A COMPARATIVE PHYTOCHEMICAL APPROACH FOR SUBSTITUTION OF STEM BARK WITH SMALL BRANCHES IN BUCHANANIA LANZAN FOR MEDICINAL USE
HTML Full TextA COMPARATIVE PHYTOCHEMICAL APPROACH FOR SUBSTITUTION OF STEM BARK WITH SMALL BRANCHES IN BUCHANANIA LANZAN FOR MEDICINAL USE
Bhavana Srivastava * 1, Ravindra Singh 2, Vandana Bharthi 1, A. K. Meena 3 and Om Prakash 1
Regional Ayurveda Research Institute for Drug Development 1, Gwalior - 474009, Madhya Pradesh, India.
Central Council for Research in Ayurvedic Sciences 2, New Delhi - 110058, Delhi, India.
Captain Srinivas Murthy Regional Ayurveda Drug Development Institute 3, Chennai - 600106, Tamil Nadu, India.
ABSTRACT: Overexploitation of many traditional medicinal plants like Buchanania lanzan Spreng. has become a threat and shortly, many medicinal plants may be unavailable for use in the medicinal industry. The present study outlines the concept of plant part substitution. Stem bark and small branches of B. lanzan are compared based on physicochemical analysis, phytochemical analysis, total phenolic content, total flavonoid content and High-Performance Thin Layer Chromatography (HPTLC) to evaluate the possibilities of using small branches in place of stem bark. Results suggest that small branches of this plant may be recommended in place of its stem bark after comparison and confirmation of same for pharmacological activities. The study provides the base for further study to use small branches of B. lanzan Spreng. as a substitute for its stem bar.
| Keywords: |
Buchanania lanzan Spreng, Plant part substitution, Phytochemical analysis, HPTLC profile
INTRODUCTION: Majority of the population believes in traditional medicines because of easy availability and lesser side effects. But despite their importance, medicinal plants are misused with no concern for their conservation. Unrestricted collection and destructive harvesting of medicinal plants are resulting in an over-exploitation of natural resources, due to which many useful medicinal plant species are becoming endangered or threatened. In the traditional system of medicines, most of the medicinal plants are slow growing trees, bulbous and tuberous plants, with bark, heartwood and underground parts being the parts mainly utilized.
Destructive harvesting of these plant parts is leading to the reduction of their natural populations and is of great concern for resource managers. Studies have shown that forest trees are highly susceptible to excessive exploitation, mainly because the mature bark is the commonly used plant part. Bark forms a physical barrier that prevents injury to the inner portion of the tree. It keeps pathogens and insects away while holding in moisture and nutrients.
The debarked tree has a very limited chance of survival, especially if the ring barking is done by unskilled collectors. This factor is causing problems in their sustainable supply. The probable approach to come out from this problem are to set up conservation areas, put into effect laws against collecting bark, large scale cultivation and to promote healers to collect and use alternative vegetative renewable plant parts such as leaves, young stems and fruits in place of bark and underground parts like root, rhizome etc. The last suggestion, i.e. use of alternative part of the same plant is likely to be much better accepted by the patients of a traditional healer. To put this policy into practice, there is a great need to evaluate the differences and similarities between various parts of the same plant concerning chemical composition and pharmacological properties. Only a few publications covering the aspects of plant part substitution have been published during the last fifteen years 1 - 7.
B. lanzan Spreng. (Anacardiaceae) commonly known as Priyala is widely used the tree in Ayurveda. As per Ayurvedic literature, stem bark of this plant is used in Jvara (fever), Raktapitta (hemorrhage), Trsa (thirst) and Rakatisara (dysentery) 8. The stem bark is also reported for various pharmacological activities like geno-toxicity, oxidative stress 9, antioxidant 10, 11, 12, anti-microbial 11. Stem bark mainly contains alkaloids, tannins, saponins, reducing sugars, triterpenoids and flavonoids 8. Removal of stem bark from the trunk of this tree may make this plant weak and susceptible to damage by insects and natural elements due to which availability of this plant may be difficult in the near future for use in Indian system of medicine.
Hence, in this study possibilities of using small branches of B. lanzan in place of its stem bark were evaluated with the aim of the conservation and sustainable use of this plant in medicine. Standard physicochemical parameters of small branches of B. lanzan have not been worked out yet, so work was also carried out to establish preliminary physicochemical standards of small branches.
