ANTIMICROBIAL ACTIVITY OF BRAZILIAN HONEY AND ALOE VERA
HTML Full TextANTIMICROBIAL ACTIVITY OF BRAZILIAN HONEY AND ALOE VERA
K. A. Almeida * 1, L. R. Tiso 2, T. N. Cunha 3 and E. L. Costa 3
16 Brechin Court 1, Greenvale, VIC, 3059, Australia.
Rua Maria Cristina 2 507-1, Jd. Casqueiro, CEP 11553-160, Cubatão, São Paulo, Brazil.
Av. Conselheiro Nébias 3, 300, Vila Matias, CEP 11015-002, Santos, São Paulo, Brazil.
ABSTRACT: Honey is commonly used by the indigenous population in Brazil due to its large variety of physiological properties such as ulcers and dermatitis healing. The goal of this paper is to select and to test different types of Brazilian honey to be used associated with Aloe vera as an accessible wound treatment. Three types of honey were analyzed: two produced by Tetragonisca angustula in different regions of Brazil and one honey produced by Apis mellifera scutellata, associated with both water and Aloe vera. Quantitative assays, where bacterial growth curve was analyzed by optical density, were performed to test the antimicrobial activity of the samples. From the results obtained, we observed that the three types of honey studied presented a satisfactory antimicrobial activity. The association between honey and Aloe vera, besides demonstrating better antimicrobial action than each compound itself, it demonstrated better effectiveness against Enterobacteriaceae species (E. coli and S. typhimurium).
| Keywords: |
Aloe vera, Antimicrobial activity, Medicinal honey, Tetragonisca angustula, Apis mellifera scutellata
INTRODUCTION: Honey is one of the oldest medicinal substances of history. In ancient Egypt, honey was found in around 500 of the 900 drugs existing at that time 1, 2. The composition of honey depends on both the bee species and the plant’s nectar. Climatic conditions and beekeeper’s handling can also influence honey’s characteristics 3. Honey is composed of different sugars, mostly monosaccharides, and disaccharides. It also contain flavonoids 4, proteins, amino acids, enzymes, organic acids, minerals, pollen 2, 5, proteins, amino acids, enzymes, organic acids, minerals, pollen 5, 6 and hydrogen peroxide, which is considered a factor for its antimicrobial activity 4, along with the high osmolarity of the product 4, 5.
As it is a high nutritional value product, it is used to treat and prevent physical and mental fatigue. Moreover, it is claimed to contain properties that assist with the treatment of ulcers 4, and it shows anti-inflammatory activity 6. Honey can be classified by its origin, therefore, it is divided into two categories: floral honey or honeydew. Floral honey is obtained by the bee’s feeding on the nectar of the flowers, and it can be subdivided into either monofloral or poly floral. Monofloral honey is composed by bee’s feeding on flowers from the same family, genus or species only, and poly floral honey is obtained by bee’s feeding on several different varieties of flowers. On the other hand, honeydew is constituted from a nectar sucker insect’s excretion 7.
In Brazil, Tetragonisca angustula and Apis mellifera scutellata bee’s honey are popularly known and employed due to its medical properties, such as hydrating, antioxidant, anti-seborrheic, anti-dandruff, anti-psoriasis, anti-inflammatory, anti-microbial, wound healing, and others, 4, 6, 7 being an accessible alternative to Manuka honey, a therapeutical New Zealand honey commonly used for the treatment of wounds 4. Additionally, according to Gupta et al., bandages containing honey accelerates wound resolution, to make wounds sterile in less time and present better healing results in hypertrophic and burning wounds when compared to silver sulfadiazine dressing 8.
Phytotherapic drugs constituted by honey are largely used in several places around the globe. The main use of these associations is the treatment of cough and respiratory tract issues. Recently, honey is being associated with Aloe vera, a plant used as a medicine for over 1000 years for the treatment of wounds and burns due to its hydrating, anti-inflammatory, antimicrobial and scaring activity 9. The scaring activity of Aloe vera is a result of manose-6-phosphate, present in its pulp. Functional polysaccharides of Aloe vera promotes fibroblast proliferation 10, angiogenesis and hyaluronic acid and hydroxyproline production; substances that are important on extracellular matrix remodeling during wound scarring 11.
Clinical trials performed on humans proved the efficacy of Aloe vera in the treatment of superficial and deep burn wounds. Trials performed on mice by Tabandeh et al., showed that isolated polysaccharide from Aloe vera induced formation of metallopeptidase matrix (MMP-3) and increased TIMP2 (metallopeptidase inhibitor) expression during skin wound resolution 12. Aloe vera’s anti-inflammatory activity is a result of direct inhibition of cyclooxygenase metabolic path and reduction of prostaglandin E2 production, which presents an important role in inflammatory processes. In the study performed by Tomasin and Gomes-Marcondes, the association between honey and A. vera reduced growth, proliferation and increased apoptosis of induced tumors in Wistar rat 9.
