of the various parts. According to the results, Acacia leaf contain the highest amount of phenols followed by stem bark and twig, and Prosopis leaf also contains the highest amount followed by twig and bark. Prosopis leaf has a maximum percentage, and bark contain the lowest amount. According to the result tabulated in the table.
Total Flavonoid Content: Results obtained from the present study shows the level of flavonoids in the methanolic extract of the various parts. Total flavonoid content was much higher in the leaf followed by stem bark, and twig. Leaf of P. juliflora contains the maximum amount of flavonoid and twig contain minimum. According to the result tabulated in the table.
CONCLUSION: It is concluded that a given plant is A. arabica and P. juliflora. I have done the comparative pharmacognostical study between A. arabica and P. juliflora and conclude that A. arabica plays a more significant role and has a more scientific value. The present study was aimed at pharmacognostical study. Plants A. arabica and P. juliflora were studies for pharmacognostical, physi-chemical, parameters which can be have utilized in identification and authentication of plants.
ACKNOWLEDGEMENT: The authors thankful with our deepest core of heart to Mr. Vikram Singh (Assistant Professor), for his valuable guidance.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Bijauliya RK, Alok S, Jain SK Singh VK, Singh D and Singh M: Herbal and allopathic medicine for kidney, gallbladder and urinary stones: a review. Int J Pharm Sci Res 2017; 8(5): 1935-52. doi: 10.13040/IJPSR.0975-8232. 8(5).1935-52.
- Chanchal DK, Niranjan P, Alok S, Singh S and Saurabh: An update on Ayurvedic Herb Kachhnar (Bauhinia purpurea)- A review. Int J Pharmacognosy 2015; 2(8): 381-90:.doi link: http://dx.doi.org/10.13040/IJPSR 0975-8232.IJP.2(8).381-90.
- Mukherjee PK: Exploring Botanicals in Indian System of Medicine-Regulatory Perspectives, Vol. 20, 2003: 249-264.
- Smith G: Herbs in medicine Triple Helix Autumn 04, 12.
- Raghavendra1 MP: Alkaloid extracts of Prosopis juliflora (Sw.) DC. (Mimosaceae) against Alternaria alternate. Journal of Biopesticides 2009; 2(1): 56-59.
- Wickramasinghe and Bandaranayake M: Quality control, screening, toxicity, and regulation of Herbal drugs, modern phytomedicine. Turning medicinal plants into drugs, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2006.
- Thomas J: Medicinal and aromatic plants research in India. In UNDP. 1997. Proc. Training course on Industrial Exploitation of Indigenous Medicinal and Aromatic Plants. Beijing, China, 1997.
- Kokate CK: Pharmacognosy’ Nirali Prakashan, Edition 37th, 2008: 105-120, 1-3.
- Agrawal SS and Paridhavi M: Herbal drug technology; Universities Press (India) Private Limited 2007: 1-5.
- Chanchal DK, Niranjan PS, Alok S and Rashi S: Evaluation of Macroscopical and Microscopical Study, Phytochemical Analysis, TLC and HPTLC Fingerprinting of Bauhinia purpurea Leaves. Int J Pharm Sci Res 2016; 7(8): 3539-44.
- Wickramasinghe and Bandaranayake M: Quality control, screening, toxicity, and regulation of Herbal drugs, modern phytomedicine. Turning medicinal plants into drugs, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2006.
- Montgomery R: Determination of glycogen. Arch Biochem Biophys 1957; 67: 378-386.
- Anonymous: Official methods of analysis of the association of Official Analytical Chemists (AOAC), Virginia, US 1984.
- Bray HC and Thorpe WV: Analysis of phenolic compounds of interest in metabolism. Meth Biochem Analysis 1954; 1: 27-52.
- Ez Ordon LAA: Antioxidant activities of Sechium edule (Jacq.) Swart extracts 2006; 97: 452-458621-1623.
How to cite this article:
Nigam S, Singh V, Dongray A and Chanchal DK: Comparative study on physio-chemical parameters of Acacia arabica & Prosopis julifera. Int J Pharmacognosy 2018; 5(11): 705-16. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.5(11).705-16.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
3
705-716
1,051
651
English
IJP
S. Nigam *, V. Singh, A. Dongray and D. K. Chanchal
College of Pharmacy, SRGI Ambabai, Jhansi, Uttar Pradesh, India.
sarikabbd@gmail.com
12 September 2018
13 October 2018
23 October 2018
10.13040/IJPSR.0975-8232.IJP.5(11).705-16
01 November 2018