MATERIAL AND METHODS:
Plant Material: Stem bark and small branches of B. lanzan Spreng. were collected from Jabalpur (Madhya Pradesh), India. Plant material was identified and authenticated by a botanist of the Institute, and a voucher specimen (A.No.10108) was deposited in Institute.
Instrumentation: CAMAG HPTLC system (Muttenz, Switzerland) equipped with semi-automatic TLC applicator Linomat IV, twin trough plate development chamber, Win CATS software version 1.4.2 and Hamilton (Reno, Nevada, USA) Syringe (100 μl).
Material and Reagents: All chemicals, reagents, and solvents used during the experiments were of analytical grade, and HPTLC plates were pur-chased from E. Merck Pvt. Ltd., (Mumbai, India).
Physicochemical Parameters: Stem bark and small branches were studied for various physicochemical standards like foreign matter, loss on drying at 105 °C, total ash, insoluble acid ash, alcohol-soluble extractive, water-soluble extractive and pH of 10% aqueous solution using standard methods 13, 14.
Preliminary Phytochemical Screening: n-hexane, ethyl acetate and ethanol extract of both stem bark and small branches were screened for the presence of phenols, tannins, carbohydrates, saponins, amino acids, alkaloids, flavonoids, steroids, coumarins, quinone, furanoids, and terpenoids by the standard methods 15, 16.
Estimation of Total Phenolic and Flavonoid Content: Five grams of each of shade-dried plant material was pulverized into coarse powder and subjected to ethanolic extraction using Soxhlet apparatus.
Extracts were concentrated to dryness. Dried residues were then dissolved in 100 ml of 95% ethanol. Extracts were used for total phenolic and flavonoid assay. Total phenolics content was determined by using Folin-ciocalteu assay 17. An aliquot (1 ml) of extracts or standard solution of gallic acid (20, 40, 60, 80 and 100 μg/ml) was added to a 25 ml volumetric flask, containing 9 ml of distilled water. A reagent blank was prepared using distilled water. One milliliter of Folin-ciocalteu phenol reagent was added to the mixture and shaken. After 5 min, 10 ml of 7% Na2CO3 solution was added to the mixture. Volume was then made up to the mark. After incubation for 90 min at room temperature, absorbance against reagent blank was determined at 550 nm with a UV/Vis spectrophotometer. Total phenolics content was expressed as mg gallic acid equivalents (GAE) per gm of dry weight.
Total flavonoid content was measured by aluminum chloride colorimetric assay 18. An aliquot (1 ml) of extracts or standard solutions of quercetin (20, 40, 60, 80 and 100 μg/ml) was added to a 10 ml volumetric flask containing 4 ml of distilled water. To the flask, 0.3 ml of 5% NaNO2 was added, and after 5 min, 0.3 ml of 10% AlCl3 was added. After 5 min, 2 ml of 1M NaOH was added, and the volume was made up to 10 ml with distilled water. The solution was mixed, and absorbance was measured against the blank at 510 nm. Total flavonoid content was expressed as mg quercetin equivalents (QUE) per gm of dry weight.
HPTLC Profiles: HPTLC studies were carried out by following the methods of Sethi 19, Stahl 20 and Wagner et al., 21. Stem bark and small branches were powdered coarsely. Ten-gram powdered samples of each of stem bark and small branches were accurately weighed and exhaustively extracted by n-hexane, ethyl acetate and ethanol (each 100 ml) separately using Soxhlet apparatus. Extracts were filtered and concentrated under reduced pressure and made up to10 ml in standard flasks separately. The mobile phase used for developing the n-hexane, ethyl acetate and ethanol extracts of stem bark and small branches was toluene: ethyl acetate (7:3 v/v). Samples were spotted in the form of bands of width 10 mm with a 100 μl Hamilton syringe on aluminum TLC plates precoated with silica gel 60 F254 of 0.2 mm thickness with the help of TLC semi-automatic applicator Linomat IV attached to CAMAG HPTLC system, which was programmed through Win CATS software version 1.4.2. 10 μl of each extract of stem bark and small branches were applied in two tracks as 10 mm bands at a spraying rate of 10 sec/μl. Track 1 was stem bark, and track 2 was small branches for each of extracts applied.