Thus, the goal of this study is to evaluate the antimicrobial activity of Brazilian honey from different origins produced by different species of bees, furthermore to verify its synergetic potential when associated with Aloe vera.
MATERIALS AND METHODS: This experiment took place in 2016 at the Scientific and Technological Research Institute, Catholic University of Santos, Santos, Brazil. The honey samples used in this study were acquired by purchase. The first sample was produced by Tetragniscaangustula bee, in the Atlantic Forest, situated in the south region o Brazil (T-AF); the second one was produced by the same bee species but in the Pantanal Forest, midwest region of Brazil (T-MP); the last sample was produced by Apis mellifera scutellata, from Mimosa scabrella tree secretion, also in the Atlantic Forest, in the south of Brazil Atlantic forest (A-AF).
The bacterial strains used in this study were: Escherichia coli (ATCC 25922), Staphylococcus aureus (ATCC 6538), Pseudomonas auruginosa (ATCC 27853) and Salmonella typhimurium (ATCC 14028). The strains were obtained commercially in the form of impregnated paper discs and, posteriorly activated by 18 h after incubation in 10 ml of brain heart infusion (BHI) each, until reaching a turbidity corresponding to 4 in MacFarland turbidity standard.
To analyze bacterial growth by optical density, each honey sample was analyzed in three different concentrations (50%, 30% and 10% of the honey in distilled water) for each bacterial strain. Samples containing each type of honey in different concentrations were added to tubes containing the different bacteria. The tubes were incubated under constant agitation on 36 ºC in a Marconi MA830 incubator. The optical density of the samples was measured every 1 hour, during 6 h and after 24 h the first measurement, in a spectrophotometer (Brand: La Motte, Model: Smart Spectro), on 600nm wavelength.
Tetragonisca angustula honey (T-AF) from the Atlantic Forest of the South of Brazil, the honey type that presented the best bacterial inhibition results, was used for the elaboration of a compound with Aloe vera barbadensis pulp gel in the concentrations of 50% and 25% Aloe vera-honey (v/v). The Aloe vera pulp gel was prepared by extraction and homogenization of the leaf parenchyma.
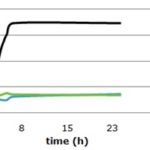
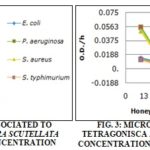
RESULTS AND DISCUSSION: This study showed that every honey tested presented antimicrobial activity, independently to its origin. Fig. 1 shows the behavior profile of Escherichia coli in relation Apis mellifera scutellata honey from the Atlantic Forest, South of Brazil Atlantic forest (A - AF) in different concentrations. At the concentration of 10% of honey, E. coli left the latency (Lag) phase after, approximately, 3.5 h of incubation. The beginning of exponential (Log) phase can be defined as the period when optical density corresponding to the double of the initial value. Due to the low concentration of inhibition agent, the bacteria reached the stationary phase after 5 h of incubation. However, when the honey is at concentrations of 30% and 50%, E. coli was not able to reach exponential phase, which means it prevented bacterial growth at those concentrations. Moreover, when a concentration of 50% of honey, there was an initial decrease in the bacteria population, that later returned to initial values after approximately 6 h. Fig. 1 also shows a dose-response effect, although it is not linear behavior, can be easily interpreted based on the bacterial growth velocity, as seen in Fig. 2, 3 and 4 that shows the microbial growth with the three kinds of honey studied: A-AF, T-AF, and T-MP honey, respectively, for E. coli, Salmonella typhimurium, S. aureus, and P. aeruginosa.
FIG. 1: ESCHERICHIA COLI GROWTH CURVE IN RELATION APIS MELLIFERA SCUTELLATA HONEY (A-AF) AT DIFFERENT CONCENTRATIONS
Apis mellifera scutellata honey (A-AF) showed to be more efficient against P. aeruginosa and S. aureus than for the Enterobacteriaceae micro-organisms tested. A-AF honey showed anti-microbial activity against P. aeruginosa and S. aureus with honey at the concentration of 10%, 30%, and 50%. For E. coli and S. typhimurium, honey at the concentration of 30% and 50% were efficient to reduce these bacteria’s speed of multiplication, preventing them from reaching Log phase, even after 24 h of incubation.
T-AF and A-AF honey, both from the same region, showed similar inhibition results as shown in Fig. 3, therefore the dose-response effect for S. aureus and P. aeruginosa is more evident and the honey at 10% didn't cause inhibition of Enterobacteriaceae microorganisms. T-PM honey, probably for presenting different flavonoid compounds, even in the concentration of 10%, showed an inhibitory effect over the Enterobacteriaceae used in this study. Resulting in similar value to the ones obtained for Pseudomonas and S. aureus.