Development of plate up to a migration distance of 80 mm was performed at 27 ± 2 °C with mobile phase for each extract in a CAMAG HPTLC chamber previously saturated for 30 min. After development, the plate was dried at 60 °C in an oven for 5 min and visualized under wavelength 254 nm and 366 nm for ultraviolet detection. Developed plate was then dipped in anisaldehyde sulphuric acid reagent for derivatization and dried at 105 °C in hot air oven till the color of the band appears and visualized under white light. Images were captured by keeping plates in photo documentation chamber and Rf values were recorded by Win CATS software.
RESULTS: Physico-chemical parameters like foreign matter, loss on drying at 105 ºC, ash values, acid insoluble ash, extractive values, and pH are given in Table 1. These data may be useful to pharmaceutical industries for the authentication and batch to batch consistency of the commercial samples.
TABLE 1: PHYSICO-CHEMICAL PARAMETERS OF STEM BARK AND SMALL BRANCHES OF B. LANZAN
| S. no. | Parameters | Results | |
| Stem bark | Small branches | ||
| 1 | Foreign matter (% w/w) | Nil | Nil |
| 2 | Loss on drying (% w/w) | 7.98 | 6.91 |
| 3 | Total ash (% w/w) | 14.47 | 12.71 |
| 4 | Acid insoluble ash (% w/w) | 0.83 | 0.40 |
| 5 | Alcohol soluble extractive value (% w/w) | 22.32 | 11.72 |
| 6 | Water soluble extractive value (% w/w) | 22.18 | 9.42 |
| 7 | pH of 10 % aqueous solution | 5.06 | 5.01 |
Preliminary phytochemical analysis of different extracts of stem bark and small branches are shown in Table 2. Results reveal the presence of similar phytochemicals in n-hexane and ethanol extracts of stem bark and small branches. In ethyl acetate extract phenols, tannins and terpenoids were found present only in stem bark. The total amount of phenolics and flavonoids content of ethanolic extract of stem bark and small branches of B. lanzan are summarized in Table 3. Results indicate that in comparison to small branches, stem bark had high total phenolic and flavonoid contents.
TABLE 2: PHYTOCHEMICAL ANALYSIS OF EXTRACTS OF STEM BARK AND SMALL BRANCHES OF B. LANZAN
| Phytochemicals | Stem bark | Small branches | ||||
| n-hexane | Ethyl acetate | Ethanol | n-hexane | Ethyl acetate | Ethanol | |
| Phenols | -ve | +ve | +ve | -ve | -ve | +ve |
| Tannins | -ve | +ve | +ve | -ve | -ve | +ve |
| Alkaloids | +ve | +ve | +ve | +ve | +ve | +ve |
| Carbohydrates | +ve | +ve | +ve | +ve | +ve | +ve |
| Saponins | -ve | -ve | +ve | -ve | -ve | +ve |
| Amino acids | -ve | -ve | -ve | -ve | -ve | -ve |
| Steroids | +ve | +ve | +ve | +ve | +ve | +ve |
| Flavonoids | -ve | +ve | +ve | -ve | +ve | +ve |
| Coumarins | -ve | -ve | -ve | -ve | -ve | -ve |
| Quinone | -ve | +ve | +ve | -ve | +ve | +ve |
| Furanoids | -ve | +ve | +ve | -ve | +ve | +ve |
| Terpenoids | -ve | +ve | +ve | -ve | -ve | +ve |
(+ve) = Present, (-ve) = Negative
TABLE 3: TOTAL PHENOLIC AND TOTAL FLAVONOID CONTENT OF ETHANOLIC EXTRACTS OF STEM BARK AND SMALL BRANCHES OF B. LANZAN
| S.
no. |
Plant
parts |
Total phenolics
mg of GAE/ g dry weight* |
Total flavonoids
mg of QUE/ g dry weight* |
| 1 | Stem bark | 133.23 ± 1.77 | 39.73 ± 0.78 |
| 2 | Small branches | 66.26 ± 1.54 | 22.45 ± 1.18 |
*Values are expressed as Mean ± SD
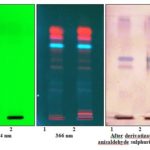
HPTLC profile of n-hexane extract of stem bark and small branches Table 4 and Fig. 3 showed no band and two bands, respectively when visualized under UV at 254 nm. At UV 366 nm stem bark and small branches showed two and four bands, respectively out of which one band at Rf 0.64 (fluorescent blue) was found similar.
Visualization under white light after derivatization with anisaldehyde sulphuric acid reagent, stem bark, and small branches showed three and five bands, respectively and all the three bands at Rf 0.43 (purple), 0.52 (blue), 0.87 (blue) of stem bark were found present in small branches.
FIG. 3: HPTLC PROFILE OF n-HEXANE EXTRACTS OF STEM BARK AND SMALL BRANCHES OF B. LANZAN. (TRACK 1: STEM BARK, TRACK 2: SMALL BRANCHES)
TABLE 4: Rf VALUE OF n-HEXANE EXTRACT OF B. LANZAN
| S.
no. |
Wavelength | Rf value | |
| Stem bark | Small branches | ||
| 1 | 254 nm | No band | 0.66, 0.81 |
| 2 | 366 nm | 0.64, 0.69 | 0.64, 0.71, 0.76, 0.82 |
| 3 | Visible light after derivatization | 0.43, 0.52, 0.87 | 0.43, 0.52, 0.64, 0.79, 0.87 |
HPTLC profile of ethyl acetate extract of stem bark and small branches Table 5 and Fig. 4 showed two and one bands, respectively at UV at 254 nm and band at Rf 0.70 was found common in both the parts. At UV 366 nm stem bark and small branches showed nine and seven bands, respectively out of which five bands at Rf 0.07 (red), 0.66 (fluorescent blue), 0.72 (blue), 0.75 (red), 0.80 (red) were found similar. Visualization under white light after derivatization with anisaldehyde sulphuric acid reagent stem bark and small branches showed six and four bands, respectively, out of which three bands at Rf 0.07 (blue), 0.54 (blue), 0.88 (blue) were found similar in both the parts.
FIG. 4: HPTLC PROFILE OF ETHYL ACETATE EXTRACTS OF STEM BARK AND SMALL BRANCHES OF B. LANZAN. (TRACK 1: STEM BARK, TRACK 2: SMALL BRANCHES)
TABLE 5: Rf VALUE OF ETHYL ACETATE EXTRACT OF B. LANZAN
| S. no. | Wavelength | Rf value | |
| Stem bark | Small branches | ||
| 1 | 254 nm | 0.26, 0.70 | 0.70 |
| 2 | 366 nm | 0.07, 0.11, 0.24, 0.32, 0.54, 0.66, 0.72, 0.75, 0.80 | 0.07, 0.45, 0.54, 0.66, 0.72, 0.75, 0.80 |
| 3 | Visible light after derivatization | 0.07, 0.26, 0.49, 0.54, 0.80, 0.88 | 0.07, 0.46, 0.54, 0.88 |
HPTLC profile of ethanol extract of both stem bark and small branches Table 6 and Fig. 5 showed no band when visualized under UV at 254 nm. At UV 366 nm, both stem bark and small branches showed ten bands and all were found similar.
TABLE 6: RF VALUE OF ETHANOL EXTRACT OF B. LANZAN
| S. no. | Wavelength | Rf value | |
| Stem bark | Small branches | ||
| 1 | 254 nm | No band | No band |
| 2 | 366 nm | 0.06, 0.27, 0.34, 0.47, 0.54, 0.64, 0.67, 0.69, 0.75, 0.82 | 0.06, 0.27, 0.34, 0.47, 0.54, 0.64, 0.67, 0.69, 0.75, 0.82 |
| 3 | Visible light after derivatization | 0.49, 0.56, 0.94 | 0.49, 0.56, 0.94 |
FIG. 5: HPTLC PROFILE OF ETHANOL EXTRACTS OF STEM BARK AND SMALL BRANCHES OF B. LANZAN. (TRACK 1: STEM BARK, TRACK 2: SMALL BRANCHES)
Visualization under white light after derivatization with anisaldehyde sulphuric acid reagent, both stem bark and small branches showed three bands, and again all were found similar.
DISCUSSION: Stem bark of B. lanzan Spreng. is an important ingredient in many Ayurvedic and other traditional formulations and considered to be very effective in various diseases. Ever increasing demand for this medicinal bark, commercialization, and overharvesting is creating a major threat to this species. Removal of bark from the tree may damage phloem or expose it to desiccation and fungal or parasite attack. Damage of phloem may disrupt the conduction of nutrients and hormones involved in flower bud production 22, 23 decreasing flower induction and therefore fruit and seed set. Studies have shown that overharvesting of bark can reduce fruit production and threaten reproduction 24. This may affect the survival of plant due to which this plant may be difficult to available shortly for use in traditional medicine industry. Therefore, serious concern regarding the conservation and protection of this plant has become a matter of urgency. Given this, a comparative phytochemical study was carried out to evaluate the possibility of using small branches of B. lanzan in place of its stem bark. The study is based on the concept of plant part substitution and will help sustainable utilization.