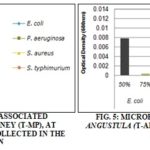
According to Tabandeh MR et al., 21, Aloe vera is an effective antimicrobial agent against M. smegmatis, K. pneumoniae, E. faecalis, M. luteis, C. albincans and B. sphericus, although it has no effect against E. coli, Staphylococcus aureus, Salmonella typhimurium and Pseudonomas auruginosa, microorganisms that are commonly found in necrotic or non necrotic wounds. According to the results presented in Fig. 5, it is evident that the speed of microbial growth in the presence of honey associated to Aloe vera, is possible to see a synergetic effect between both substances, with an increase of antimicrobial activity for three of the four microorganisms tested.
Two different concentrations were tested: 50% (honey: Aloe, v/v) and 75% (honey: Aloe, v/v). Multiplication speed of E. coli was reduced in approximately 25 times with the employment of solution in 75%, for P. aerurginosa, the solution in 75% (honey: Aloe vera, v/v) reduced the speed of multiplication in 6,4 times compared to the solution in 50% (honey: Aloe vera, v/v). For S. typhimurium, bacterial multiplication was lowered by approximately half when exposed to the solution in 75% (honey: Aloe vera, v/v). For S. aureus there was no alteration on antimicrobial activity in the different Aloe vera concentration, maintaining its low bacterial multiplication speed.
CONCLUSION: The tested honey at the concentration of 30% presented a similar effect as at the concentration of 50%, a positive factor for a suggested phytotherapic economic viability. The elaboration of an association Aloe vera and T. angustula honey compound, besides demonstrating better antimicrobial action than each compound itself, it demonstrated better effectiveness against Enterobacteria species (E. coli and S. typhimurium).
From the results obtained, we noticed that the three types of honey studied presented a satisfactory antimicrobial activity when associated with water or Aloe vera and that the antimicrobial activity varies according to honey concentration.
ACKNOWLEDGEMENT: The authors are grateful for the financial support given to this project by the Scientific and Technological Research Institute, Catholic University of Santos, Santos, Brazil and The Brazilian National Council for Scientific and Technological Development (CNPq) for the student fellowship. (Grant Number <147130/2015-8>)
CONFLICT OF INTEREST: The authors report no conflict of interest.
REFERENCES:
- Couto RHN and Couto LA: Apicultura: manejo e produtos. Funep, Edition 3rd, 2006; 193.
- Gouveia MC, Silva JBA, Mesquita LX and Maracajá PB: As análises de mel: Revisão. Revista Caatinga 2009; 22: 7-14.
- Anacleto DDA, Souza BDA, Marchini LC and Moreti ACD: Composition of the honey of samples originated from Jataí bees (Tetragonisca angustula Latreille, 1811). Food Science and Technology 2009; 29: 535-541.
- Vandamme L: Honey in modern wound care: a systematic review. Burns 2013; 39: 1514-1525.
- Bangroo AK, Khatri R and Chauhan S: Honey dressing in pediatric burns. Journal of Indian Association of Pediatric Surgeons 2005; 10: 3-172-175.
- Hadagali MD and Chua LS: The antiinflammatory and wound healing properties of honey. Eur Food Res Technol 2014; 239: 1003-1014.
- Ministério da Agricultura, Pecuária e Abastecimento. Instrução Normativa n. 11, de 20 de outubro de 2000. Regulamento técnico de identidade e qualidade do mel. http://sistemasweb.agricultura.gov.br/ sislegis/action/detalhaAto.do?method=consultar. Legislacao Federal.
- Finco FDB, Moura LL and Silva IG: Propriedades físicas e químicas do mel de Apis mellifera Ciênc e Tecnol de Aliment 2010; 30: 706-712.
- Gupta SS, Singh O, Bhagel PS, Moses S, Shukla S and Mathur RK: Honey dressing silver sulfadiazine dressing for wound healing in burn patients: A retrospective study. J Cutan Aesthet Surg 2011; 4: 183-7.
- Tomasin R and Gomes-Marcondes MCC: Oral administration of vera and honey reduces walker tumor growth by decreasing cell proliferation and increasing apoptosis in tumor tissue. Phytotherapy Research 2011; 25: 619-23.
- Danof IE and McAnalley W: Stabilised vera: Effect on human skin cells. Drug Cosmetic Ind, 1983; 133: 105-106.
- Subramanian S, Kumar DS and Arulselvan P: Wound healing potential of Aloe vera leaf gel studied in experimental rabbits. Asian J Biochem2006; 1: 178-185.
- Tabandeh MR, Oryan A and Mohammadalipour A: Polysaccharides of Aloe vera induce MMP-3 and TIMP-2 gene expression during the skin wound repair of the rat. International Journal of Biological Macromolecules 2014; 65: 424-430.
How to cite this article:
Almeida KA, Tiso LR., Cunha TN and Costa EL: Antimicrobial activity of Brazilian honey and Aloe vera. Int J Pharmacognosy 2018; 5(7): 414-18. doi link: http://dx.doi.org/10.13040/IJPSR.0975-8232.IJP.5(7).414-18.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.