Data of physicochemical parameters Table 1 may be useful to traditional medicine industries for authentication and batch to batch consistency of commercial samples. Many similarities in the results for the qualitative phytochemical analysis of various extracts of stem bark and small branches of B. lanzan Table 2 indicate the presence of many analogous compounds in both the parts of this plant.
HPTLC profile can be used as a diagnostic tool for the proper identification of constituents in medicinal plants. Comparative evaluation of HPTLC profiles of n-hexane, ethyl acetate and ethanol extracts of stem bark and small branches of B. lanzan carried out to reveal the chemical pattern showed many similar bands which again indicates the presence of many similar compounds in stem bark and small branches of B. lanzan. A careful look at the results of both phytochemical analysis Table 2 as well as HPTLC profiles of various extracts of stem bark and small branches Table 4-6, Fig. 3-5 indicate that the stem bark and small branches are comparable chemically and may be investigated in detail for use as a substitute of stem bark based on chem profile and pharmacological action of compared plant parts. Less quantity of active phytochemicals like total phenolics and total flavonoids in small branches Table 3 may be compensated by using more quantity of small branches in formulations because this approach may satisfy the requirements of sustainable harvesting, yet simultaneously provide the drug for primary health care needs.
The above results may be of great importance for a scientist because physicians may substitute the parts of the same plant they traditionally use. The study provides a base for the possibilities of plant part substitution in this plant. Phytochemical investigation can never substitute pharmacological investigations in determining the therapeutic value of the plant material, so further investigation on comparison and confirmation of above study for pharmacological activities on these aspects is needed to support the findings and coming to a conclusion.
Results of qualitative evaluation of HPTLC profiles will also be helpful in the identification and quality control of the drug and can provide standard HPTLC profiles with the selected solvent system. This study is an initiative to add solution inputs to global concern for the management of traditional medicinal plant resources which nowadays has become a matter of urgency.
CONCLUSION: Similarities in HPTLC profiles and phytochemical analysis of various extracts of stem bark and small branches of B. lanzan suggests that small branches may have many similar active constituents like stem bark and may be recommended as a substitute of stem bark after comparison and confirmation of same for pharmacological activities. The study provides the base for further study covering an aspect of pharmacology and toxicity to use small branches in place of stem bark which can save the plant from destruction. Plant part substitution is the need of the hour for the conservation of medicinal plants as many medicinal plants are becoming red listed. It can provide greater scope for the physicians to utilize raw drugs that are easily available, cost-effective and most appropriate for the clinical conditions.
We, therefore, suggest that every investigation on underground parts or stem bark of medicinal plant may include an investigation on aerial parts of the plant also, even though those might not be the parts traditionally used. Investigations like this, may protect more species from extinction, and allow the recovery of threatened medicinal plants.
ACKNOWLEDGEMENT: Authors are thankful to CCRAS, New Delhi for financial assistance and Director General, CCRAS, New Delhi for providing facilities.
CONFLICT OF INTEREST: Nil
REFERENCES:
- Zschocke S, Rabe T, Taylor JLS, Jäger AK, and Van SJ: Plant part substitution a way to conserve endangered medicinal plants. Journal of Ethno Pharmacology 2000; 71(1-2): 281-92.
- Sulaiman CT and Balachandran I: Plant part substitution for medicinal use in Aegle marmelos a phytochemical approach. Journal of Tropical Medicinal Plants 2013; 14(1/2): 19-22.
- Srivastava B, Sharma VC, Pant P, Pandey NK and Jadhav AD: Evaluation for substitution of stem bark with small branches of Myrica esculenta for medicinal use- A comparative phytochemical study. Journal of Ayurveda and Integrative Medicine 2016; 7: 218-23.
- Srivastava B, Sharma VC, Singh R, Pant P and Jadhav AD: Substitution of roots with small branches of Rauwolfia serpentina for therapeutic uses a phytochemical approach. Ayushdhara 2015; 2(6): 373-8.
- Srivastava B, Sharma VC, Vashisth E, Singh R, Pandey NK and Jadhav AD: Comparative physicochemical, phytochemical and HPTLC study of stem bark versus small branches of Flacourtia indica. Journal of Scientific and Innovative Research 2015; 4(6): 227-31.
- Srivastava B, Sharma VC, Sharma H, Pant P and Jadhav AD: Comparative physicochemical, phytochemical and high-performance thin layer chromatography evaluation of heartwood and small branches of Aquilaria agallocha International Journal of Ayurveda Pharma Research 2016; 4(1): 1-6.
- Srivastava B, Sharma VC, Singh A, Singh R and Jadhav AD: Evaluation for substitution of heartwood with small branches of Acacia catechu for therapeutic use - A comparative phytochemical approach. Journal of Pharmacognosy and Phytochemistry 2016; 5(4): 254-8.
- The Ayurvedic Pharmacopoeia of India, Ministry of Health and Family Welfare, Department of Indian System of Medicine and Homeopathy, Government of India, New Delhi, Edition 1st, Part I, Vol. IV, 2008.
- Jain R and Jain SK: Effect of Buchanania lanzan Bark extract on cyclophosphamide-induced genotoxicity and oxidative stress in mice. Asian Pacific Journal of Tropical Medicine 2012; 5(3): 187-91.
- Kumari A and Kakkar P: Screening of antioxidant potential of selected barks of Indian medicinal plants by multiple in-vitro Biomedical and Environmental Sciences 2008; 21(1): 24-9.
- Venkata SP, Murali MC, Kandra P, Sravani R and Akondi RB: Screening of antimicrobial and antioxidant potentials of Acacia caesia, Dillenia pentagyna and Buchanania lanzan from Maredumilli forest of India. Journal of Pharmacy Research 2012; 5(3): 1734-8.
- Mehta SK, Mukherjee S and Jaiprakash B: Comparative antioxidant activity studies of Buchanania lanzan methanolic extract. Biomedical Pharmacology Journal 2009; 2(2): 441-4.
- Mukherjee PK: Quality control of Herbal drugs. Business Horizons Pharmaceutical Publishers, New Delhi 2008.
- Indian Herbal Pharmacopoeia. Indian Drug Manufacturers Association. Mumbai: Indian Herbal Pharmacopoeia; Revised Edition, 2002.
- Harborne JB: Phytochemical methods. A guide to modern techniques of plant analysis. New York: Hall Chapman and Co; Edition 3rd, 1998.
- Kokate CK, Purohit AP and Gokhale SB: Pharmacognosy. Pune, India: Nirali Prakashan; Edition 39th, 2007.
- Singleton VL and Rossi Jr. JA: Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Amer Jour of Enol and Viticul 1965; 16: 144-58.
- Zhishen J, Mengcheng T and Jianming W: The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chemistry 1999; 64: 555-9.
- Sethi PD: High-Performance Thin Layer Chromatography. New Delhi: CBS Publishers and distributors; Student Edition, Vol. X, 1996.
- Stahl I: Thin Layer Chromatography. A laboratory handbook. Berlin: Springer - Verlag; 1969.
- Wagner H and Bladt S: Plant drug analysis a Thin Layer Chromatography. Germany, Atlas: Springer-Verlag; Edition 2nd, 1984.
- Primack RB: Relationship among flowers, fruits and seeds. Annual Review of Ecology and Systematics 1987; 18: 409-30.
- Mohr H and Schopfer P: Plant physiology. Berlin: Springer-Verlag; Edition 4th, 1995.
- Gaalensoue OG and Ticktin T: Impacts of bark and foliage harvest on Khaya senegis (Meliaceae) reproductive performance in Benin. Journal of Applied Ecology 2008; 45(1): 34-40.
How to cite this article:
Srivastava B, Singh R, Bharthi V, Meena AK and Prakash O: A comparative phytochemical approach for substitution of stem bark with small branches in Buchanania lanzan for medicinal use. Int J Pharmacognosy 2018; 5(10): 658-65. doi link: http://dx.doi.org/10.13040/ IJPSR.0975-8232.IJP.5(10).658-65.
This Journal licensed under a Creative Commons Attribution-Non-commercial-Share Alike 3.0 Unported License.
Article Information
3
658-665
682
691
English
IJP
B. Srivastava *, R. Singh, V. Bharthi, A. K. Meena and O. Prakash
Regional Ayurveda Research Institute for Drug Development, Gwalior, Madhya Pradesh, India.
bhavanakan@gmail.com
01 August 2018
26 September 2018
30 September 2018
10.13040/IJPSR.0975-8232.IJP.5(10).658-65
01 October 2018